Rotation can cause non-intuitive effects in fluid dynamical systems. UCLA Spinlab’s newest video tackles the problem using four demonstrations. The first two deal with droplets released in air, first in a non-rotating environment and then in a rotating one. As one would expect, in a non-rotating environment, droplets fall through the tank in a straight line. When rotating, though, the droplets follow a deflected, straight-line path due to centrifugal effects. This is the same as the way passengers in a car feel like they’re being thrown to the outside of a turn on a curvy road. When the experiment is repeated with a tank of water instead of air, the results are different. The densities of the creamer and water are much closer to one another, so the droplet falls much slower than before. The tank now rotates faster than time it takes the drop to fall. This smaller timescale means that the droplet experiences more acceleration from Coriolis forces than centrifugal forces in the rotating tank of water. Thus, instead of being thrown outward, the drop now forms a column aligned with the axis of rotation. (Video credit: UCLA Spinlab; submitted by Jon B.)
Search results for: “droplet”

Inside a Rocket
Rockets often utilize liquid propellants for their combustion. To maximize the efficiency during burning, the liquid fuel and oxidizer must mix quickly and break up into an easily vaporized spray. One method to achieve this is to inject the fuel and oxidizer as liquid jets that collide with one another. For high enough flow rates, this creates a highly unstable liquid sheet that quickly atomizes into a spray of droplets. The animation above shows an example of two impinging jets, but rockets using this method would typically have more than just two injection points. Other rockets use co-axial or centrifugal injectors to mix and atomize the fuel and oxidizer prior to combustion. (Image credit: C. Inoue; full-scale GIF)

Giant Bubbles
In their latest video, Gavin and Dan of The Slow Mo Guys demonstrate what giant bubbles look like in high-speed video from birth to death. Surface tension, which arises from the imbalance of intermolecular forces across the soapy-water/air interface, is the driving force for bubbles. As they move the wand, cylindrical sheets of bubble film form. These bubble tubes undulate in part because of the motion of air around them. In a cylindrical form, surface tension cannot really counteract these undulations. Instead it drives the film toward break-up into multiple spherical bubbles. You can see examples of that early in the video. The second half of the video shows the deaths of these large bubble tubes when they don’t manage to pinch off into bubbles. The soap film tears away from the wand and the destructive front propagates down the tube, tearing the film into fluid ligaments and tiny droplets (most of which are not visible in the video). Instead it looks almost as if a giant eraser is removing the outer bubble tube, which is a pretty awesome effect. (Video credit: The Slow Mo Guys)

“Clourant”
Photographers Cassandra Warner and Jeremy Floto produced the “Clourant” series of high-speed photographs of colorful liquid splashes. The artists took special care to disguise the origin of splashes, making them appear like frozen sculptures. The photos are beautiful examples of making fluid effects and instabilities. Many of them feature thin liquid sheets with thicker rims just developing ligaments. In other spots, surface tension has been wholly overcome by momentum’s effects and what was once ligaments has exploded into a spray of droplets. (Photo credit: C. Warner and J. Floto; submitted by jshoer; via Colossal)
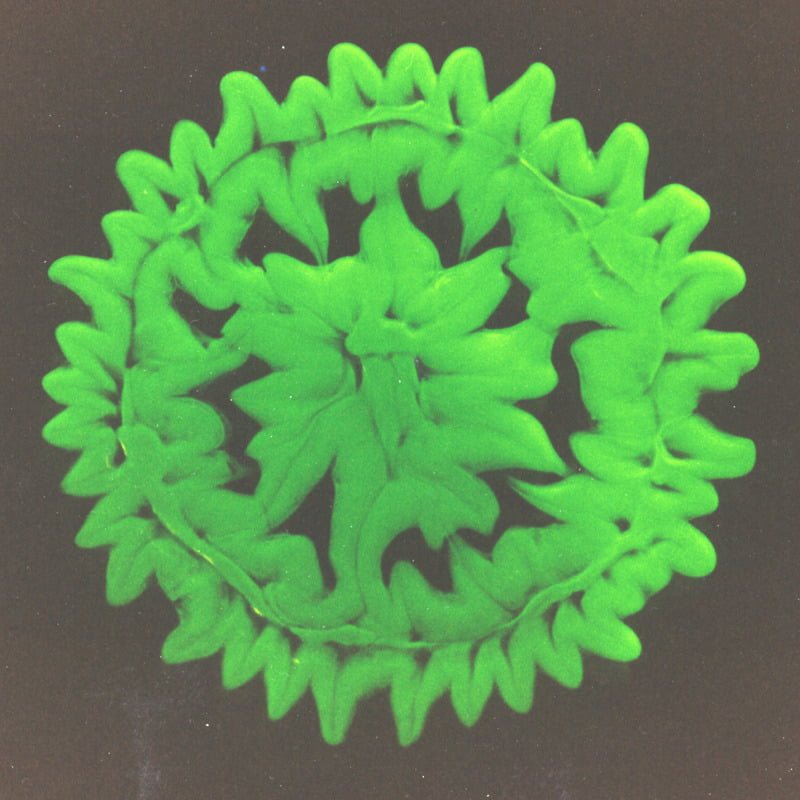
Distorted Rings
The Marangoni effect is generated by variations in surface tension at an interface. Such variations can be temperature-driven, concentration-driven, or simply due to the mixing between fluids of differing surface tensions as is the case here. The pattern in the image above formed after a dyed water droplet impacted a layer of glycerin. The initial impact of the drop formed an inner circle and outer ring. This image is from 30 seconds or so after impact, after the Marangoni instability has taken over. The higher surface tension of the water pulls the glycerin toward it, resulting in a flower-like pattern. (Photo credit: E. Tan and S. Thoroddsen)

Forming a Jet
Many situations can generate high-speed liquid jets, including droplet impacts, vibrated fluids, and surface charges. In each of these cases, a concave liquid surface is impulsively accelerated, which causes the flow to focus into a jet. The image above shows snapshots of a microjet generated from a 50 micron capillary tube visible at the right. This jet formed when the meniscus inside the capillary tube was disturbed by a laser pulse that vaporized fluid behind the interface. Incredibly, the microjets generated with this method can reach speeds of 850 m/s, nearly 3 times the speed of sound in air. Researchers have found the method produces consistent results and suggest that it could one day form the basis for needle-free drug injection. You can read more in their freely available paper. (Photo credit: K. Tagawa et al.)

Colliding in Microgravity
On Earth, it’s easy for the effects of surface tension and capillary action to get masked by gravity’s effects. This makes microgravity experiments, like those performed with drop towers or onboard the ISS, excellent proving grounds for exploring fluid dynamics unhindered by gravity. The video above looks at how colliding jets of liquid water behave in microgravity. At low flow rates, opposed jets form droplets that bounce off one another. Increasing the flow rate first causes the droplets to coalesce and then makes the jets themselves coalesce. Similar effects are seen in obliquely positioned jets. Perhaps the most interesting clip, though, is at the end. It shows two jets separated by a very small angle. Under Earth gravity, the jets bounce off one another before breaking up. (The jets are likely separated by a thin film of air that gets entrained along the water surface.) In microgravity, though, the jets display much greater waviness and break down much quicker. This seems to indicate a significant gravitational effect to the Plateau-Rayleigh instability that governs the jet’s breakup into droplets. (Video credit: F. Sunol and R. Gonzalez-Cinca)

The March of Drops
I love science with a sense of humor. This video features a series of clips showing the behavior of droplets on what appears to be a superhydrophobic surface. In particular, there are some excellent examples of drops bouncing on an incline and droplets rebounding after impact. For droplets with enough momentum, impact flattens them like a pancake, with the rim sometimes forming a halo of droplets. If the momentum is high enough, these droplets can escape as satellite drops, but other times the rebound of the drop off the superhydrophobic surface is forceful enough to overcome the instability and draw the entire drop back off the surface. (Video credit: C. Antonini et al.)

Space Balls (of Water!)
The microgravity environment of space is an excellent place to investigate fluid properties. In particular, surface tension and capillary action appear more dramatic in space because gravitational effects are not around to overwhelm them. In this animation, astronaut Don Petit injects a jet of air into a large sphere of water. Some of the water’s reaction is similar to what occurs on Earth when a drop falls into a pool; the jet of air creates a cavity in the water, which quickly inverts into an outward-moving jet of water. In this case, the jet is energetic enough to eject a large droplet. Meanwhile, the momentum, or inertia, from the air jet and subsequent ejection causes a series of waves to jostle the water sphere back and forth. Surface tension is strong enough to keep the water sphere intact, and eventually surface tension and viscosity inside the sphere will damp out the oscillations. You can see the video in full here. (Image credit: Don Petit/Science off the Sphere)

Pointed Drops
When water droplets sit on a cold substrate, they freeze into a shape with a pointed tip. At first glance, this behavior seems very odd since surface tension usually acts to prevent such sharp protrusions. The shape is, however, a result of water’s expansion as it freezes. The droplet freezes from the substrate upward, with a concave shape to the solidification front. The angle of the point does not depend on the substrate temperature or the wetting angle between the water and surface. Instead, it turns out that this concave front shape and water’s expansion are the key factors that determine the pointed cusp’s angle, and that the final geometry of the cusp is essentially universal. (Video credit: M. Nauenberg; additional research credit: A. Marin et al.)




