Both synchronized swimming and water polo require competitors to hold themselves stable above the water’s surface without touching the pool’s bottom. One of the basic techniques for doing so in both sports is known as the eggbeater kick, shown above. The eggbeater kick is very similar to the motion for the breaststroke’s kick, but it’s performed upright and with alternating leg motions, sweeping a clockwise circle with the left leg and a counterclockwise one with the right.
A swimmer typically stays afloat due to a buoyant force equal to the weight of the volume of water the swimmer displaces. Rising further out the water means reducing the buoyant force, so the swimmer must generate other forces to counter their weight. The eggbeater kick does this two ways. First, as the swimmer sweeps their foot around, it acts like a hydrofoil, generating lift that holds the swimmer up. Second, other parts of the kick cycle force water downward, which, by Newton’s third law, pushes the swimmer up.
Keeping a wide stance and sweeping the legs alternately allows the athlete to balance the horizontal forces their motions create while keeping the upward forces generated relatively constant. This gives them a stable, arms-free platform that’s a foundation for everything else their sport requires. (Image credits: GettyImages; The Studio WLV, source)
Previously: How buoyancy helps swimmers
Join us throughout the Rio Olympics for more fluid dynamics in sports. If you love FYFD, please help support the site!

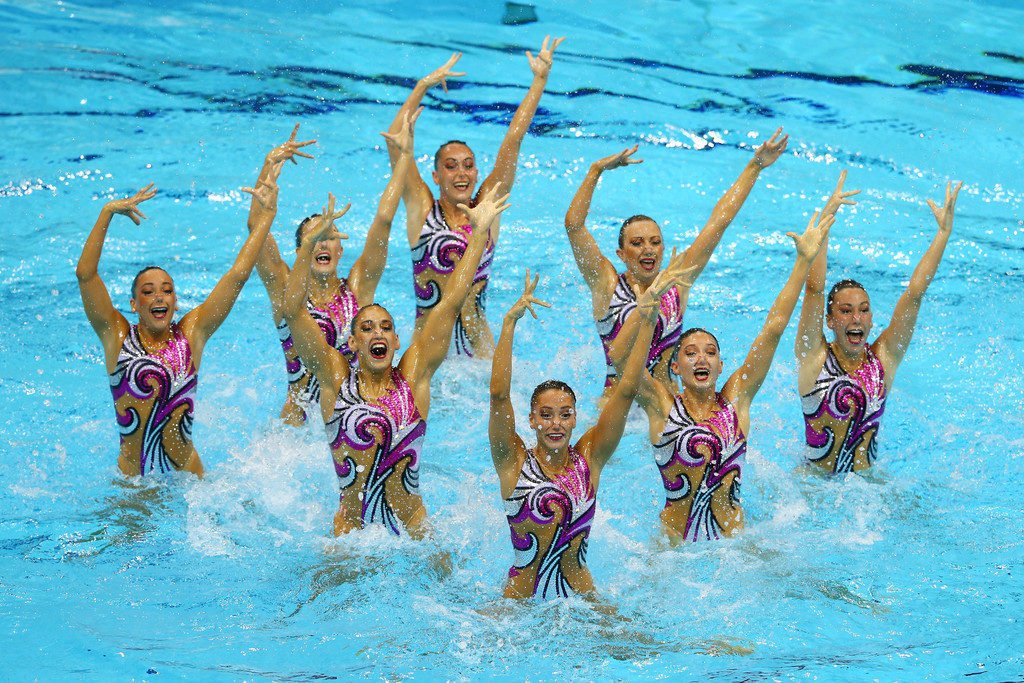











 Strange as it seems, elite swimmers are faster when swimming underwater than they are at the surface. So much so, in fact, that they’re restricted to being underwater only 15 m after a dive or turn. To see just how stark a difference this makes,
Strange as it seems, elite swimmers are faster when swimming underwater than they are at the surface. So much so, in fact, that they’re restricted to being underwater only 15 m after a dive or turn. To see just how stark a difference this makes, 












