Being hot isn’t always enough to make water boil. To form vapor bubbles, water and other liquids need imperfections that serve as seeds. In the absence of these, the liquid can become superheated, reaching temperatures higher than its boiling point without forming bubbles. Superheated water can be quite dangerous because it appears to be cooler, but once it’s disturbed – thereby breaking its surface tension – vapor bubbles form rapidly and explosively. You can see in the animation above just how quickly and unsteadily a sudden vapor bubble expands as it rises to the surface. (Image credit: C. Kalelkar and K. Raj, source)
Search results for: “surface tension”

Reader Question: White Caps
Reader eclecticca asks:
I really like the last two posts about waves, and they left me with another question… My dad had a little boat he used to take us ocean fishing on quite a bit. I always noticed that some days we just had big waves (swells) when out from the coastline and in fairly deep water (a hundred feet to hundreds of feet according to the depth sounder) and other days those swells would “break” and curl and foam and crash in on themselves, being what we called “breakers” or “white caps”. There is no shore to create the breakers in this case, so what is happening? Is it due to wind? current a combination of factors? Always been kind of curious about this really…
You’re exactly right: those open ocean white caps are due to wind. Strictly speaking, the wind is what’s causing all* of the waves out in open, deep waters. But once the wind is strong enough, it starts breaking up the crests of waves, creating those foamy white tops.
According to one study, the break-up happens when the wind transfers more energy to the wave than surface tension can withstand. When the wave crest breaks up into a mixture of air, spray, and foam, it effectively gives the wind more surface area to push against and continue transferring energy. (Image credit: M. Moers)
* With a few notable exceptions, like in the case of a tsunami.
Breaking Up
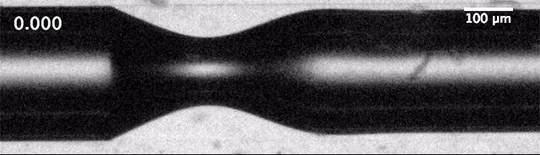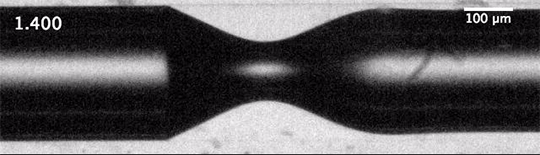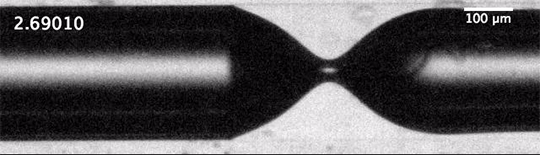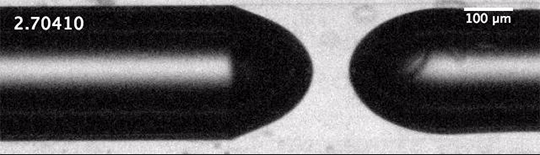
The dripping of a faucet and the break-up of a jet into droplets is universal. That means that the forces – the inertia of the fluid, the capillary forces governed by surface tension, and the viscous dissipation – balance in such a way that the initial conditions of the jet – its size, speed, etc. – don’t matter to the process of break-up.
We’d expect that the inverse situation – the breakup of a gas into bubbles in a liquid – would be similarly universal, but it’s not. When unconfined bubbles pinch off, the way they do so is heavily influenced by initial conditions. But that changes, according to a new study, if you confine the gas to a liquid-filled tube before pinch-off. Confinement forces a different balance between viscous and capillary effects, one which effectively erases the initial conditions of the flow and restores universality to the pinch-off process. (Image and research credit: A. Pahlavan et al.; via phys.org)

Collecting Dew
In areas of the world where fresh water is scarce, one potential source is dew collection. Scientists have been working in recent years on making overnight dew collection more efficient. The challenge is that drops won’t begin to slide down an inclined surface until they are large enough for gravity to overcome the surface tension forces that pin the drop. Most efforts have focused on reducing the critical size where drops begin to slide through surface treatments and chemical coatings.
A recent study, however, uses a different tactic. Instead of aiming to reduce the critical drop size, these researchers built a grooved surface designed to encourage drops to grow faster. By helping the droplets coalesce quickly, their surface (right side) is able to start shedding droplets much faster than a smooth surface (left side). Under test conditions, the grooved surface was shedding droplets after only 30 minutes, whereas the smooth surface shed its first drops after 2 hours. (Image and research credit: P. Bintein et al.; see also APS Physics)

Lensing in a Straw
While doing the sort of experiment only a kid or a scientist would pursue – namely, staring down a straw – Dianna noticed that water in a straw creates a lens-like magnification effect as the straw moves or down. This happens thanks to the curvature of the air-water-straw interface. Because water has strong surface tension, it curves dramatically as it meets the wall of the straw, and moving the straw up or down will drag some of the fluid with it, enhancing the curvature. When light refracts across that interface, it gets bent the same way it would through a lens, thereby shrinking or magnifying the objects beneath. (Video credit: D. Cowern/Physics Girl)

Pouring a Liquid Mirror
In this video, the Slow Mo Guys play with liquid gallium, giving us a chance to see how molten metals behave (outside of, say, the Terminator movies). Near its melting point, gallium is about six times denser than water, with a viscosity three times higher, and a surface tension about ten times greater. So how do those properties affect its behavior?
You may be surprised that when watching the gallium vibrate on a speaker or get poured into a pan, it doesn’t look all that different from water. Yes, it’s highly reflective, but, on the whole it doesn’t look radically different from a distance. We can use the Reynolds number to quantify what’s going on here. It’s a dimensionless number that compares the fluid’s inertial force to the viscous force. Imagine we have two versions of an experiment, one where we pour gallium at a given speed and one pouring at the same volume and speed but with water. If we compared the Reynolds numbers of the water and the gallium, they only differ by a factor of two. Overall, that’s not very much. That’s why the two pours look similar.
The story is different, though, if we look at individual drops of gallium and water, like when the first few drops of our pour hit the surface. Check out the gallium drops below. They’re conical on either end! This looks very different from what we expect with water droplets. You might think that’s because the metal is more viscous, but if we compare a water drop with a gallium drop of the same characteristic size and impact speed, we find a different story. For this, we’ll use the Ohnesorge number, which compares the viscous forces to a combination of inertia and surface tension. In this case, we find that the gallium drop’s Ohnesorge number is almost an order of magnitude smaller than the water droplet’s. That means that viscosity isn’t a major factor for our gallium drop. Both surface tension and inertia are more important.
But if the surface tension is so high, then why aren’t the droplets spherical? Mostly because they don’t have time to form spheres before they hit. Their shape suggests that they’ve only just broken into droplets, which makes sense if the pour is fast and the surface tension is strong. (Video and image credit: The Slow Mo Guys)

“Aurora”
In “Aurora”, artist Rus Khasanov uses fluids to create a short film full of psychedelic color and cosmic visuals. As in a soap bubble, the bright colors – as well as the pure black holes – come from the interference of light rays. The colors directly relate to the thickness of fluid, and they allow us to see all the subtle flows caused by variations in surface tension. (Video and image credit: R. Khasanov)

Transporting Droplets
Transporting droplets easily and reliably is important in many microfluidic applications. While this can be done using electric fields, those fields can impact biological characteristics researchers are trying to measure. As an alternative, a group of researchers have developed the concept of “mechanowetting,” a technique that uses surface tension forces to hold droplets on a traveling wave.
Now visually, it’s a bit tough to see what’s going on here. In the animations, it looks like the droplets are just sticking to a moving surface, but that’s an illusion. The surface the droplet is sitting on is fixed and unmoving. It’s a thin silicone film that covers a ridged conveyor belt. The belt underneath can (and does) move. This creates a traveling wave. Instead of that wave simply passing beneath the droplet, it triggers an internal flow and restoring force that helps the drop follow the wave. The effect is strong enough that small droplets are even able to climb up vertical walls or stick upside-down. (Image, research, and submission credit: E. de Jong et al.)

Oil-on-Water Impact
Although many people have studied droplet impacts over the years, there’s been remarkably little work done with oil-on-water impacts. One of the things that makes this situation different is that the oil and water are completely immiscible, which means we can see aspects of the impact process that are invisible with, say, water-on-water impacts.
The animation above shows an underwater view of the oil droplet’s impact. The energy of the initial impact creates an expanding crater and an unstable crown splash. That crown splash contains both water and oil. After it ejects some droplets, the rim stabilizes, but we can still see small perturbations along its edge as it starts to retract. In the water, high surface tension damps out these perturbations. Not so for the oil! As the crater retracts, the small disturbances along the rim get stretched into mushroom-shaped fingers that point inward toward the impact site. Because the index of refraction is different between oil and water, we can see the fingers clearly near the end of the animation. (Image and research credit: U. Jain et al.; submitted by Utkarsh J.)

Ink Explosion
Sometimes beautiful flows come from simple combinations. Here the artists of Chemical Bouillon combine ink and hydrocarbons to create lovely explosions of color. Eschewing quick cuts between views, they allow us to linger and explore the flow ourselves as it changes. Differences in surface tension drive streaming flows along the surface, but there seem to be some chemical reactions contributing as well. Watch along the edges and you may even see convection pulling ink down and back. The whole video is only 2 minutes long and worth a full watch. (Image and video credit: Chemical Bouillon)








