Though we may not often consider it, our bodies are full of fluid dynamics. Blood flow is a prime example, and, in this video, researchers describe their simulations of flow through the left side of the heart. Beginning with 3D medical imaging of a patient’s heart, they construct a computational domain – a meshed virtual heart that imitates the shape and movements of the real heart. Then, after solving the governing equations with an additional model for turbulence, the researchers can observe flow inside a beating heart. Each cycle consists of two phases. In the first, oxygenated blood fills the ventricle from the atrium. This injection of fresh blood generates a vortex ring. Near the end of this phase, the blood mixes strongly and appears to be mildly turbulent. In the second phase, the ventricle contracts, ejecting the blood out into the body and drawing freshly oxygenated blood into the atrium. (Video credit: C. Chnafa et al.)
Category: Research

Soap Film Grass
In the summer months, a breeze can set long grasses waving in an impressive display. Similar behaviors are seen in aquatic plants during tides. Researchers simulate the behavior in two-dimensions using a flowing soap film and nylon filaments. Flow visualization reveals the strong differences between flow above and between the grass. Vortices recirculate between the filaments at speeds much slower than the flow overhead. The instantaneous interaction of the high-speed freestream, the unsteady vortices, and the resistance of the grass results in familiar synchronous waves of grain. (Video credit: R. Singh et al.)

Balloon Explosion
These photos are shadowgraphs of a hydrogen flame exploding inside a balloon. The shadowgraph optical technique highlights density and temperature variations through their effect on a fluid’s refractive index. Here we see that the hydrogen flame has a strong cellular structure and is more turbulent than a methane flame. The cellular structure is a sign of an instability in the curved flame front. The instability and accompanying cellular appearance are a result of the complicated transport and reaction of fuel and oxidizer inside the flame. (Photo credits: P. Julien et al.)

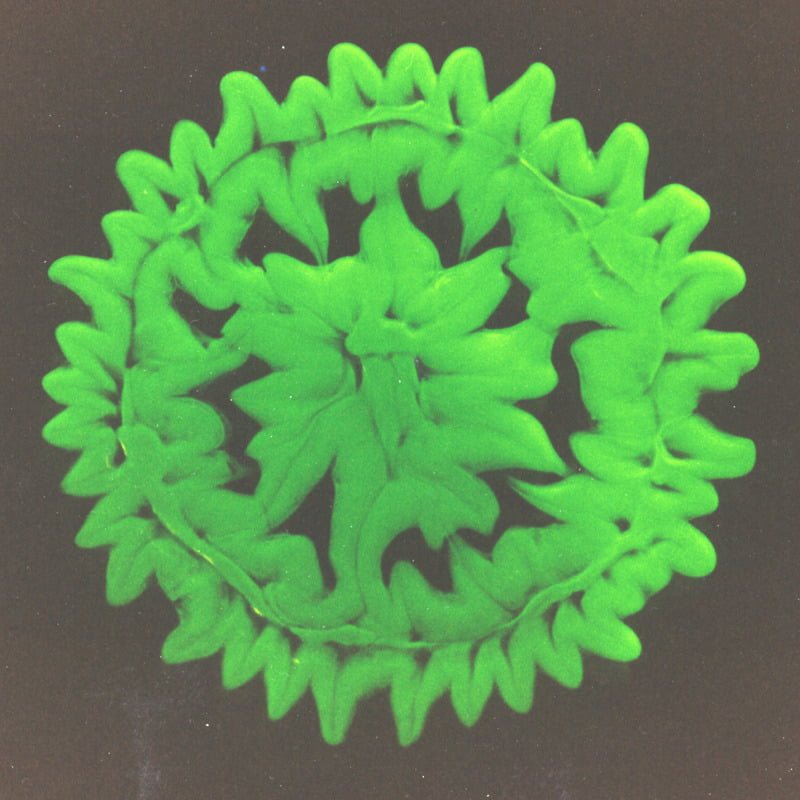
Distorted Rings
The Marangoni effect is generated by variations in surface tension at an interface. Such variations can be temperature-driven, concentration-driven, or simply due to the mixing between fluids of differing surface tensions as is the case here. The pattern in the image above formed after a dyed water droplet impacted a layer of glycerin. The initial impact of the drop formed an inner circle and outer ring. This image is from 30 seconds or so after impact, after the Marangoni instability has taken over. The higher surface tension of the water pulls the glycerin toward it, resulting in a flower-like pattern. (Photo credit: E. Tan and S. Thoroddsen)

Stirring Up
When a viscoelastic non-Newtonian fluid is stirred, it climbs up the stirring rod. This behavior is known as the Weissenberg effect and results from the polymers in the fluid getting tangled and bunched due to the stirring. You may have noticed this effect in the kitchen when beating egg whites. In this video, researchers explore the effect using rodless stirring. The first example in the video shows a viscous Newtonian fluid being stirred. The stirring action creates a concave shape in the glycerin-air interface, and dye injection shows a toroidal vortex formed over the stirrer. Fluid near the center of the vortex is pulled downward and circulates out to the sides. In contrast, the viscoelastic fluid bulges outward when stirred. Dye visualization reveals fluid being pulled up the center into the bulge. It then travels outward, forming a mushroom-cap-like shape before sinking down the outside. This is also a toroidal vortex, but it rotates opposite the direction of the Newtonian one. Exactly how the polymers create this change in flow behavior is a matter of active research. (Video credit: E. Soto et al.)

Brazuca
Since 2006, Adidas has unveiled a new football design for each FIFA World Cup. This year’s ball, the Brazuca, is the first 6-panel ball and features glued panels instead of stitched ones. It also has a grippy surface covered in tiny nubs. Wind tunnel tests indicate the Brazuca experiences less drag than other recent low-panel-number footballs as well as less drag than a conventional 32-panel ball. Its stability and trajectory in flight are also more similar to a conventional ball than other recent World Cup balls, particularly the infamous Jabulani of the 2010 World Cup. The Brazuca’s similar flight performance relative to a conventional ball is likely due to its rough surface. Like the many stitched seams of a conventional football, the nubs on the Brazuca help trip flow around the ball to turbulence, much like dimples on a golf ball. Because the roughness is uniformly distributed, this transition is likely to happen simultaneously on all sides of the ball. Contrast this with a smooth, 8-panel football like the Jabulani; with fewer seams to trip flow on the ball, transition is uneven, causing a pressure imbalance across the ball that makes it change its trajectory. For more, be sure to check out the Brazuca articles at National Geographic and Popular Mechanics, as well as the original research article. (Photo credit: D. Karmann; research credit: S. Hong and T. Asai)

Going Supercritical
Supercritical fluids exist at temperatures and pressures above the critical point, in a region of the phase diagram where there is no clear boundary between the liquid and gaseous state. Supercritical fluids have some of the properties of each state: they can move as freely as a gas, but they are still capable of dissolving materials like a liquid does. They also have no surface tension because there is no interface between liquid and solid. These properties make supercritical fluids very useful in industrial applications, including decaffeination and chemical deposition. Interestingly, the temperatures and pressures on Venus are so high that scientists think the atmosphere at the surface is a supercritical fluid. (Video credit: SCFED Project)

The Upside-Down Jellyfish
The upside-down jellyfish, Cassiopea, rests its bell against the ocean floor and points its frilly oral arms up toward the sun for the benefit of the symbiotic algae living on it. In return, the algae provide some of the nutrients the jellyfish needs. The rest it obtains by filter feeding for zooplankton. The video above shows how a combination of flow visualization and simplified computational modeling can reveal the jellyfish’s methods for eating. A simple pulsing bell has limited fluid flow in the region of the jellyfish’s mouths, but the addition of a permeable layer (representative of the oral arms) significantly enhances mixing. (Video credit: T. Rodriguez et al.)

Wave Tank
A new wave tank facility opening at the University of Edinburgh promises new capabilities to simulate ocean wave behavior. The circular 25m diameter wave tank is lined with 168 wave makers and is equipped with 28 submerged flow-drive units. Together, these allow the tank to simultaneously simulate nearly any wave type as well as tidal currents up to 1.6 m/s. The facility is intended for 1/20th scale modeling; projected to full-size, this means that the tank is capable of making waves representative of 28 m high ocean waves and tidal currents in excess of 12 knots. It’s expected to be particularly valuable in the development and testing of wave and tidal motion generators for clean energy. For more, see BBC News and FloWave’s own website. (Image credit: Brightspace/BBC News; submitted by srikard)

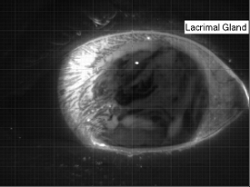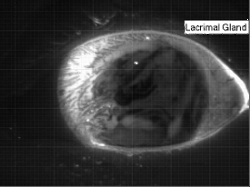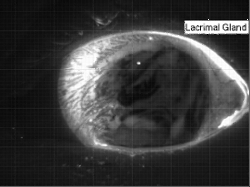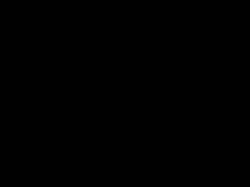
Tear Films
The human eye has a thin tear film over its surface to maintain moisture and provide a smooth optical surface. The film consists of multiple layers: a lipid layer at the air interface to decrease surface tension and delay evaporation; an aqueous middle layer; and an inner layer of hydrophilic mucins that keep the film attached to the eye. The entire film is a few microns thick, with the lipid layer estimated to be only 50-100 nm thick and the mucin layer just a few tenths of a micron. The aqueous portion of the tear film is supplied from the lacrimal gland in the corner of the eye. In the animation above, the fresh aqueous fluid is fluorescent. It gathers in the corner of the eye several seconds after a blink due to reflex tearing. The tear fluid then flows around the outer edges of the eye until the subject blinks and the fresh tear gets distributed throughout the film. (Research credit: L. Li et al.; original video)