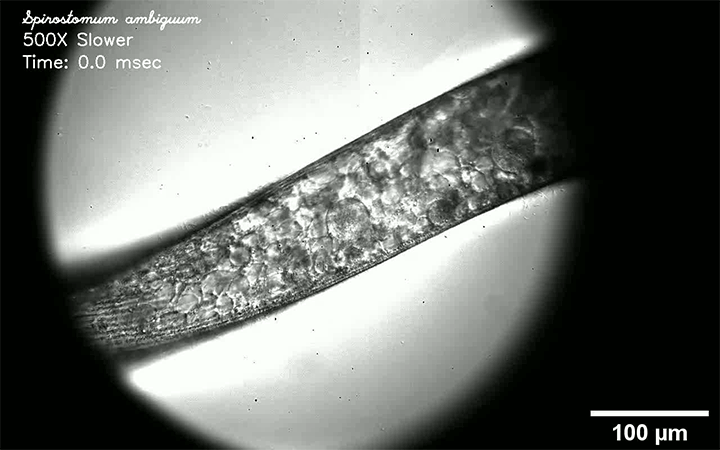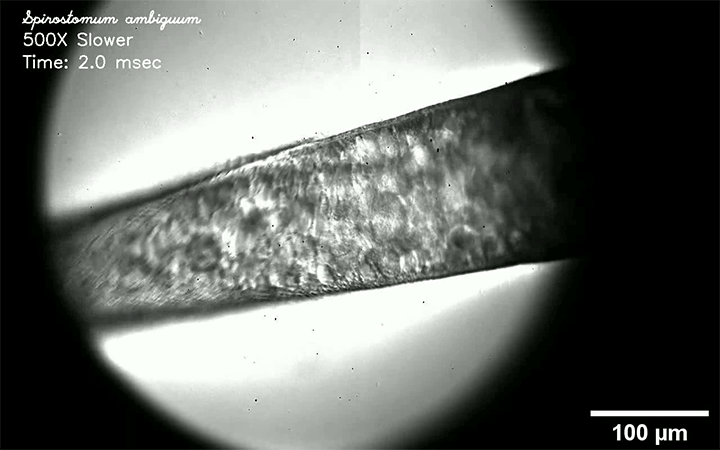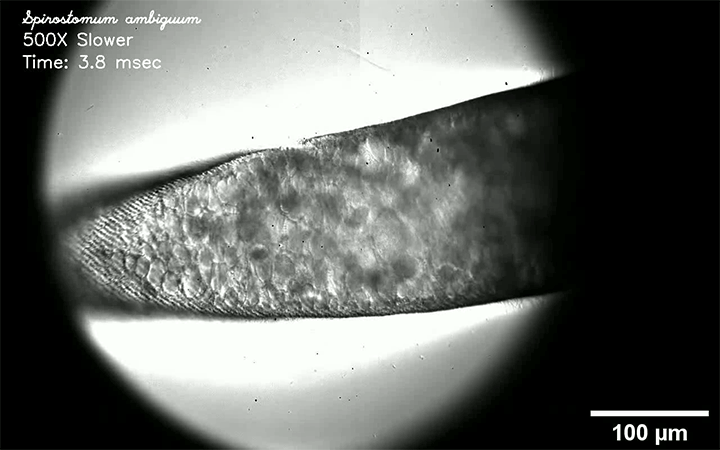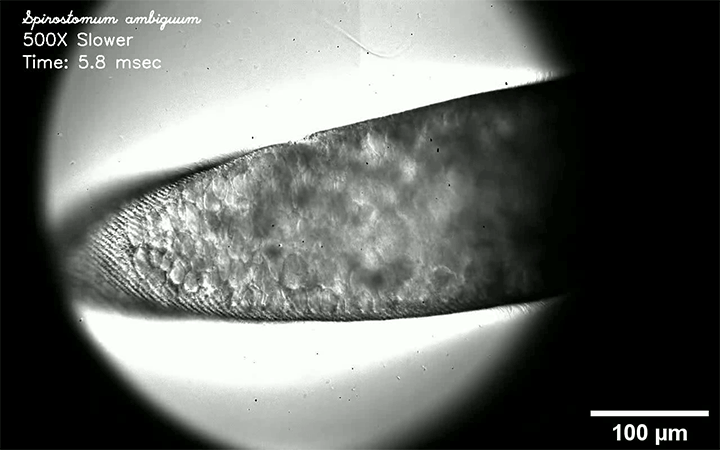
Respiratory diseases like measles, flu, tuberculosis, and COVID-19 are all transmitted by droplets. Some are tiny and airborne, capable of traveling long distances. Other drops are larger and only capable of traveling short distances. A new review paper consolidates what we know about these droplets and categorizes them by size and origin.
It turns out that a droplet’s size can tell us where it originated in the body. The largest type of droplets come from our mouths, lips, and tongues. Some form from filaments of saliva that stretch across our mouths and burst during exhalation. Others originate in our nasal passages where a sneeze can destabilize the mucus film there. These types of droplets are best suited to transmitting diseases that reside in the upper respiratory tract. Coughing, sneezing, singing, and speaking all produce these droplets, but breathing does not.
In contrast, the smallest classes of droplets come from the bronchial passages of the lungs, where films form after exhalation closes a passage. When we inhale again, the passage reopens, the film breaks up, and tiny droplets flow further into the lungs before getting exhaled. Breathing alone is enough to create and spread these tiny droplets, which are well-suited to spreading diseases that reside deep in the lungs, like tuberculosis.
In between these extremes are medium-sized droplets created from movement around our vocal cords. The formation mechanism for these droplets is least understood, but they are connected to breathing, coughing, speaking, singing, and so on.
Ultimately, understanding the mechanics of disease transmission is about knowing how to best prevent transmission. Knowing the size of droplets responsible for transmission lets us prioritize responses that work. For example, if large droplets are the primary transmission mechanism, loose-fitting masks and face masks will stop the spread. But for smaller droplets, ventilation measures and well-fitted N-95 respirators are the better choice. (Image credit: Anton; research credit: M. Pöhlker et al.; via APS Physics)