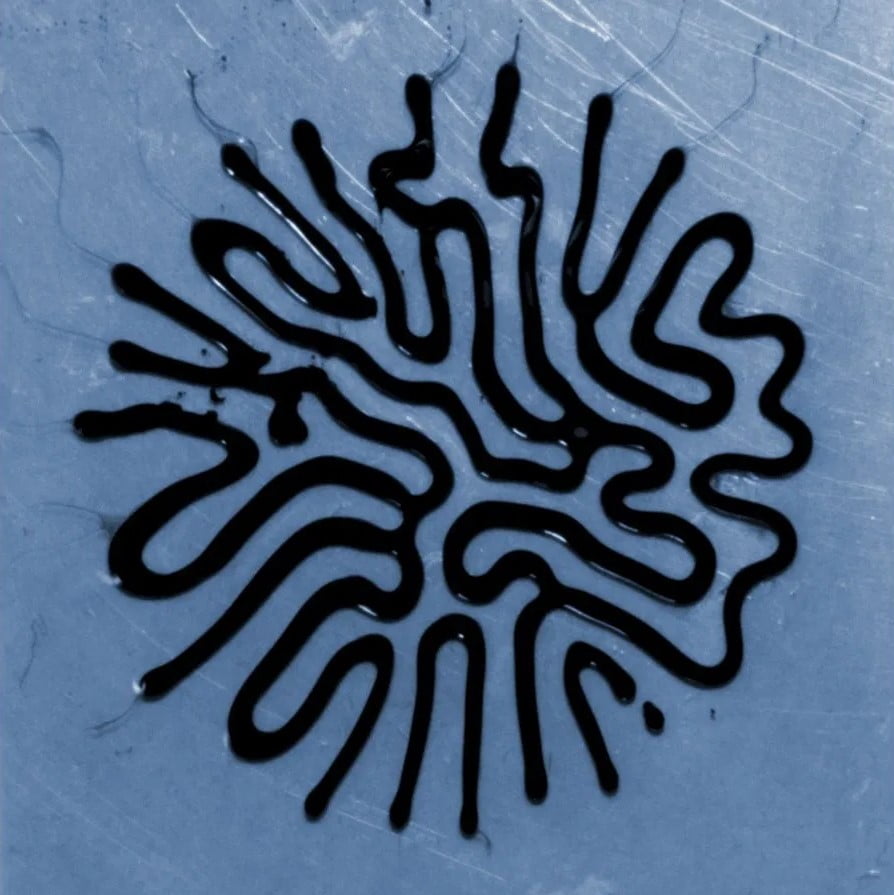
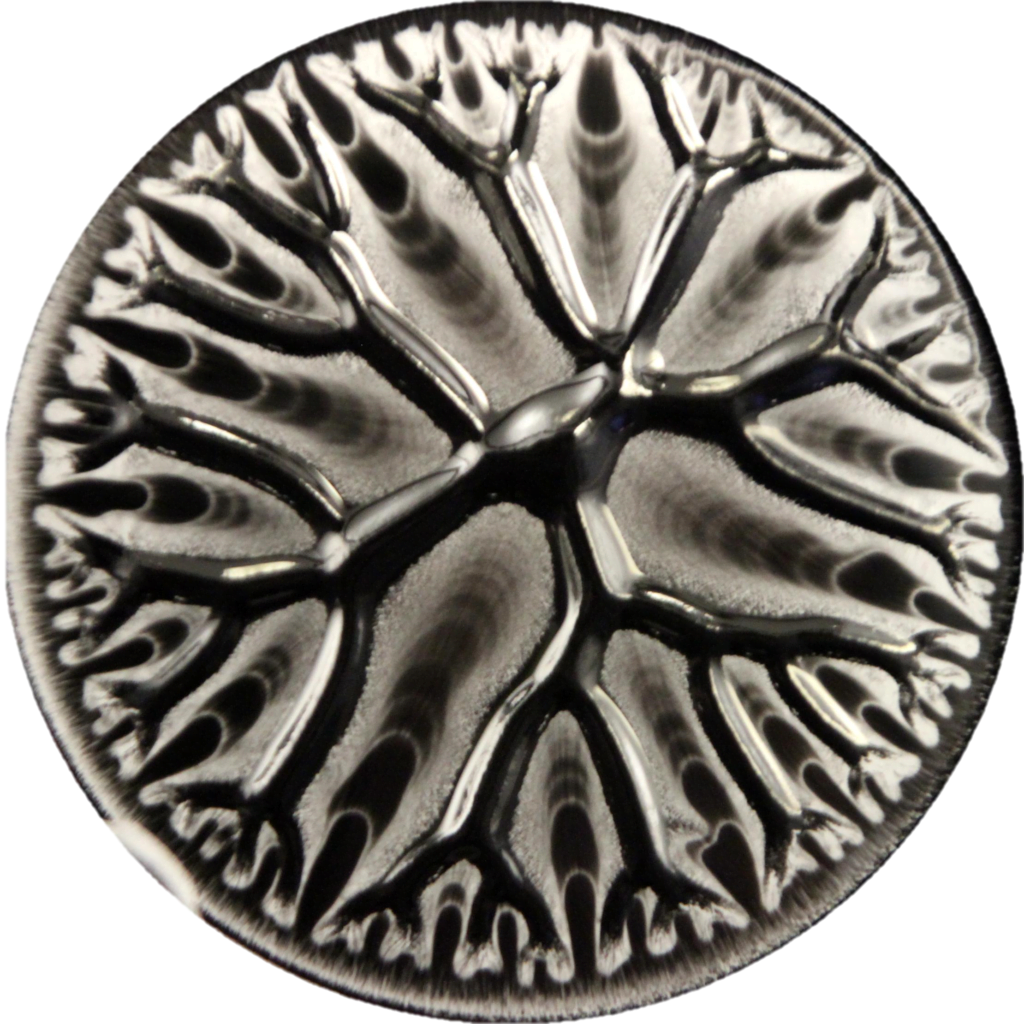
We’re used to seeing ferrofluids — with their suspended iron nanoparticles — as spiky fluids when exposed to a magnetic field. But this is not always the case. Here, the ferrofluid is immersed in a thin liquid layer — window cleaner, in this case — and when a magnet is brought near, it forms snake-like, labyrinthine lines. (Image credit: M. Carter et al.)
Tag: physics

Ultrasound in Medicine
When you hear the term “ultrasound,” your brain likely jumps to grainy black and white images of unborn babies, but this technology has a lot more medical uses than just that! Ultrasound is used to image many parts of the body — earlier this year, I got to see my own heart in action through an echocardiogram, for example. But the technology has therapeutic uses as well. At higher energies, ultrasound is used to break up kidney stones (through cavitation), treat tremors, and alleviate some sources of pain. To learn more, check out Explore Sound’s page on biomedical acoustics. (Video and image credit: Acoustical Society of America)

Branching Gels
If you sandwich a viscous fluid between two plates, then pull the plates apart, you’ll often get a complex branching pattern that forms as air pushes its way into the fluid. But the exact results depend strongly on what kind of viscous fluid you used. A new study looks specifically at what happens when that fluid is a yield-stress gel.
Yield-stress fluids behave like a solid until a critical amount of force causes them to flow. Think about your toothpaste. When you take the cap off, the toothpaste stays put until you squeeze the tube enough to make it flow. The gels used in this experiment behave similarly.
The researchers found that their gels required a critical energy input in order to branch and flow. If the energy applied in pulling the plates apart was too low, no branching occurred (Image 1). But beyond that critical energy, separating the plates created intricate branching patterns consistent with those seen in simpler, Newtonian fluids. (Image, research, and submission credit: T. Divoux et al.; via APS)

Speeding Sedimentation
Did you know that particles settle faster in an inclined container instead of a vertical one? This sedimentation phenomenon is known as the Boycott effect, after the researcher who first described it. Boycott noticed that red blood cells settled out of samples faster when the test tubes were inclined.
The inclined walls give particles a much larger area to settle on. As the particles gather on the wall, it creates a buoyant, particle-free layer of fluid above. That fluid quickly rises to the top of the container, helping to push the sediment further toward the bottom. As you can see in the video below, the Boycott effect drastically reduces settling time. (Video and image credit: C. Kalelkar)

Crystalline Critters
In 5th grade, I grew crystals by evaporating solutions of salt water from miniature pie tins. The results were white, boxy crystals whose size depended on how much salt I’d managed to dissolve into the water. But it turns out I could have gotten much cooler results if I’d evaporated my salt water a drop at a time on a hot superhydrophobic surface. That’s how these researchers formed the “crystal critters” shown in the video above.
Initially, the evaporating salt water drop is what we would expect, but once enough water is gone to leave a shell of salt, the drop grows legs and lifts off the surface. From that point, all growth occurs from the surface up. Because the surface is heated, evaporation happens quickest at that point of contact, and the water that remains is drawn down the legs, providing more fluid for evaporation as well as additional salt to grow the crystal. (Video, image, and research credit: S. McBride et al.)

Slow Mo Espresso
High-speed photography gives us an alternate glimpse of reality. Here it provides an all-new perspective on making espresso. Surface tension plays a starring role, first in pulling together the film that forms over the exit, then in creating the drips and drops that follow. The break-up of espresso into individual droplets is an example of the Plateau-Rayleigh instability, where surface tension drives any wobble in the falling jet to pinch off. For more slow-motion espresso, you can also check out this behind-the-scenes video. (Video and image credit: J. Hoffmann; submitted by Jerrod H.)

Fungal Fluid Dynamics
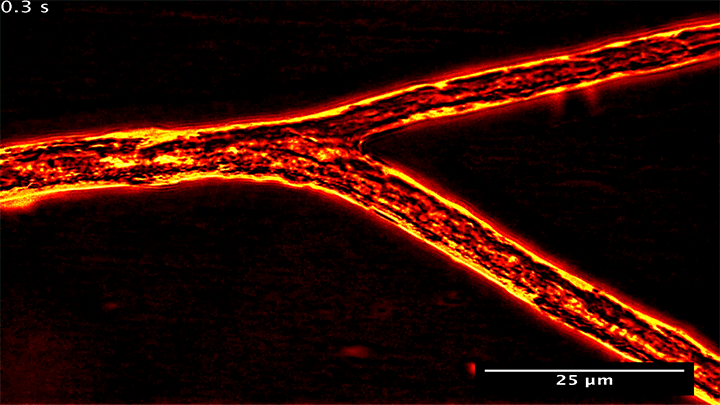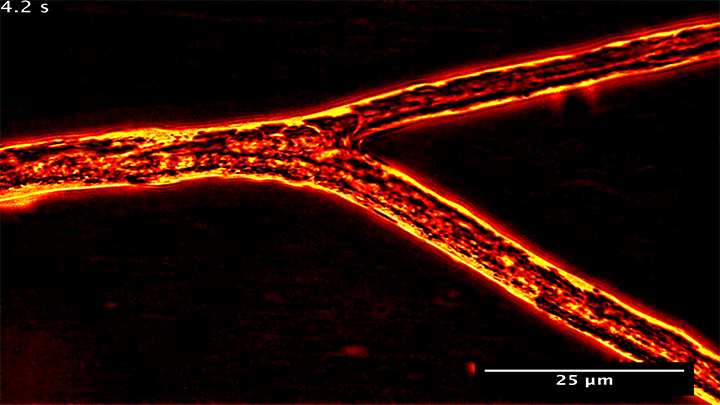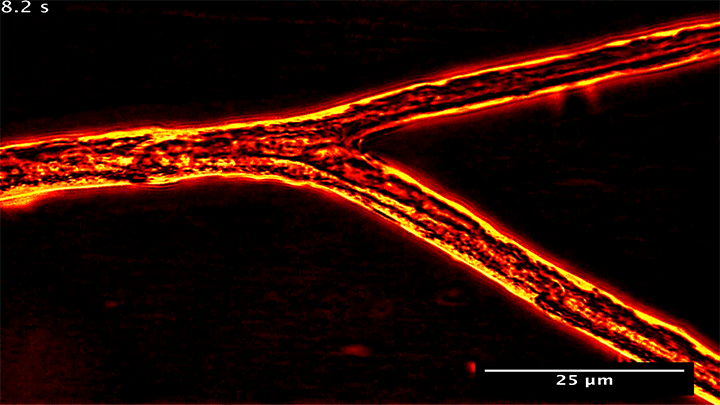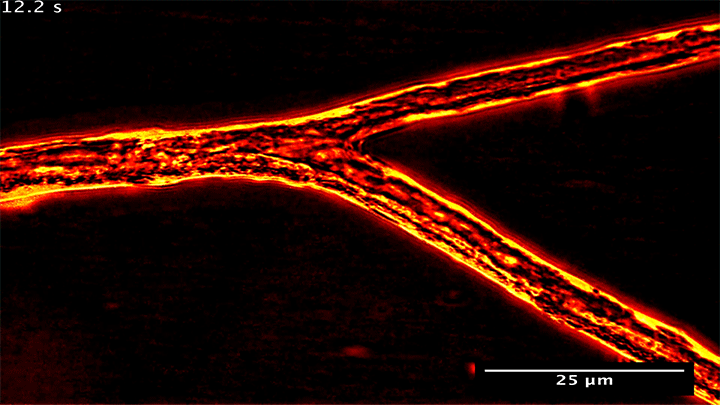
Many plants gain the soil-bound nutrients they need by trading with symbiotic fungi. Underground, these fungi spread networks that gather and store phosphorus, which they then trade with host plants to get the carbon they need. Research shows that the fungi can be shrewd traders, moving phosphorus from nutrient-rich areas to poorer ones in order to maximize their trade gains.
What you see above are snapshots of some of this transport within the fungal network. Notice how flow within the branching network changes direction. The fungus can force these flow reversals in a matter of seconds, allowing it to move nutrients to wherever the best returns are found. (Image and research credit: M. Whiteside et al.)

Sediment and Coral
As rivers wash sediment toward the sea, they carve elaborate deltas like that of the Rio Cauto in Cuba. Over time these sediments build up marshes, swamps, lagoons, and other wetlands that provide critical habitat and flood control. Sediment also washes into the bay, where it interacts with the coral reefs (light green lines on the lower left) and the species that live there. (Image credit: L. Dauphin/USGS; via NASA Earth Observatory)


Molten Thermite
This glowing, molten liquid captured by the Slow Mo Guys is thermite. The chemical reaction behind thermite is highly exothermic, hence its intense glow. There’s some great fluid dynamics hiding in this video. First, there’s the dripping thermite (Image 1), which breaks up into droplets via the Plateau-Rayleigh instability before shattering when it hits the ground.
Then there are the sequences (Images 2 and 3) of thermite dripping into water. The heat of the reacting thermite vaporizes a layer of water around it, creating a bubble that completely envelops the thermite. In other words, the falling thermite is supercavitating! That layer of air significantly reduces drag on the thermite and it insulates the thermite from the cooler temperature of the water. (Video and image credit: The Slow Mo Guys)

Droplets From Jets
On the ocean, countless crashing waves are creating bubbles. When they burst, those bubbles generate jets and droplets that spray into the sky, carrying sea salt, dust, and biological material into the atmosphere. Researchers know these droplets and their evaporation are important for understanding environmental processes, but figuring out how to capture that importance in models continues to be a challenge.
In a new study, researchers concentrated on a simplified problem: the bursting of a single bubble in pure water. By studying a wide range of conditions, the team found that jets from these bubbles could eject as many as 14 droplets apiece. And though existing models have mostly ignored all but the first droplet, their work showed that all of the droplets should be accounted for in any evaporation models. (Image credit: C. Couto; research credit: A. Berny et al.)