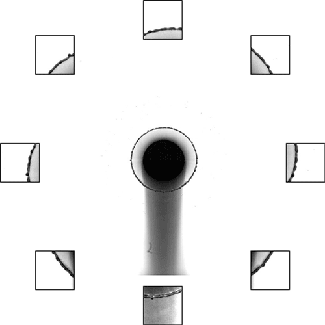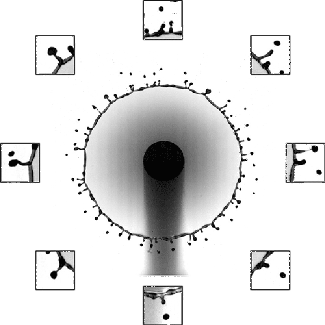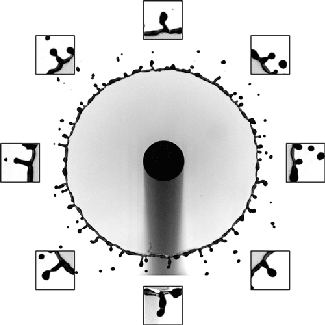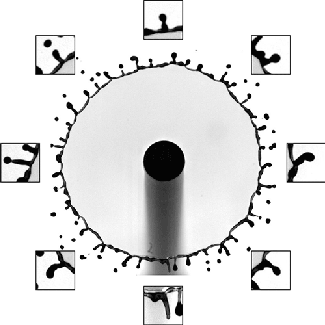
Sand and other granular materials can be strikingly fluid-like. Here the impact of a solid sphere on sand generates a splash remarkably similar to what’s seen with water. When the ball hits, it creates a crater in the surface and sends up a bowl-like spray of sand. As the ball continues falling through the sand, the grains try to fill the empty space left behind. The walls of sand collapsing around the void meet somewhere between the surface and the depth of the ball. This generates the tall jet we observe, as well as a second one under the surface that we can’t see. We know that collapse traps an air bubble under the surface because of the eruption that occurs as the jet falls. That’s the air bubble reaching the surface. (Image credit: T. Nguyen et al., source; see also R. Mikkelsen et al.)
Category: Research

Giving Droplets a Kick
Giving droplets a kick by accelerating the surface they sit on creates elaborate shapes as the drops respond. As the surface accelerates upward, the droplet flattens into a pancake. When the plate slows down, the droplet continues rising, stretching into a cone as its rim flies upward and its lower surface adheres to the surface. The rim retracts with a constant acceleration while the drop detaches with a constant velocity. That velocity depends on how well it adheres to the surface. The interplay between those two variables determines how conical or cylindrical the drop appears. See more in the full video below. (Image and video credit: P. Chantelot et al.)

Using Embolisms to Fight Cancer
Blocking blood vessels by creating embolisms is, under most circumstances, very bad. But researchers are exploring ways to fight cancer by intentionally and strategically creating these blockages. In gas embolotherapy, researchers inject fluid droplets, which can carry chemotherapy drugs, into the bloodstream. Once they circulate into a cancerous tumor, they use ultrasound to vaporize the droplet and create a gas bubble. Those bubbles lodge inside the capillaries of the tumor, starving it of fresh blood and trapping the chemotherapy drugs inside. It’s a one-two punch to the cancer. Without blood flow, the cancer cells die, and, since the cancer-killing drugs get mostly trapped inside the tumor, patients may require lower dosages and endure fewer side effects. The technique is currently in animal testing, but hopefully it will be a valuable therapy for human patients in the future. (Image credit: Chemical & Engineering News; research credit: Y. Feng et al.; via AIP)

Rim Break-Up
Splashing drops often expand into a liquid sheet and spray droplets from an unstable rim. Although this behavior is key to many natural and industrial processes, including disease transmission and printing, the physics of the rim formation and breakup has been difficult to unravel. But a new paper offers some exciting insight into this unsteady process.
The researchers found that if they carefully tracked the instantaneous, local acceleration and thickness of the rim, it always maintained a perfect balance between acceleration-induced forces and surface tension. That means that even though different points on the rim appear very different, there’s a universality to how they behave. They found that this rule held over a remarkably large range of situations, including across fluids of different viscosities and splashes on various surfaces. (Image and research credit: Y. Wang et al.; via MIT News; submitted by Kam-Yung Soh)

Turbulence and Star Formation
Space, as I’ve discussed previously, is surprisingly full of matter, especially clouds of dust. And yet the rate of star formation we observe is bizarrely low; the Milky Way, for example, produces only about one solar mass worth of new stars every year. If gravity were the sole force driving star formation, we’d see far more stars forming. Recent research suggests that turbulence plays a major role in regulating the star formation process, both by countering gravity’s attempts to collapse gases into a proto-star and by creating supersonic shocks that drive material together to jump-start star formation. There seem to be other important ingredients as well: young stars tend to form jets that blow material back into the interstellar clouds they’re forming in, feeding the turbulent background. For more, check out Physics Today. (Image credit: ESA/NASA/Hubble/ESO, via APOD; research credit: C. Federrath)

The Coexistence of Order and Chaos
One of the great challenges in fluid dynamics is understanding how order gives way to chaos. Initially smooth and laminar flows often become disordered and turbulent. This video explores that transition in a new way using sound. Here’s what’s going on.

The first segment of the video shows a flat surface covered in small particles that can be moved by the flow. Initially, that flow is moving in right to left, then it reverses directions. The main flow continues switching back and forth in direction. This reversal tends to provoke unstable behaviors, like the Tollmien-Schlichting waves called out at 0:53. Typically, these perturbations in the flow start out extremely small and are difficult or even impossible to see by eye. So researchers take photos of the particles you see here and analyze them digitally. In particular, they are looking for subtle patterns in the flow, like a tendency for particles to clump together with a consistent spacing, or wavelength, between them. Normally, researchers would study these patterns using graphs known as spectra, but that’s where this video does something different.
Instead of representing these subtle patterns graphically, the researchers transformed those spectra into sound. They mapped the visual data to four octaves of C-major, which means that you can now hear the turbulence. When the audio track shifts from a pure note to an unsteady warble, you’re hearing the subtle disturbances in the flow, even when they’re too small for your eye to pick out.

The last part of the video takes this technique and applies it to another flow. We again see a flat plate, but now it has a roughness element, like a tiny hockey puck, stuck to it. As the flow starts, we see and hear vortices form behind the roughness. Then a horseshoe-shaped vortex forms upstream of it. Aside from the area right around the roughness, this flow is still laminar. But then turbulence spreads from upstream, its fingers stretching left until it envelops the roughness element and its wake, making the music waver. (Video and image credit: P. Branson et al.)

Building Smart Swimmers
Scientists have long wondered whether the schooling of fish is driven by hydrodynamic benefits, but the complexity of their environment makes unraveling this complex motion difficult. A recent study uses a different tactic, combining direct numerical simulation of the fluid dynamics with techniques from artificial intelligence and machine learning to build and train autonomous, smart swimmers.
The authors use a technique called deep reinforcement learning to train the swimmers. Essentially, the swimmer being trained is able to observe a few variables, like its relative position to the lead swimmer and what its own last several actions have been – similar to the observations a real fish could make. During training, the lead swimmer keeps a steady pace and position, and the follower, through trial and error, learns how to follow the leader in such a way that it maximizes its reward. That reward is set by the researchers; in this case, one set of fish was rewarded for keeping a set distance from their leader, one intended to keep them in a position that was usually beneficial hydrodynamically. Another set of fish was rewarded for finding the most energy-efficient method for following.
Once trained, the smart swimmers were set loose behind a leader able to make random decisions. Above you can see the efficiency-seeker chasing this leader. Impressively, even though this smart swimmer had the option to go it alone (and had never followed such a dynamic leader), it does an excellent job of keeping to the leader’s wake. Compare it with real swimmers and there’s a definite similarity in their behavior, which suggests the technique may be capturing some of an actual fish’s intuition. (Image and research credit: S. Verma et al., source; thanks to Mark W. for assistance)

Bouncing Off a Moving Wall
There are many ways to repel droplets from a surface: water droplets will bounce off superhydrophobic surfaces due to their nanoscale structures; a vibrating liquid pool can keep droplets bouncing thanks to its deformation and a thin air layer trapped under the drop; and heated surfaces can repel droplets with the Leidenfrost effect by vaporizing a layer of liquid beneath the droplet. But all of these methods will only work for certain liquids under specific circumstances.
More recently, researchers have begun looking at a different way to repel droplets: moving the surface. The motion of the plate drags a layer of air with it; how thick that layer of air is depends on the plate’s speed. (Faster plates make thinner air layers.) Above a critical plate speed, a falling droplet will impact without touching the plate directly and will rebound completely. This works for many kinds of liquids – the researchers used silicone oil, water, and ethanol – across many droplet sizes and speeds. The key is that the air dragged by the plate deforms the droplet and creates a lift force. If that lift force is greater than the inertia of the droplet, it bounces. (Image and research credit: A. Gauthier et al., source)

Using Air to Break Up Jets
One method of breaking a liquid into droplets, or atomizing it, uses a slow liquid jet surrounded by an annulus of fast-moving gas. The gas along the outside of the liquid shears it, creating waves that the wind blowing past can amplify. This draws the liquid into thin ligaments that then break into droplets. This is a popular technique in rocket engines, where cryogenic liquid fuels often need to be atomized for efficient combustion. When things aren’t working exactly right, however, the liquid jet may start flapping instead of breaking up. In this case, the jet will swing back and forth, but only part of it will atomize. For a rocket engine, this would mean slower and less efficient combustion – never desirable outcomes! (Image credit: A. Delon et al.)

Dissolving Candy
In nature, solid surfaces often evolve over time in conjunction with the flows around them. This is how stalactites, canyons, and hoodoos all form and change over time. Here researchers examine a surface formed from hard candy that is dissolving from below. Over time, the initially flat surface develops a pitted appearance (top image, scale bar is 1 cm) with roughness that is approximately 1 mm in scale. Flow visualization (bottom row) suggests that these pits result from local flow where narrow, millimeter-sized dense plumes fall away from the surface.
As material dissolves from the candy, it forms a dense layer of sugar-water mixture near the solid surface. Once that layer grows to a critical thickness, it will be too unstable for viscosity to counter. At that point, the Rayleigh-Taylor instability takes over, causing the dense sugar-water layer to break up into narrow, sinking plumes. Although each area is evolving independently, the rate at which material dissolves is uniform everywhere, so the dissolving body retains the same shape over time. (Image and research credit: M. Davies Wykes et al., source)