Stretching from the southern tip of Florida, a chain of low-lying islands, known as keys or cays, formed underwater during a warm interglacial period some 125,000 years ago. Originally coral reefs and sand bars, the islands hardened and fossilized when sea levels dropped during an ice age. These natural-color satellite images hint at the keys’ impressive ecosystems. The bright blue streak is a giant coral reef separating the deeper waters of the Atlantic from the shallow waters and sea-grass beds lying between the islands. Formations like these, along with mangrove forests, are part of nature’s way to mitigate the damage and flooding caused by hurricanes. Unfortunately, warmer seas and rising sea levels now threaten the keys. (Image credit: L. Dauphin/USGS; via NASA Earth Observatory)
Month: August 2022

Encapsulating Drops
Sometimes a droplet needs a little protection while it’s traveling to its destination. When that’s the case, we often try to encapsulate it in a layer of material that won’t be affected by whatever environment the drop is traveling through. In this study, researchers aimed to give their drops not one but two layers of protection — in as simple a way as possible.
The team began with three layers of liquid. The lowest layer was water, the middle layer was an oil, and the top layer was a mixture of water and isopropyl alcohol. Next, they added glass particles that were denser than the alcohol, but less dense than the oil. This caused the particles to form a clump — a granular raft — along the interface between the alcohol and the oil (not shown). When the layer of particles became heavy enough, it began to sink into the oil, carrying some of the alcohol with them. This conglomeration formed the initial droplet of alcohol mixture encased in an armor of glass beads.
As this armored droplet sank, it approached the second interface: the oil-water interface. At this juncture, the team observed three different outcomes. When the glass particles were small or light, the armored drop would come to a rest at the oil-water interface. As the drop deformed, water would pierce the armor, causing the whole drop to rupture (Image 1).
In the second case, heavier particles caused the armored drop to sink through the oil-water interface, but a low oil viscosity meant that the oil film drained from the bottom of the drop before the drop was fully encapsulated. Once again, this let the water through and ruptured the droplet (Image 2).
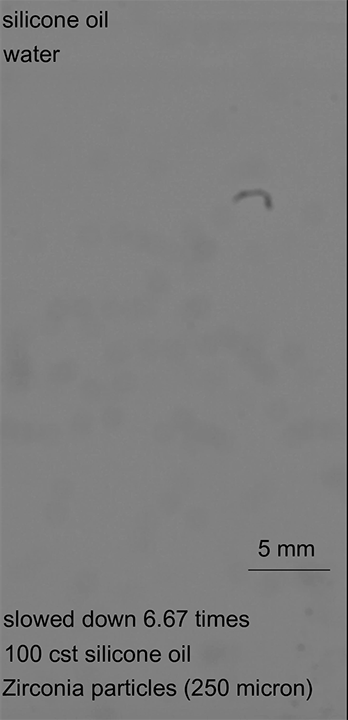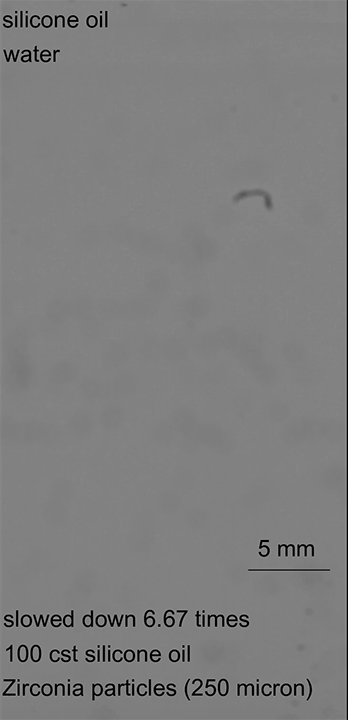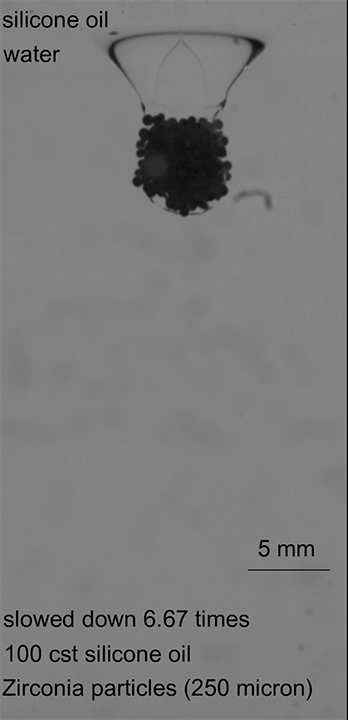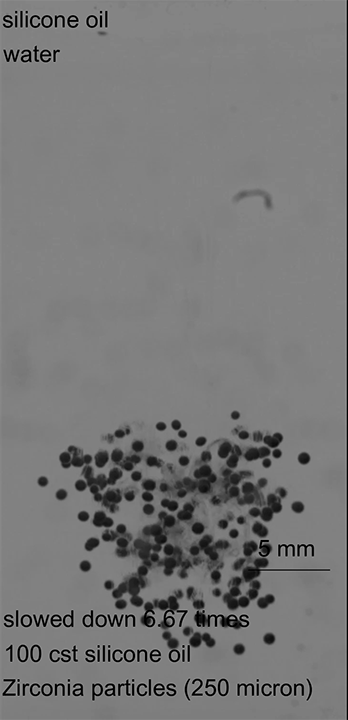
In the final case, armored drops with just the right bead density and oil viscosity would sink through the oil-water interface until the oil pinched off behind the drop. This pinch-off allowed the oil to redistribute around the drop, encapsulating it in layers of both oil and particles, thereby protecting it as it continued its journey (Image 3). (Image credits: top – Girl with red hat, experiment – A. Hooshanginejad et al.; research credit: A. Hooshanginejad et al.)

When Reservoirs Run Dry
With the ongoing megadrought in the U.S. Southwest, more and more reservoirs are reaching historic low water levels. So it’s worth asking: what happens when a reservoir runs dry? And what, exactly, does a reservoir do in the first place? In this Practical Engineering video, Grady tackles both questions and takes a look at the many disciplines — beyond just civil engineering — that go into making a functional reservoir. (Image and video credit: Practical Engineering)

“Art of Paint”
Filmmaker Roman De Giuli is always coming up with spectacular and visually fascinating new ways to manipulate ink and other liquids. In “Art of Paint,” he applies thin layers atop a custom plate that can be tilted in any direction. The results sometimes resemble acrylic paint pours, sometimes Marangoni flows, and sometimes look more like salt fingers or Rayleigh-Taylor instabilities. The extreme variety of forms is quite unique among these sorts of films and is well worth taking the time to view in fullscreen. (Image and video credit: R. De Giuli)

Slow to Relax
Oobleck is a decidedly weird substance. Made from a dense suspension of cornstarch in water, oobleck is known for its mix of liquid-like and solid-like properties, depending on the force that’s applied. In a recent study, researchers took a look at what happens when you really push oobleck to the extreme. When the force applied to oobleck is small or slowly added, the water between cornstarch particles helps keep the particles apart and free of contact. It’s when the force is large that those particles start jamming up against each other and having friction between them, and then the oobleck suddenly acts like a solid. But what happens once that force is removed?
When the force is gone, we expect the particles to repel and for water to squeeze back into the spaces between them, breaking up the friction and allowing the oobleck to relax back to a liquid-like form. But the team found that sometimes the oobleck doesn’t relax as easily as expected; instead, it seems to retain some memory of its solid-like state, due to persisting friction between particles. (Image credit: T. Cox; research credit: J. Cho et al.)

The Tea Leaves Effect
If you’ve ever stirred a cup of tea with loose leaves in it, you’ve probably noticed that the leaves tend to swirl into the center of the cup in a kind of inverted whirlpool. At first, this behavior can seem counter-intuitive; after all, a spinning centrifuge causes denser components to fly to the outside. In this video, Steve Mould steps through this phenomenon and how the balance of pressures, velocities, densities, and viscosity cause the effect. (Note that Mould uses the term “drag,” but what he’s really referring to is the boundary layer across the bottom of the container. But who wants to explain a boundary layer in a video when they can avoid it?) (Video and image credit: S. Mould)

When liquid in a cup is stirred, the densest layers move to the center. 
Bubbles in Turbulence
In nature and industry, swarms of bubbles* often encounter turbulence in their surrounding fluid. To study this situation, researchers used numerical simulation to observe bubbles across a range of density, viscosity, and surface tension values relative to their surroundings. They found that density differences between the two fluids made negligible changes to the way bubbles broke or coalesced.
In contrast, viscosity played a much larger role. More viscous bubbles were less likely to deform and break, thanks to their increased rigidity. When looking at small deformations along the bubble interface, both density and viscosity had noticeable effects. With increasing bubble density, they observed more dimples on the interface; increasing the viscosity had the opposite effect, making the bubbles smoother. (Image credit: Z. Borojevic; research credit: F. Mangani et al.)
*We usually think of bubbles as air or another gas contained within a liquid. But this study’s authors use the term “bubble” more broadly to mean any coherent bits of fluid in a different surrounding fluid. Colloquially, this means their results apply to both bubbles and drops.

Actinoform Clouds
Flower-shaped actinoform clouds, like those seen on the left side of this satellite image, were only discovered in the 1960s once satellite imagery allowed meteorologists to identify cloud structures that were too large to recognize from the ground. Often appearing over the ocean, these clouds can stretch over hundreds of kilometers, bringing drizzling rain.
This particular set of actinoform clouds have some distinctive neighbors in the right side of the image, where V-shaped slashes through the cloud cover mark the origins of two von Karman vortex streets. The vortex streets appear downwind of two rocky islands, Alejandro Selkirk Island and Robinson Crusoe Island. (Image credit: L. Dauphin; via NASA Earth Observatory)

“Timedrift II”
As a teenager, I climbed Mount Kilimanjaro. The final ascent began around midnight, and we climbed through the dark, through sunrise, and into the early morning. I remember pausing at one point, just as the sun was rising, and looking out at the clouds thousands of meters below. From that height, they looked like an ocean, rippled with lavender waves. Timelapse films like this one, by filmmaker Martin Heck, remind me of that morning and the sense that I had of the sky as an ocean, flowing, crashing, and surging in ways we cannot appreciate until we slow down and look closer. (Image and video credit: M. Heck/Timestorm Films)

Rotating Waves of Grains
Rotating drums are a popular way to explore granular dynamics. Here, researchers fill a cylinder (seen below) with heavy grains and a low-viscosity fluid, then rotate the mixture about a horizontal axis. This sets up a contest between centrifugal forces and gravitational forces on the grains. At the right rotation rates, the grains form annular rings around the outside of the cylinder, where they rotate at a different speed than the fluid. This difference in speed between the two layers can trigger a Kelvin-Helmholtz instability and cause waves along the interface between the grains and the fluid, as seen in the examples above. (Image and research credit: V. Dyakova and D. Polezhaev; top image adapted by N. Sharp)