Dyed isopropyl alcohol atop a thin layer of acrylic medium spreads in a fractal fingering pattern. Although the shapes are reminiscent of the viscous fingers seen in in the Saffman-Taylor instability, these patterns are most likely a result of surface tension. The lower surface tension of the alcohol causes Marangoni forces to pull it outward. The branching shapes indicate an instability, likely driven by surface tension, but the details of the mechanism behind it are unclear. (Image credits: J. Nahabetian)
Search results for: “viscous”

Breaking Ground
Pushing a fluid into a porous granular material can fracture it into branching, lightning-like patterns. Here, air is injected into wet grains as a laboratory analog to hydrocarbon extraction or fracturing to treat contaminated soil. The injection of air compacts grains along the branch boundaries, keeping individual branches separated from one another. The patterns that form change with grain shape and ultimately result from the interactions of pressure, surface tension, friction and viscous forces. Studies like these help optimize fluid flow, decontaminate polluted soil faster, and determine risk in gas-driven fracturing of hydrocarbon reservoirs. (Image and video credit: J. Campbell et al.; submitted by B. Sandnes)

Sunset Flow
Day and night mix in this flow visualization of watercolor pigments and ferrofluid. The former, as suggested by their name, are water-based, whereas ferrofluids typically contain an oil base. This means the two fluids are immiscible. Like oil and vinegar in salad dressing, the only way to mix them is to break one into tiny droplets floating in the other. This is what happens near their boundary, where brightly-colored paint droplets float in a network of dark channels. To the right, the paint and ferrofluid have been swirled around to create viscous mixing patterns among the paint colors with occasional intrusions of thin ferrofluid fingers. (Image credit: G. Elbert)
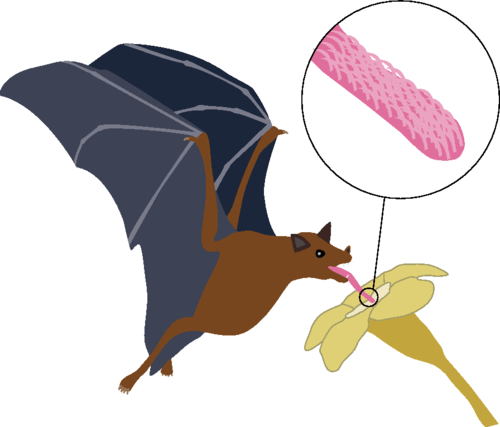
Hairy Tongues Help Bats Drink
Nectar-drinking bats, honey possums, and honeybees all use hair-like protrusions on their tongues to help them drink. In bats, these papillae have blood vessels that swell when drinking, stiffening the hairs. To investigate this drinking mechanism, researchers built their own version of a bat tongue by fabricating hairy surfaces and testing how well they trapped viscous oil when dipped and withdrawn. Through a combination of experiment and mathematical modeling, the researchers found that the optimal fluid uptake depended on the density of hairs, fluid viscosity, and the withdrawal speed. When they compared their results to actual bats, honey possums, and honeybees, they found that those animals’ tongues have hair densities very close to the predicted optimal value, suggesting that their model is capturing the important physical mechanisms that have driven evolutionary advantages for these species. (Image and research credit: A. Nasto et al.; submitted by Kam-Yung Soh)

Growing Fingers
Branching, tree-like structures are found throughout nature. Take a thin layer of a viscous fluid pressed between two glass plates and inject a less viscous fluid like air and you’ll get branch-like structures. These are the result of the Saffman-Taylor instability and usually result in a fairly random outcome because of the instability’s sensitivity to small variations. In a new study, researchers use multiple air injection ports to finely control the formation and growth of air fingers, allowing them to build well-ordered branching structures like the one above. By placing the air ports in an array, the same technique can be used to create fluid meshes. The authors suggest this new technique could have wide-ranging applications including the design of heat exchangers and the growth of artificial tissues. (Image and research credit: T. ul Islam and P. Gandhi, source)

Swimming Like a Balloon
For humans, swimming is relatively easy. Kick your legs, wheel your arms, and you’ll move forward. But for microswimmers, swimming can be more complicated. For them, the world is a viscous place, and the rules that we swim by can’t help them get around. In a highly viscous world, flows are reversible. Kick one limb down and you might move forward, but when you pull the limb up, you’ll be sucked right back to where you started. So microswimmers must use asymmetry in their swimming. In other words, their recovery stroke cannot be the mirror-image of their power stroke.
A new study suggests that simple elastic spheres could make good microswimmers through cyclic inflation and deflation. When the sphere deflates, it buckles, making a shape unlike its inflating one. This difference in shape change is enough to propel the sphere a little with each cycle. Right now the test system is a macroscale one, but the researchers hope to continue miniaturizing. (Image and research credit: A. Djellouli et al.; via APS Physics; submitted by Kam-Yung Soh)

Building with Sand
Sand and water make a remarkable team when it comes to building. But the substrate – the surface you build on – makes a big difference as well. Take a syringe of wet sand and drip it onto a waterproof surface (bottom right), and you’ll get a wet heap that flows like a viscous liquid. Drop the same wet sand onto a surface covered in dry sand (bottom left), and the drops pile up into a tower. Watch the sand drop tower closely, and you’ll see how new drops first glisten with moisture and then lose their shine. The excess water in each drop is being drawn downward and into the surrounding sand through capillary action. This lets the sand grains settle against one another instead of sliding past, giving the sand pile the strength to hold its weight upright. (Video and image credit: amàco et al.)

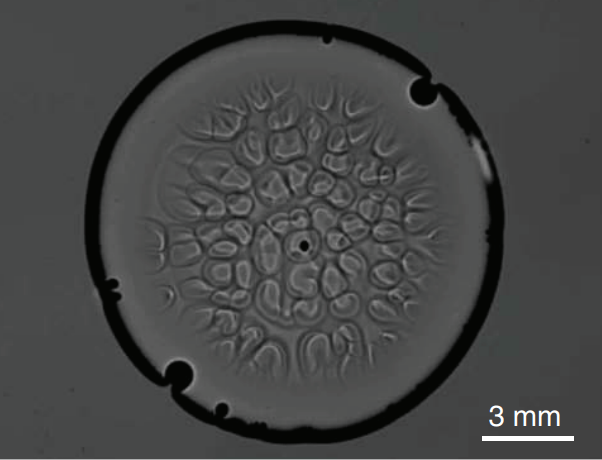
Convection Without Heat
Glycerol is a sweet, highly viscous fluid that’s very good at absorbing moisture from the ambient air. That’s why a drop of pure glycerol in laboratory conditions quickly develops convection cells – even when upside-down, as shown above. This is not the picture of Bénard-Marangoni convection we’re used to. There’s no temperature or density change involved; in fact, there’s no buoyancy involved at all! This convection is driven entirely by surface tension. As glycerol at the surface absorbs moisture, its surface tension decreases. This generates flow from the center of a cell toward its exterior, where the surface tension is higher. Conservation of mass, also known as continuity, requires that fresh, undiluted glycerol get pulled up in the wake of this flow. It, too, absorbs moisture and the process continues. (Image credit: S. Shin et al., pdf)
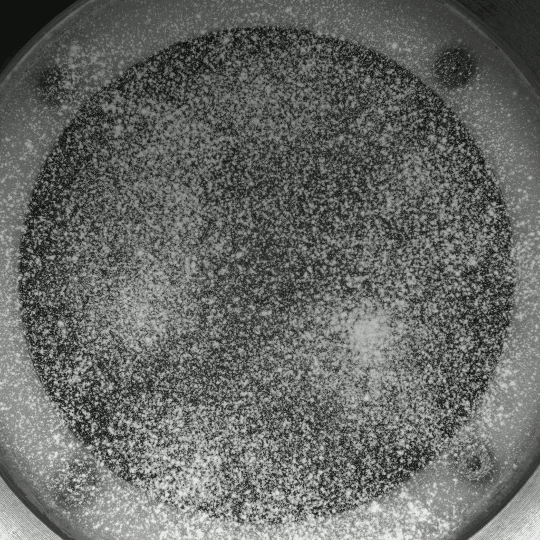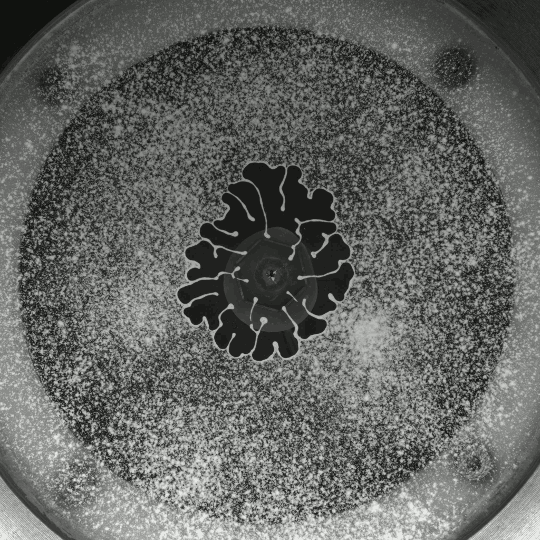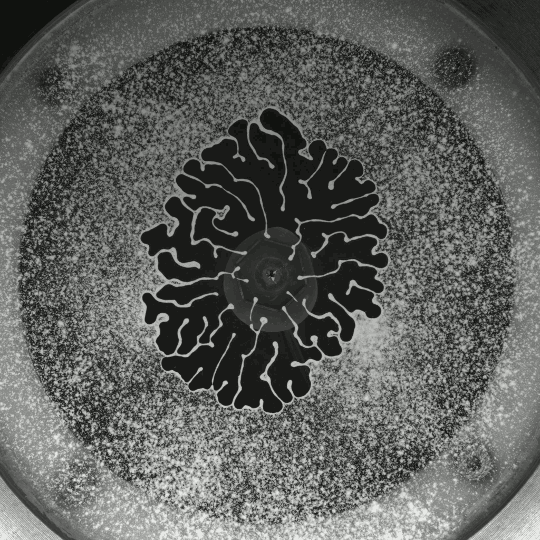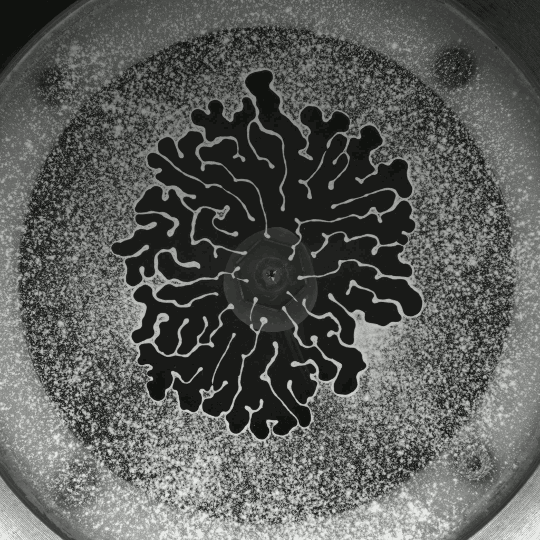
Porous Fingers
If you inject a less viscous fluid, like air, into a narrow gap between two glass plates filled with a more viscous fluid, you’ll get a finger-like instability known as the Saffman-Taylor instability. If you invert the situation – injecting something viscous like water into air – the water will simply expand radially; you’ll get no fingers. But that situation doesn’t hold if there are wettable particles in the air-filled gap. Inject water into a particle-strewn air gap and you get a pattern like the one above. In this case, as the water expands, it collects particles on the meniscus between it and the air. Once the concentration of particles on the meniscus is too high for more particles to fit there, the flow starts to branch into fingers. This creates a greater surface area for interface so that more particles can get swept up as the water expands. (Image and research credit: I. Bihi et al., source)
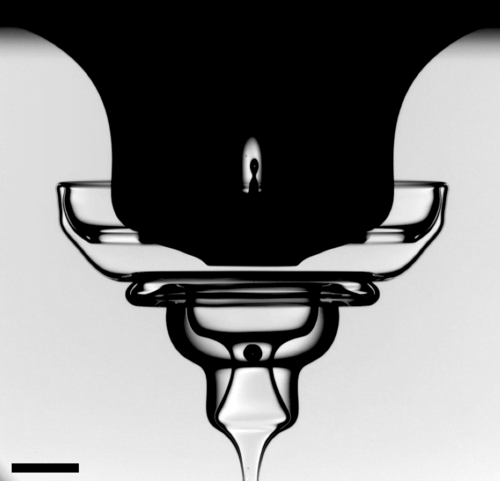
Wrinkling Drops
When a viscous drop falls into a pool of a less viscous liquid, the drop can deform into some beautiful and complex shapes. Typically, shear forces between the drop and its surroundings cause a vortex ring to roll up and advect downward, thereby stretching the remainder of the drop into thin sheets that can buckle and wrinkle. Here the drop is about 150 times more viscous than the pool and impacts at 1.45 m/s, making a rather energetic entry. The vortex ring (not visible) has stretched the drop’s remains downward while a buoyant bubble caught by the impact pulls some of the drop back toward the surface. As a result, the thin sheets of the drop’s fluid are buckling and folding back on themselves like an elaborate and delicate glass sculpture. This entire paper is full of gorgeous images and videos. Be sure to check them out! (Image and research credit: E. Q. Li et al.; see supplemental info zip for videos)
