Hurricanes are a frequent and potentially deadly occurrence for many parts of the world. Although forecasting models have improved, there is still a lot about the physics of these storms that we don’t fully understand, in part because getting direct measurements from the real thing is so difficult and hazardous. Researchers at the University of Miami have instead built their own hurricane generator, capable of sustained 200 mph winds – strong enough to create Category 5 hurricane conditions. In this facility, they can study details of the storm up close, allowing them to distinguish effects from the scale of large waves down to the physics of the sea spray. Learn more and see the facility in action in the Science Friday video below. (Video credit: L. Groskin/Science Friday; image credits: L. Groskin/Science Friday, University of Miami, SUSTAIN Lab; submitted by Guillaume D.)
Category: Research

Soap Film Catenoid
Even very simple fluid systems can have surprising complexity. What you see here is a catenoid – the hourglass-like soap film that forms between two rings. In this case, the space in the center of the catenoid has a secondary film separating the top and bottom halves of the catenoid. When the rings are pulled apart, the waist of the catenoid and the secondary film inside it collapse. The secondary film gets thicker as its diameter decreases. (The fluid has to go somewhere, after all.) As the film thickens, the pressure inside it rises, eventually pushing some of the fluid out through the catenoid. This is what causes the fingers flowing down the lower half of the catenoid in the bottom two images. (Image and research credit: R. Goldstein et al.)

The Catherine Wheel
When particles of different sizes fall in an avalanche, they separate out by size. Smaller particles form one layer with another layer of larger particles over the top. This happens because the smaller particles tend to fall in between the larger ones, similar to the percolation theory in the Brazil nut effect. In a slowly rotating drum, this size segregation during an avalanche forms a distinctive pattern (above) called a Catherine wheel pattern. Here, the gray layers form from smaller iron particles, while the white layers are large particles of sugar. Notice that the pattern starts to form during each avalanche, but it freezes in place after grains pile up against the drum wall and cause a shock wave to run back up the avalanche. (Image credit: J. Gray and V. Chugunov, reprinted in J. Gray, source)

Hydrofoils and Stability
Today’s fastest boats use hydrofoils to lift most of a boat’s hull out of the water. This greatly reduces the drag a boat experiences, but it can also make the boat difficult to handle. One style of hydrofoil boat, called a single-track hydrofoil, uses two hydrofoils in line with one another to support and steer the boat. The pilot can steer the lead hydrofoil into the direction of a fall to correct it. Stability-wise, this is the same way that you keep a bicycle upright. On a boat, the situation is a bit tougher to manage, and, like riding a bike, it takes practice. A group of students published a full mathematical model for the dynamics of this kind of boat, which allows designers to test a prototype’s stability early in the design process and enables student teams to use computer simulators to train their pilots to drive a boat before putting them out on the water, similar to the way that airplane pilots train. (Image credit: TU Delft Solar Boat Team, source; research credit: G. van Marrewijk et al., pdf; via TU Delft News; submitted by Marc A.)

Bubble Trains in a Microchannel
Trains of bubbles flowing through a microchannel get distorted by periodic expansions and constrictions. In these images, flow is from left to right, and the narrow point of the channel is about 250 microns across. In narrow regions, the front of the bubble tends to move faster, while in wider areas, the back of the bubble speeds up. This causes the distinctive shape changes we see. Microfluidic channels with these exaggerated shifts in geometry allow researchers to study the physics behind liquids and gases seeping through the interstitial gaps of a porous media, like when water and gases move through rock and soil. (Image and research credit: M. Sauzade and T. Cubaud)

The Disintegrating Splash
A drop of blue-dyed glycerine impacts a thin film of isopropanol, creating a spectacular splash and breakup. The drop’s impact flings a layer of the isopropanol into the air, where air currents make the thin sheet buckle inward and break into a spray of droplets. Meanwhile, the liquid from the drop forms a thick, blue crown that rises and expands outward. When tiny droplets of the isopropanol hit the splash crown, their lower surface tension causes the blue glycerine to pull away, due to the Marangoni effect. This opens up holes in the crown, which grow quickly, until the entire sheet breaks apart. (Image and research credit: A. Aljedaani et al., source)

Snowmelt
Much of the rain that falls on Earth began as snow high in the atmosphere. As it falls through warmer layers of air, the snowflakes melt and form water droplets. The details of this melting process have been difficult to capture experimentally, but a new computational model may provide insight. The basic process has a couple stages. As snow begins to melt, surface tension draws the water into concave areas nearby. When those regions fill up, the water flows out and merges with neighboring liquid, forming water droplets around a melting ice core.
Although this same sequence was observed for many types of snow, scientists also observed some important differences between rimed and unrimed snowflakes. Rime forms when supercooled water droplets freeze onto the surface of a snowflake. Lightly rimed snow still looks light and fluffy, like the animation above, but heavily rimed snow forms denser and more spherical chunks. Because there are lots of porous gaps in heavily rimed snow, water tends to gather there during initial melting. Rimed snow was also more likely to form one large water droplet rather than breaking into multiple droplets like snow with less rime. For more, check out NASA’s video and the Bad Astronomy write-up. (Image credit: NASA, source; research credit: J. Leinonen and A. von Lerber; via Bad Astronomy; submitted by Kam Yung-Soh)

Martian Mantle Convection
Over geological timescales – on the order of millions of years – even hard substances like rock can flow like a fluid. Heat from the Earth’s core drives convection inside our mantle, and that fluid motion ultimately drives the plate tectonics we experience here at the surface. But most other planetary bodies, including those with mantle convection similar to ours, don’t have a surface that shifts like our tectonic plates. Mars and Venus, for example, have solid, unmoving surfaces. The images above provide a peek at what goes on beneath. The upper image shows a simulation of mantle convection inside Mars over millions of years. The lower image is a timelapse of dye convecting through a layer of glucose syrup being heated from below. Notice how both examples show evidence of convective cells and plumes that help circulate warm fluid up and colder fluid downward. (Image credit: Mars simulation – C. Hüttig et al, source; N. Tosi et al., source; submitted by Nicola T.)

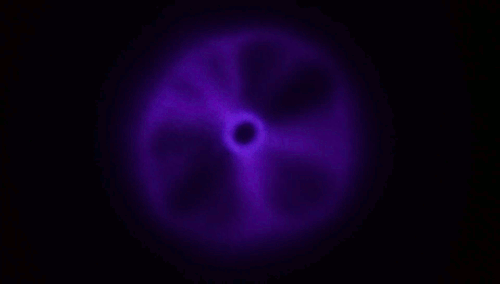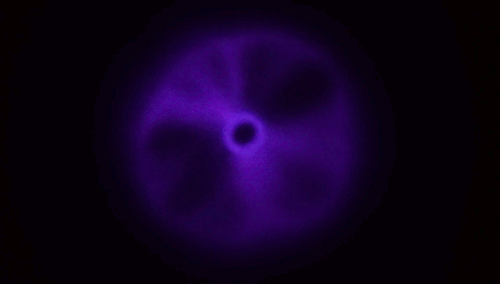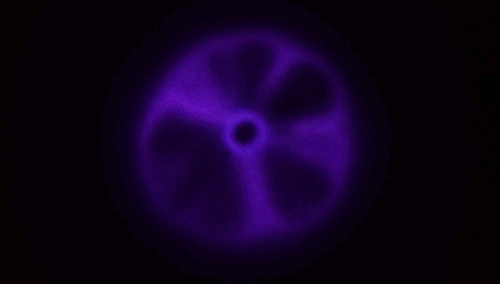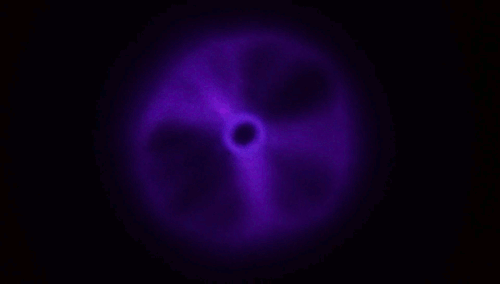
Plasma From a Jet of Water
There aren’t many naturally occurring plasmas in our daily lives; by far the most common one is lightning. So it’s something of a surprise that a stream of water hitting a material like glass is able to produce a toroid of plasma like the one above. The key here, though, is that the jet has to be fast – to the tune of 200 meters per second or faster. When a jet of deionized water strikes a surface at that speed, the water has to take a very sharp, 90-degree turn, and, thanks to the polar nature of water, this causes a (negative) charge to build up at that turn. It’s akin to rubbing a balloon to build up a static charge, and it’s known as a triboelectric effect. At rest (and without high shear rates), water and glass in contact tend to create in a positive charge in the water. The plasma is created when an arc forms through air between those two charged areas.
Experiments in helium environments create a different color of plasma, confirming that the arc definitely travels through the gas. Similarly, if you use regular water instead of deionized water, the conductivity of the dissolved salts in the water is enough to prevent the necessary build up of charge. (Image and research credit: M. Gharib et al.; video credit: Applied Science; submitted by Kam-Yung Soh)

Breaking Ground
Pushing a fluid into a porous granular material can fracture it into branching, lightning-like patterns. Here, air is injected into wet grains as a laboratory analog to hydrocarbon extraction or fracturing to treat contaminated soil. The injection of air compacts grains along the branch boundaries, keeping individual branches separated from one another. The patterns that form change with grain shape and ultimately result from the interactions of pressure, surface tension, friction and viscous forces. Studies like these help optimize fluid flow, decontaminate polluted soil faster, and determine risk in gas-driven fracturing of hydrocarbon reservoirs. (Image and video credit: J. Campbell et al.; submitted by B. Sandnes)