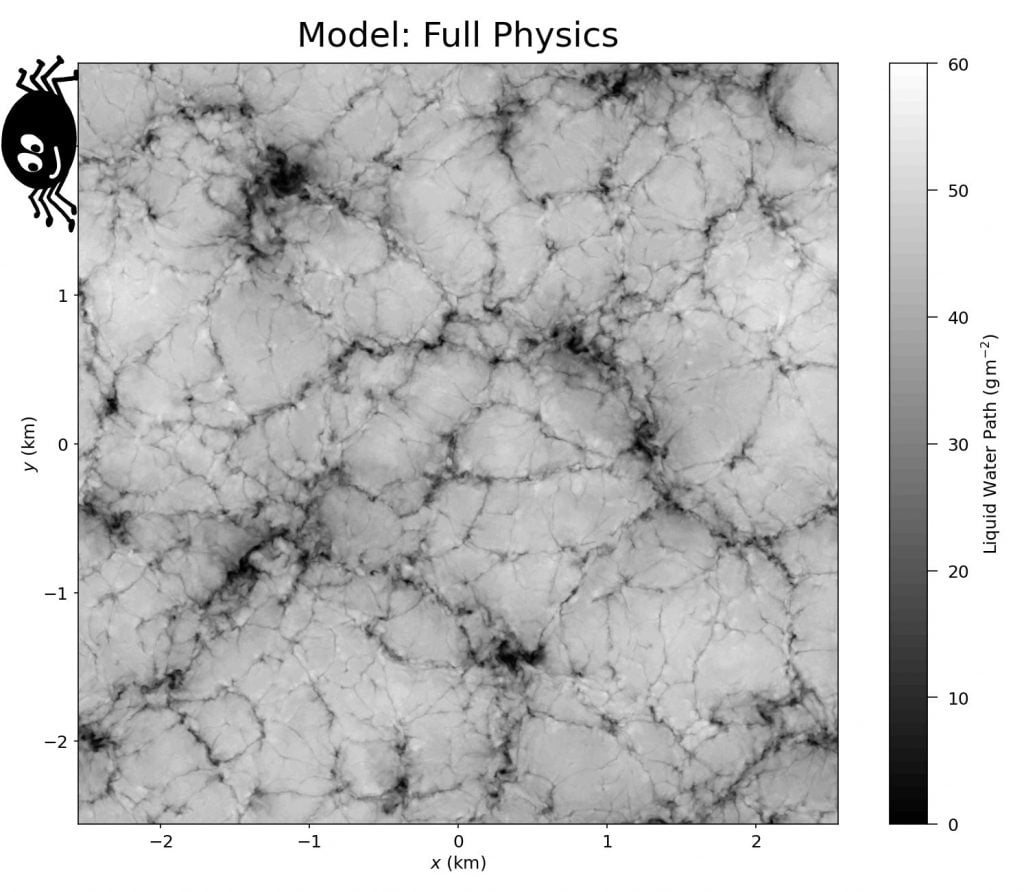
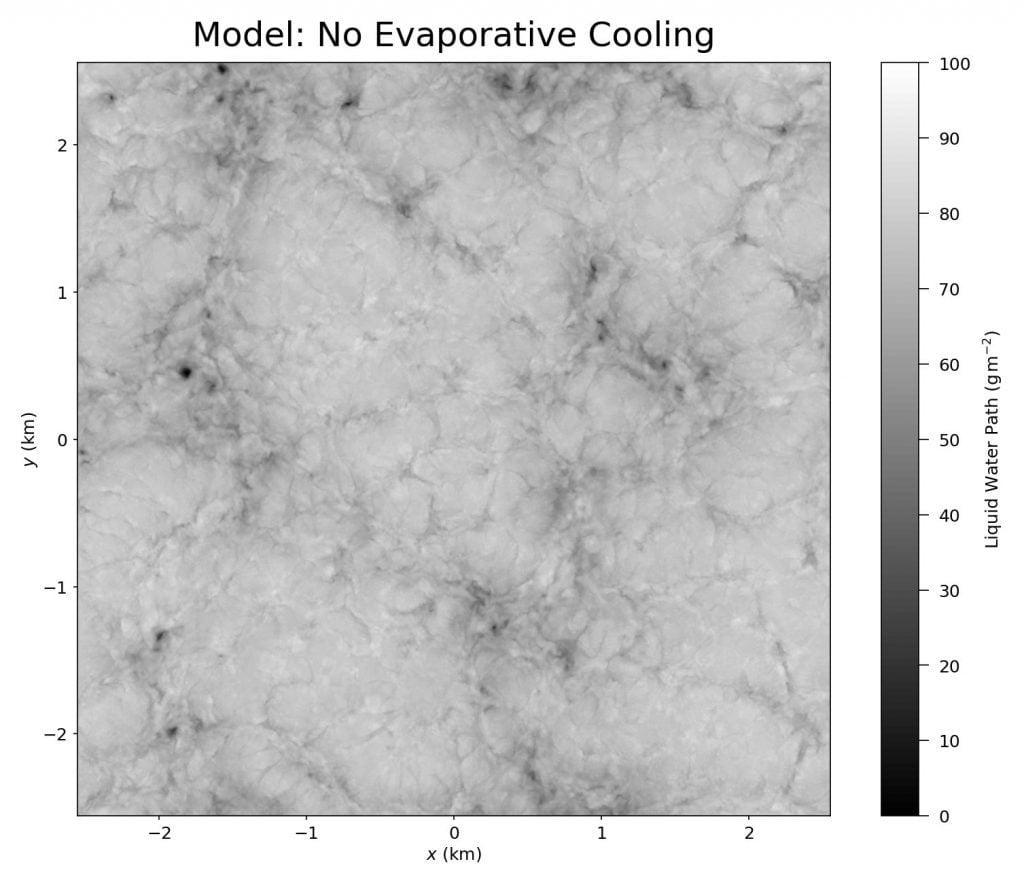
Stratocumulus clouds cover about 20% of Earth’s surface at any given time, and they form distinctive patterns of lumpy cells separated by thin slits. Because of their interconnectedness, researchers nicknamed these narrow regions spiderwebs. New simulations show that evaporative cooling along the cloud tops drives the formation of these spiderwebs (Image 2). Without it (Image 3), the cloud pattern looks very different. (Image credits: featured image – L. Dauphin/MODIS, others – UConn ME 3250; research credit: G. Matheou et al.)
Tag: heat transfer

The Best of FYFD 2020
2020 was certainly a strange year, and I confess that I mostly want to congratulate all of us for making it through and then look forward to a better, happier, healthier 2021. But for tradition and posterity’s sake, here were your top FYFD posts of 2020:
- Juvenile catfish collectively convect for protection
- Gliding birds get extra lift from their tails
- How well do masks work?
- Droplets dig into hot powder
- Updating undergraduate heat transfer
- Branching light in soap bubbles
- Boiling water using ice water
- Concentric patterns on freezing and thawing ice
- Bouncing off superhydrophobic defects
- To beat surface tension, tadpoles blow bubbles
There’s a good mix of topics here! A little bit of biophysics, some research, some phenomena, and some good, old-fashioned fluid dynamics.
If you enjoy FYFD, please remember that it’s primarily reader-supported. You can help support the site by becoming a patron, making a one-time donation, buying some merch, or simply by sharing on social media. Happy New Year!
(Image credits: catfish – Abyss Dive Center, owl – J. Usherwood et al., masks – It’s Okay to Be Smart, droplet – C. Kalelkar and H. Sai, boundary layer – J. Lienhard, bubble – A. Patsyk et al., boiling – S. Mould, ice – D. Spitzer, defects – The Lutetium Project, tadpoles – K. Schwenk and J. Phillips)

A Hand in Hot Oil
In this video, Dianna from Physics Girl demonstrates a feat no one should try at home: dipping her hand into boiling oil. To stay safe, she’s relying on the Leidenfrost effect, the tendency of liquids exposed to temperatures well above their boiling point to vaporize and create a layer of gas that insulates against further heat transfer.
We’ve seen a lot of cool behaviors from Leidenfrost droplets, like surfing on herringbone surfaces, digging through sand, vibrating like a star, and, well, violently exploding. We know a lot about what can happen in this Leidenfrost state, but there are also some major unknowns, like exactly what the Leidenfrost temperature is for many liquids. That’s part of what makes Dianna’s demo so dangerous; the temperature needed to see the Leidenfrost effect — even just for water — varies wildly depending on the experimental set-up. (Video and image credit: Physics Girl)

Updating Undergraduate Heat Transfer
For many engineering students, their first exposure to fluid dynamics comes in a heat transfer class. The typical focus in these classes is not on the underlying physics but on learning to use empirical formulas and correlations that are used in engineering heat exchangers, computer fans, and other applications.
As part of this, students are presented with an extremely simplified view of classical flows like flow over a flat wall, known as a flat-plate boundary layer. Students are told that there are two main features of this and other flows: a laminar region where flow is smooth and orderly, and a turbulent region where flow is chaotic and better at mixing. The transition between these two, according to the undergraduate picture, takes place at a particular point that can be calculated as part of the correlation.
The problem with this picture is that it grossly oversimplifies the actual physics, and for students who may not take dedicated, graduate-level fluid dynamics courses, leaves future engineers with a false understanding that may impact their designs. The truth of transition is far more complicated and nuanced. Transition from laminar to turbulent flow rarely takes place at a single, predictable point; instead it takes place over an extended region and where it begins depends on factors like geometry, vibration, and the level of turbulence already present in the flow.
In an effort to bring undergraduate heat transfer correlations more in line with actual physics — as well as with real, experimental data — a new study revamps the mathematical models. Personally, I applaud any effort to add some nuance to the introduction of this important topic. (Image and research credit: J. Lienhard; via phys.org)

Energy-Efficient Deicing
Defrosting and deicing surfaces is an energy-intensive affair, with lots of heat lost to warming up system components rather than the ice itself. In a new study, researchers explore a faster and more efficient method that focuses on heating just the interface. They coated their working surface in a thin layer of iridium tin oxide, a conductive film used in defrosting. Then, once the surface was iced over, they applied a 100 ms pulse of heating to the film. That localized heat melted the interface, and gravity pulled away the detached ice. Compared to conventional defrosting methods, this technique requires only 1% of the energy and 0.01% of the time. If the method scales reliably to applications like airplane deicing, it would provide enormous savings in time and energy. (Image and research credit: S. Chavan et al.)

Avoiding Droplet Contact
Cold rain splashing on airplane wings can freeze in instants. To prevent that, researchers look for ways to minimize the time and area of contact a drop has. Hydrophobic coatings and textures can do some of the work, but they are easily damaged and don’t always work well when it comes to freezing.
The new technique shown here uses ring-shaped “waterbowls” to help deflect drops. As the drop impacts and spreads, the walls of the ring texture force the lamella up and off the surface. This reduces both the time and area of contact and, under the right circumstances, cuts the heat transfer between the fluid and surface in half. The technique is useful for more than just preventing freezing, though; it would also be helpful for waterproofing breathable fabrics, where shedding moisture quickly without clogging pores is key to keeping the wearer dry. (Image and research credit: H. Girard et al.; via MIT News and Gizmodo)

Different Kinds of Boiling
When you put a pot of water on to boil, you probably don’t give much thought to the process. In our daily lives, we pretty much only see one kind of boiling: the sort where lots of small bubbles form on a hot surface and then rise. That’s nucleate boiling (top image), and it’s typical when you have a surface close to the boiling point of a liquid.
But when you continue raising the temperature of the surface, you get a transition to a different boiling regime (middle image). In this final regime (bottom image), a film of vapor envelopes the heated surface; hence its name: film boiling. Because vapor is less efficient for heat transfer than a liquid, a surface undergoing film boiling can become much, much hotter because it cannot transfer its heat away efficiently. In this experiment, the tube starts at 375K during nucleate boiling and rises to a temperature nearly three times higher during film boiling. (Image credit: TSL, source)

Heating from Cavitation
When cavitation bubbles collapse, they can produce temperatures well over 2,000 Kelvin. Since cavitation near a surface can be so destructive, researchers have long wondered whether the high temperatures inside the bubble can be transmitted to nearby surfaces. A new set of numerical simulations provides some insight into that process. The researchers found that collapsing cavitation bubbles raised nearby wall temperatures in two ways: bubbles that were further away sent shock waves that heated the material, and nearby bubbles could contact the surface itself as they collapsed.
Heat transfer requires time, however; this is part of why quickly dunking your hand in liquid nitrogen and pulling it out likely won’t damage you. (Still, we don’t recommend it.) The cavitation bubbles could only transmit these high temperatures for less than 1 microsecond, which means that most materials won’t actually heat up to their melting temperature. The researchers did conclude, however, that softer materials exposed to frequent bubble collapses could show localized melting under the barrage. (Image credit: L. Krum; research credit: S. Beig et al.)

The Lava Lamps That Secure the Internet
A wall of lava lamps in a San Francisco office currently helps keep about 10% of the Internet’s traffic secure. Internet security company Cloudflare uses a video feed of the lava lamps as one of the inputs to the algorithms they use to generate large random numbers for encryption. The concept dates back to a 1996 patent for a product called LavaRand. The idea is that using a chaotic, unpredictable source as a seed for random number generators makes it much harder for an adversary to crack your encryption.
With lava lamps, a lot of that chaos comes from the fluid dynamics involved – without perfect knowledge of thousands of variables, it would be impossible to simulate the lava lamp wall and get the same outcome as the real one – but there’s also randomness that comes from the measurement. People walking by, shifts in lighting, and random fluctuations of individual pixels all help make the video feed unpredictable. For those interested in the details of how Cloudflare uses their lava lamps, the company explains things for both technical and non-technical readers. You can also check out Tom Scott’s video for a good overview. (Image and video credit: T. Scott; submitted by Jean H.)

Jumping Droplets
Condensation, which removes heat by changing a vapor into a liquid, is a common feature in industrial heat transfer. When droplets form on surfaces, they typically have to grow to millimeter size before gravity causes them to slide off and open up the surface to new droplet formation. Hydrophobic surfaces can shed droplets a little sooner. Droplets only 100 micrometers in size will spontaneously jump off hydrophobic surfaces due to the release of excess surface energy during droplet coalescence, but this only happens when those droplets have a small contact area with the surface. Defects in the nanoscale structure of the surface can allow water to squeeze in between posts and hold on.
To counter this, new experiments packed copper nanowires into a dense 3D array. This permits fewer defects and helps condensing droplets leap from the surface sooner. Each droplet carries away a bit of the surface’s heat. The new method is impressively efficient at it. Researchers found the new heat exchanger could remove 100% more heat than previous hydrophobic designs. (Video credit: Science; research credit: R. Wen et al.)