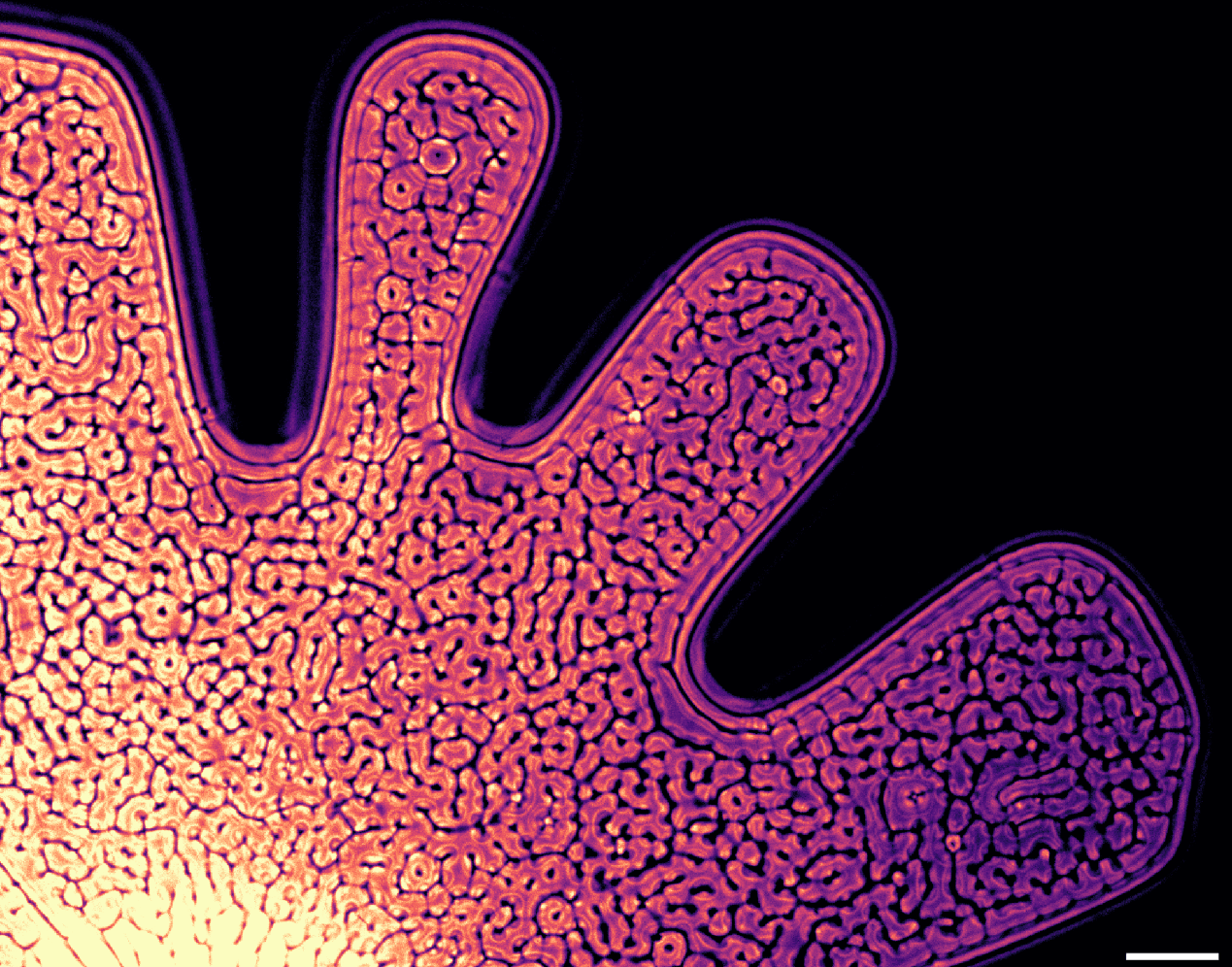
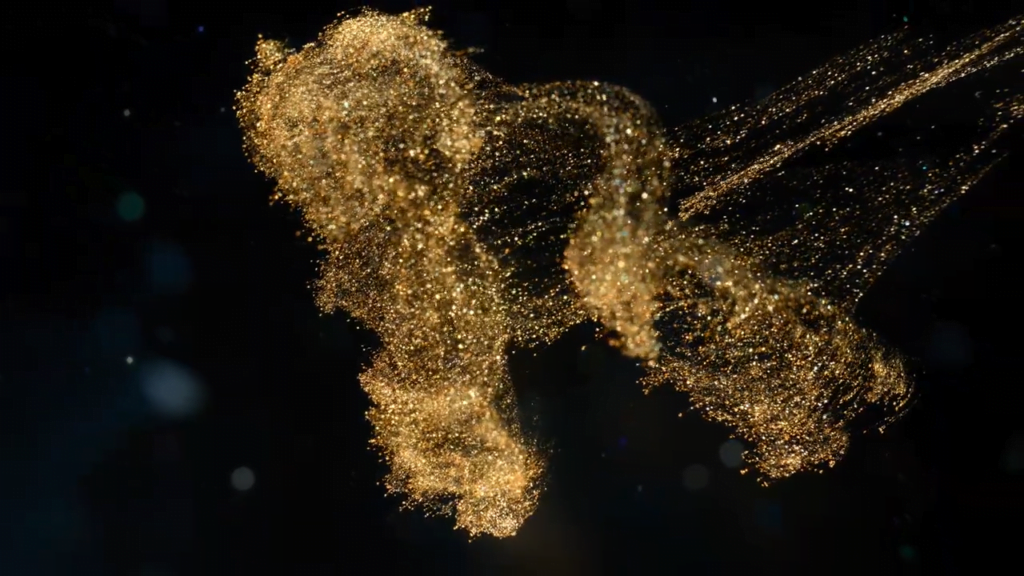Colorful chandeliers, passing spirits, sprouting mushrooms, and fountains of falling ink appear in Christopher Dormoy’s “Aquakosmos.” Driven by the slight density difference between ink and water, many of these elaborate shapes result from the Rayleigh-Taylor instability. Anytime you see mushroom-like plumes and chandelier-like splitting vortex rings, there’s probably a Rayleigh-Taylor instability behind it. Check out the full video above, and, if you want to give this kind of flow visualization a try yourself, a glass of water and vial of food coloring is a great place to start. (Video and image credit: C. Dormoy)
Tag: Rayleigh-Taylor instability

Instabilities on Instabilities
The world of fluid instabilities is a rich one. Combine fluids with differing viscosities, densities, or flow speeds and they’ll often break down in picturesque and predictable manners. Here, researchers explore the Rayleigh-Taylor instability (RTI), which occurs when a denser fluid sits above a less dense one (in a gravitational field). It’s an extremely common instability, showing up in both the cream in your ice coffee and the shape of a supernova’s explosion. It’s very difficult to set up and observe, though, which is where the real cleverness of this experiment stands out.
To study the RTI, these researchers first created another instability, the Saffman-Taylor instability. They filled the space between two glass plates with a viscous fluid, then injected a less viscous one. That created the distinctive viscous fingering pattern seen in the top image. In addition to being less viscous, the injected fluid was also less dense. As it pushed into the original fluid, it displaced some of it, creating a three-layer structure with dense fluid over less-dense fluid over dense fluid. That laid the groundwork for the Rayleigh-Taylor instability form.

A side-view through the fluid mixture shows the characteristic mushroom-like plume of the Rayleigh-Taylor instability. Check out the cell-like pattern distributed across the fluid in the top image. These are plumes formed in the RTI as dense fluid sinks into the less-dense fluid below it. From the side (see second image), each plume takes on the distinctive mushroom-like shape of a Rayleigh-Taylor instability. Given time, the two fluids mix and the cellular pattern disappears. But until then, this set-up uses one instability to study a second one. How cool is that?! (Image and research credit: S. Alqatari et al., see also)

“Turbulence”
In his recent short film, artist Roman De Giuli explores turbulence using metallic paints and inks in a fishtank. The effects are beautiful: sparkling pigments dispersing in clouds, mushroom- and umbrella-shaped Rayleigh-Taylor instabilities, and lots of swirling eddies. It’s exactly the kind of eyecandy to kick off your weekend with! (Image and video credit: R. De Giuli)

Aqueous Chandeliers
Colorful dyes falling through water form chandelier-like, branching shapes. These formations are the result of a slight density difference between the heavier dyes and the surrounding water. As the dye falls, Rayleigh-Taylor instabilities cause the mushroom-like blobs and their branches. With creativity and photographic skill, Mark Mawson turns these ephemeral shapes into bold liquid sculptures, frozen in time. See more of his work in these previous posts, on his website, and on Instagram. (Image credit: M. Mawson)

Precipitation
Chemistry and fluid dynamics often go hand-in-hand. Here chemical reactions produce visible precipitates as one chemical drops into the other. The shapes that form are distinctly fluid dynamical, with vortex rings, plumes, and instabilities all appearing.
In many applications, chemical reactions and fluid dynamics are tied inextricably to one another because the rate of chemical reaction depends on local concentrations driven by fluid dynamics, and the fluid motion is itself influenced by those concentration gradients. This is why reacting flows, like those found in combustion, are among the hardest topics in fluids. (Image and video credit: Beauty of Science)

Ghostly Chandeliers

Under a black light, highlighter fluid creates ghostly trails as it drips through water. The vortices that form and break into this chandelier-like shape are the result of density differences between the ink and water. Since ink is heavier than water, it sinks, but as the two fluids flow past, they shear one another, forming elaborate shapes. Formally, this is known as the Rayleigh-Taylor instability. While you may be most familiar with it from pouring cream into coffee, it’s also a key to mixing in the ocean and the explosions of supernovas. (Image credit: S. Adams et al.; via Flow Vis)

Mimicking Supernovas
The Hubble archives are full of incredible swirls of cosmic gas and dust, many of which were born in supernovas. Predicting the forms these massive explosions will generate is extremely difficult, thanks in large part to the complicated fluid dynamics generated by their blast waves. But new lab-scale experiments may help shed light on those underlying processes.
Researchers mimic supernovas in the lab by launching blast waves through an interface between a dense gas (shown in white) and a lighter one (which appears black). As the blast wave passes, it drives the dense fluid into the lighter one, triggering a series of instabilities. Notice how any initial perturbations in the interface quickly grow into mushroom-like spikes that rapidly become turbulent. This behavior is exactly what’s seen in supernovas (and in inertial confinement fusion)! (Video credit: Georgia Tech; research credit: B. Musci et al.; submitted by D. Ranjan)

“Magic Fluids”
In his short film, “Magic Fluids,” Roman De Giuli uses cyan, magenta, and yellow paints to generate a rainbow of macro colors. All the fluid motion you see is a practical effect, painstakingly created by layering paints and flow mediums of different densities. Like in Siqueiros’ “accidental painting” technique, the less dense paints will eventually rise through the upper layers and spread. De Giuli uses the effect for its motion, but the same physics is key for many artists who use acrylic pouring to paint. (Video and image credit: R. De Giuli)

Driving Instabilities with a Twist
Imagine that you want to study how two fluids mix when a lighter fluid is pushed into a denser one. Conceptually, it’s a straightforward situation. It would be like having a layer of oil under a layer of water and watching what happens. But how do you do that experimentally? Oil won’t naturally stay under water. If you flip the container over to start the experiment, you’ve added a bunch of extra motion from the rotation. And if you use a barrier to separate the two layers and then pull it out, you’ve added extra shear where they meet.
To deal with challenges like these, researchers at Lehigh University spent five years designing and building the rotating wheel apparatus you see in the video above. Instead of relying on gravity to force the lighter fluid into a denser one, this set-up uses centrifugal force. The test fluids start out on the loading wheel, spinning in their naturally stable configuration. Then once both sides are rotating at the desired speed, the track flips, transferring the experiment onto the other wheel, which rotates in the opposite sense. This gives the fluids a sudden change in the direction of the centrifugal force and, once the apparatus completes shake-down, should give us new insight into the sort of mixing seen in fusion. (Video credit: Lehigh University; see also Turbulent Flow Design Group)

Granular Instabilities
Granular mixtures show surprising similarities to fluids, even though their underlying physics differ. The latest example of this is a Rayleigh-Taylor-like instability that occurs when heavy particles sit atop lighter ones. By combining vertical vibration and an upward gas flow, researchers found that the lighter particles form fingers and bubbles that seep up between the heavier grains (upper left). Visually, it looks remarkably similar to a lava lamp or other Rayleigh-Taylor-driven instability (upper right).
But the physics behind the two are distinctly different. In the fluid, buoyancy drives the instability while surface tension acts as a stabilizing force. There’s no surface tension in a granular material, though. Instead, the drag force from gas flowing upward provides the vertical impetus while friction between the grains – essentially an effective viscosity – replaces surface tension as a stabilizing influence.
The similarities don’t stop there, though. When the researchers tested a “bubble” of heavy grains suspended in lighter ones (lower left), they found that, instead of sinking, the granular bubble split in two and drifted downward on a diagonal. Eventually, those daughter bubbles also split. Again, visually, this looks a lot like what happens to a drop of ink or food coloring falling through water (lower right), but the physics aren’t the same at all.
In the fluid, the breakup happens when a falling vortex ring splits. In the granular example, gas moving upward tends to channel around the heavy grains because they’re harder to move through. Eventually, this builds up a solidified region under the bubble. When the heavy grains can’t move directly down, they split and sink through the surrounding suspended particles until they build up another jammed area and have to split again. (Image credits: granular RTI – C. McLaren et al.; RTI simulation – M. Stock; bag instability – D. Zillis; research credit: C. McLaren et al.; submitted by Kam-Yung Soh)