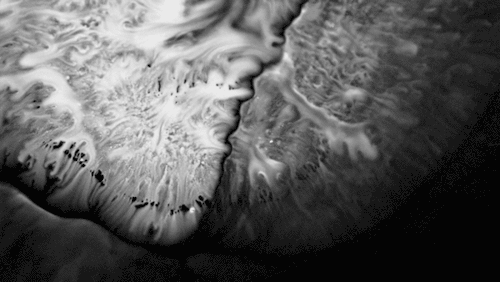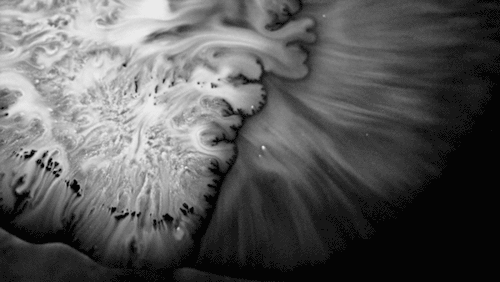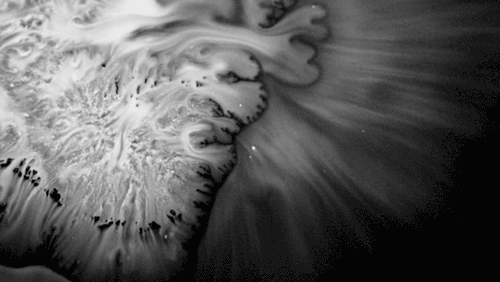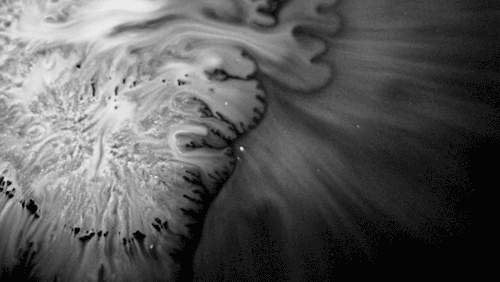
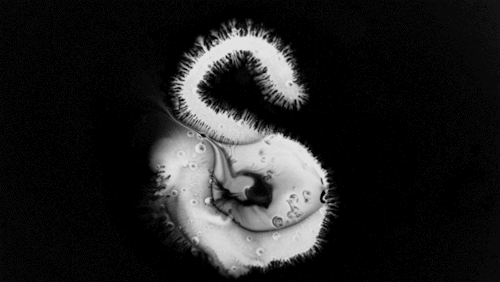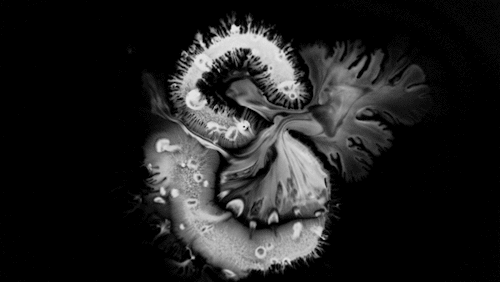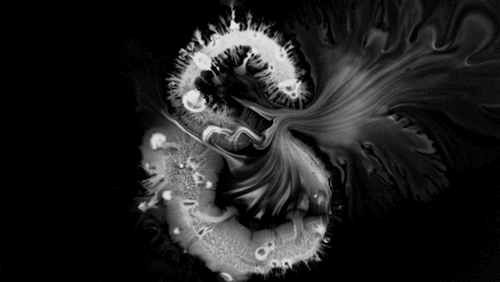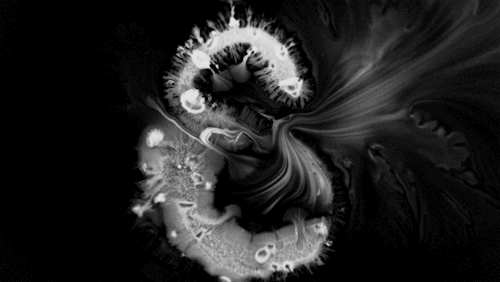
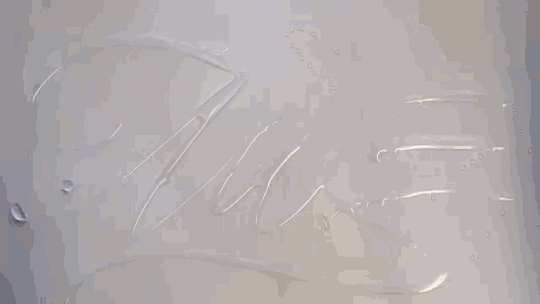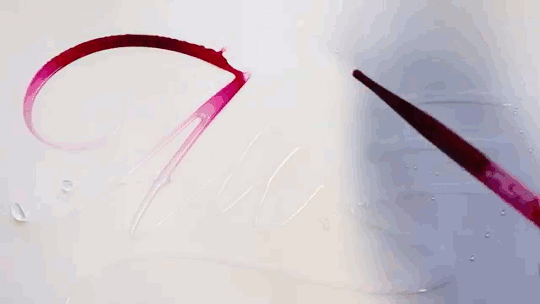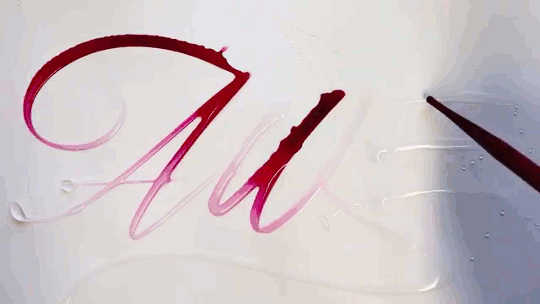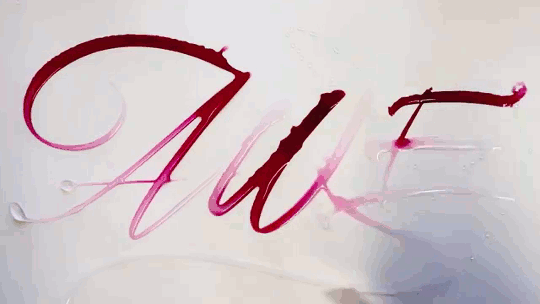In “Liquid Calligraphy,” artist Rus Khasanov’s letters dissolve once he draws them. At first, the white ink spreads in narrow fingers, probably driven by a combination of surface tension gradients, capillary action, and simple diffusion. But then, in flashes, the letters morph faster and flow outward. My best guess is that each jump is a spray from a bottle full of a low surface tension liquid like alcohol. The spray triggers faster outflows than before, like those seen when a strong difference in surface tension activates the Marangoni effect. It’s a beautiful and different artistic take on these important fluid forces. Check out more of his videos here or enjoy high-resolution stills and wallpapers in this style from his Behance page. (Image and video credit: R. Khasanov; submitted by TBBQoC)
Tag: marangoni effect

The Disintegrating Splash
A drop of blue-dyed glycerine impacts a thin film of isopropanol, creating a spectacular splash and breakup. The drop’s impact flings a layer of the isopropanol into the air, where air currents make the thin sheet buckle inward and break into a spray of droplets. Meanwhile, the liquid from the drop forms a thick, blue crown that rises and expands outward. When tiny droplets of the isopropanol hit the splash crown, their lower surface tension causes the blue glycerine to pull away, due to the Marangoni effect. This opens up holes in the crown, which grow quickly, until the entire sheet breaks apart. (Image and research credit: A. Aljedaani et al., source)

Happy 2000 Posts!
Happy Friday and happy 2000th FYFD post! To celebrate, I played with surface tension and the Marangoni effect to make some art. For a run-down on the physics, check out this previous post on water calligraphy. Two thousand posts feels like a major milestone. Not everyone realizes this, but FYFD is a one-woman operation, so 2000 posts is a whole lot of research, image editing, and writing. For fun, I’m including here eight completely random FYFD entries, representing less than one-half of one percent of my total archives:
1. Why did Chinese junks put holes in their rudders?
2. Making droplets in an ultrasonic humidifier
3. Floating on a granular raft
4. Air-trapping fur keeps otters warm
5. The physics of the knuckleball
6. What makes badminton so fast?
7. Playing with fluorescein
8. How frost formsWant to keep up the random walk? Use https://fyfluiddynamics.com/random to find random entries, or if you prefer your browsing to be more directed, check out the visual archive or the themed series page.
As always, a special thanks to those who help support FYFD through Patreon subscriptions – I couldn’t keep writing and making videos without your help! And thank you to all of you who read and share FYFD. Whether you’ve been following along for a week or for the last eight years, your enthusiasm keeps me motivated! Thank you!
(Image credits: 2k animation – N. Sharp; Chinese junk ship – Premier Ship Models; ultrasonic humidifier – S. J. Kim et al.; granular raft – E. Jambon-Puillet and S. Protiere; 3D-printed “fur” – F. Frankel; knuckleball – L. Kang; shuttlecock – Science Friday; fluorescein – Shanks FX; freezing droplets – J. Boreyko et al.)

Water Calligraphy
Artist Seb Lester creates calligraphy using ink and water, but not in the way you might expect. After writing in water, the artist applies ink a drop at a time, allowing fluid forces to spread it. There are a few effects at play here. Molecular diffusion – the random motion of molecules – can help two fluids mix, but it’s an extremely slow process. The fast, dramatic spread of ink seen in the video is more likely a Marangoni effect. The water and ink have different surface tensions, creating a gradient in surface tension that depends on the relative concentration of the two fluids. Gradients in surface tension create flow, which is why the ink spreads most quickly when it’s applied in an area that’s pure water. For similar physics, check out maze-solving soaps and the title sequence for “Marco Polo”. (Video and image credit: S. Lester, source; via Gizmodo)

Swimming Microdroplets
Simple systems can sometimes have surprisingly complex behaviors. In this video, the Lutetium Project outlines a scheme for swimming microdroplets. Most of the droplets shown are just water, but they’re released into a chamber filled with a mixture of oil and surfactants. All flow through the chamber is shut off, but the droplets swim around in complicated, disordered patterns anyway. To see why, we have to zoom way in. The surfactant molecules in the oil cluster around the droplets, orienting so that their hydrophobic parts are in the oil and their hydrophilic parts point toward the water. They actually draw some of the water out of the droplets. This creates a variation in surface tension that causes Marangoni flow, making the droplets swim. Over time, the droplets shrink and slow down as the surfactants pull away more and more of their water and the variations in surface tension get smaller. (Image and video credit: The Lutetium Project; research credit: Z. Izri et al.)


Solving Mazes
Earlier this fall, I attempted my first corn maze. It didn’t work out very well. Early on I unknowingly cut through an area meant to be impassable and thus ended up missing the majority of the maze. Soap, as it turns out, is a much better maze-solver, taking nary a false turn as it heads inexorably to the exit. The secret to soap’s maze-solving prowess is the Marangoni effect.
Soap has a lower surface tension than the milk that makes up the maze, which causes an imbalance in the forces at the surface of the liquid. That imbalance causes a flow in the direction of higher surface tension; in other words, it tends to pull the soap molecules in the direction of the highest milk concentration. But that explains why the soap moves, not how it knows the right path to take. It turns out that there’s another factor at work. Balancing gravitational forces and surface tension forces shows that the soap tends to spread toward the path with the largest surface area ahead. That’s the maze exit, so Marangoni forces pull the soap right to the way out! (Video credit: F. Temprano-Coleto et al.; research credit: F. Peaudecerf et al.)
ETA: Based on the latest research results, gravity may play less of a role than originally thought. Instead, it seems as though the soap chooses its path in part through pre-existing background levels of surfactant. As the dye advances, it compresses the background surfactant, decreasing the local surface tension until it is in equilibrium with dyed area. Because longer paths take longer to reach that equilibrium, the dye spreads preferentially toward the largest surface area.


Convection Without Heat
Glycerol is a sweet, highly viscous fluid that’s very good at absorbing moisture from the ambient air. That’s why a drop of pure glycerol in laboratory conditions quickly develops convection cells – even when upside-down, as shown above. This is not the picture of Bénard-Marangoni convection we’re used to. There’s no temperature or density change involved; in fact, there’s no buoyancy involved at all! This convection is driven entirely by surface tension. As glycerol at the surface absorbs moisture, its surface tension decreases. This generates flow from the center of a cell toward its exterior, where the surface tension is higher. Conservation of mass, also known as continuity, requires that fresh, undiluted glycerol get pulled up in the wake of this flow. It, too, absorbs moisture and the process continues. (Image credit: S. Shin et al., pdf)

“Macrocosm”
In “Macrocosm” artist Susi Sie explores a liquid world of black and white. The two colors diffuse and mix to a soundtrack of “space sounds” recorded by NASA. (Most of these are probably ionic sound rather than sound as we’re used to, but even that is somewhat fluid dynamical.) The result is beautiful, surreal, and more than a little creepy. Happy Halloween! (Video and image credit: S. Sie)



Convection
Blue paint in alcohol forms an array of polygonal convection cells. We’re accustomed to associating convection with temperature differences; patterns like the one above are seen in hot cooking oil, cocoa, and even on Pluto. In all of those cases, temperature differences are a defining feature, but they are not the fundamental driver of the fluid behavior. The most important factors – both in those cases and the present one – are density and surface tension variations. Changing temperature affects both of these factors, which is why its so often seen in Benard-Marangoni convection.
For the paint-in-alcohol, density and surface tension differences are inherent to the two fluids. Because alcohol is volatile and evaporates quickly, its concentration is constantly changing, which in turn changes the local surface tension. Areas of higher surface tension pull on those of lower surface tension; this draws fluid from the center of each cell toward the perimeter. At the same time, alcohol evaporating at the surface changes the density of the fluid. As it loses alcohol and becomes denser, it sinks at the edges of the cell. Below the surface, it will absorb more alcohol, become lighter, and eventually rise at the cell center, continuing the convective process. (Image credit: Beauty of Science, source)

Self-Healing Bubbles
Soap films have the remarkable property of self-healing. A water drop, like the one shown above, can pass through a bubble (repeatedly!) without popping it. This happens thanks to surfactants and the Marangoni effect. Surfactants are molecules that lower the surface tension of a liquid and congregate along the outermost layer of a soap film. When water breaks through the soap film, its lack of surfactants causes a higher surface tension locally. This triggers the Marangoni effect, in which flow moves from areas of low surface tension toward ones of high surface tension. That carries surfactants to the region where the drop broke through and helps stabilize and heal the soap film. Incidentally, the same process lets you stick your finger into a bubble without popping it as long as your hand is wet! (Image credit: G. Mitchell and P. Taylor, source)