Catalysts speed up chemical reactions without being consumed themselves. In “Catalysis” the Beauty of Science team shows 5 different examples of catalytic reactions, from acetone oxidation to yeast fermentation. The film is full of bubbles, sparks, and wave-like pulses of chemical reaction. As always, it’s a lovely glimpse of processes we’re not used to watching so closely. (Image and video credit: Beauty of Science)
Month: January 2021

Sand Traps
Antlion larvae catch prey by digging conical pits in sand. The steep walls of the trap are near the angle of repose, the largest angle a granular material can maintain before grains slide down. When a hapless ant wanders into the trap, the antlion throws sand from the center of the pit, triggering a sandslide that carries the ant downward. The act of flinging sand also helps the antlion maintain the pit, correcting any disruptions to the pit’s steep sides caused by its flailing prey. (Image and research credit: S. Büsse et al.; via Science)

Following the Flow
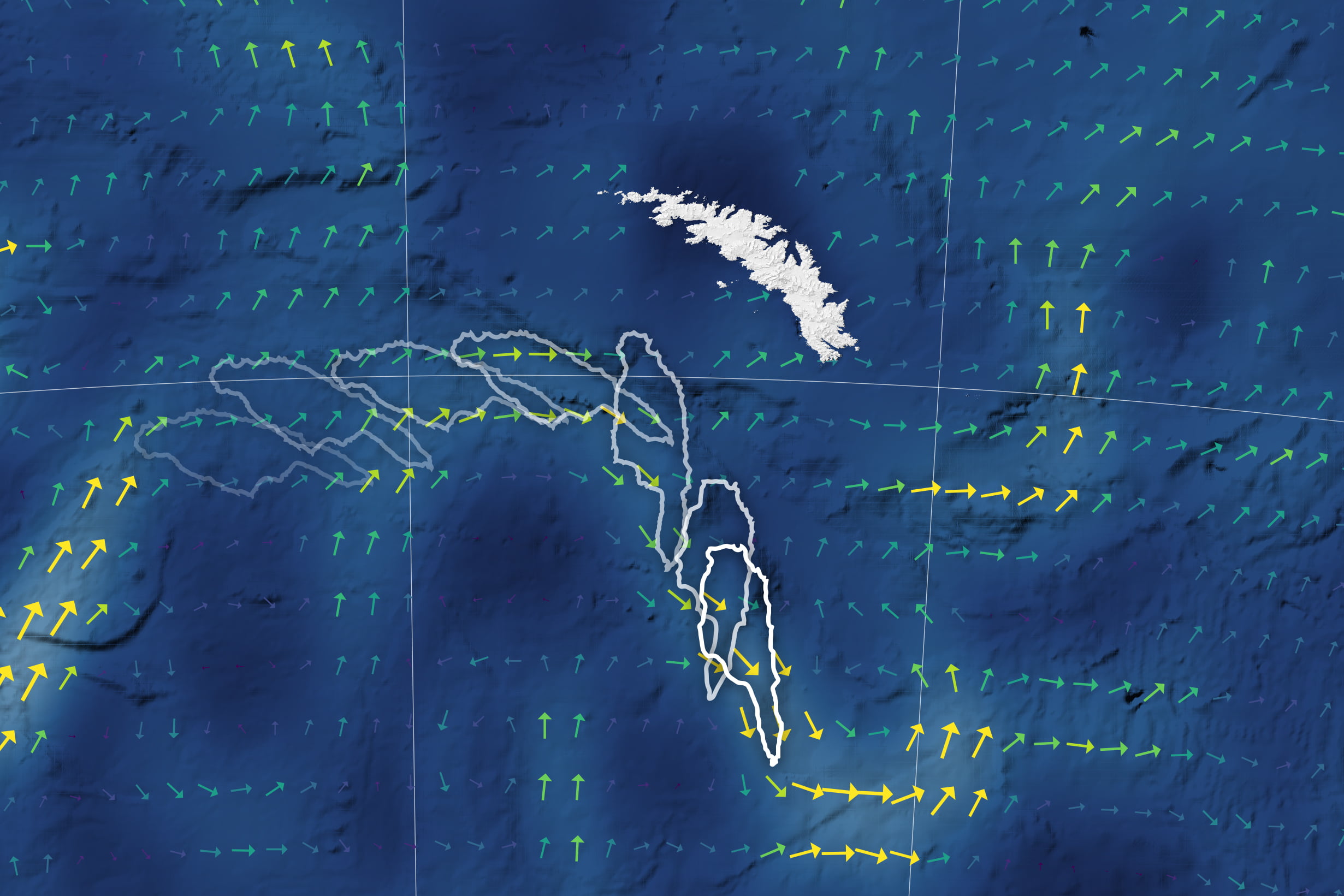
In early December 2020, the world’s largest iceberg — roughly 135 km long by 44 km wide — was heading straight for South Georgia Island. Luckily for the island, iceberg A-68A was being carried by ocean surface currents that approach the island before turning sharply southward. The enormous iceberg followed, rotating nearly 90 degrees and drifting away on faster currents.
Scientists track these large-scale — 50 to 100 km wide — currents using satellites that measure the ocean height. Currents of this size actually generate a measurable tilt to the ocean surface, which scientists measure and use as input into models that estimate the surface currents’ speed and direction. (Image credit: L. Dauphin and J. Stevens; via NASA Earth Observatory)

Uncovering Erosion Patterns
Gypsum and limestone cliffs sometimes form patterns of long, parallel grooves known as rillenkarren. Recent research shows that these patterns form when a thin layer of water flows over a dissolvable surface. As the running water picks up solute, its concentration increases, causing changes in the local hydrodynamics. What begins as a small perturbation in an otherwise flat surface grows into a groove with walls that eventually rise out of the water layer. At that point, the growth mechanism shifts because the flow is restricted to channels in the rock. (Image credit: Ymaup/Wikimedia Commons; research credit: A. Guérin et al.; via APS Physics; submitted by Kam-Yung Soh)

Breaking Bubbles
What do a nineteenth-century war ship, a sardine-hunting shark, and a viral bottle trick have in common? Cavitation! The phenomenon of cavitation occurs when a fluid is accelerated such that its local pressure drops below the vapor pressure. As a result, bubbles form and then violently collapse, creating shock waves that can damage nearby surfaces or stun prey. Dianna explains — and reveals some cool historical context that was new to me! — in the video above. (Image and video credit: Physics Girl)

Bubble Array
Surface tension tries to minimize a bubble‘s surface area, which is why bubbles assume a spherical shape. But when many bubbles clump together, a curved interface is not always the most energy efficient one. In this case, bubbles can take on many shapes and sizes while still minimizing the overall surface energy. Take a close look at this image and see what shapes you discover! (Image credit: M. Adil)

Hedgehogs Atop Waves
Since Michael Faraday, scientists have watched the curious patterns that form in a vibrating liquid. By adding floating particles to such a system, researchers have discovered spiky, hedgehog-like shapes that form near the surface. At low amplitudes, the surface patterns resemble the typical smooth rounded lobes one would expect, but as the wave amplitude increases, spikes form in the tracers, driven by the motion of the waves. (Image and research credit: H. Alarcón et al.; via APS Physics)

Stabilizing Foams
Bubbles in a pure liquid don’t last long, but with added surfactants or multiple miscible liquids, bubbles can form long-lasting foams. In soapy foams, surfactants provide the surface tension gradients necessary to keep the thin liquid layers between bubbles from popping. But what stabilizes a surfactant-free foam?
New work finds that foams in mixtures of two miscible fluids only form when the surface tension depends nonlinearly on the concentration of the component liquids. When this is true, thinning the wall between bubbles creates changes in surface tension that stabilize the barrier and keep it from popping.
In mixtures without this nonlinearity, foams just won’t form. The new results are valuable for manufacturing, where companies can avoid unintentional foams simply by careful selection of their fluids. (Image credit: G. Trovato; research credit: H. Tran et al.; via APS Physics; see also Ars Technica, submitted by Kam-Yung Soh)

The Galloping Starfish
Starfish won’t win any sprints, but they’re actually quite good at moving around as they hunt for prey. Without brains, starfish are led by their feet, which pull in the direction of food they scent. Each foot is connected to what amounts to an internal hydraulic system within the starfish. With a combination of secreted adhesive and pumping, the starfish can trundle along. (Image and video credit: Deep Look)

Oil in Water
In the decade since the Deepwater Horizons oil spill, scientists have been working hard to understand the intricacies of how liquid and gaseous hydrocarbons behave underwater. The high pressures, low temperatures, and varying density of the surrounding ocean water all complicate the situation.
Released hydrocarbons form a plume made up of oil drops and gas bubbles of many sizes. Large drops and bubbles rise relatively quickly due to their buoyancy, so they remain confined to a relatively small area around the leak. Smaller drops are slower to rise and can instead get picked up by ocean currents, allowing them to spread. The smallest micro-droplets of oil hardly rise at all; instead they remained trapped in the water column, where currents can move them tens to hundreds of kilometers from their point of release. (Image and research credit: M. Boufadel et al.; via AGU Eos; submitted by Kam-Yung Soh)