Droplet rebound is frequently associated with superhydrophobic surfaces but can also be generated by very large temperature differences. For very hot substrates, a thin layer of the drop vaporizes on contact via the Leidenfrost effect and helps a drop rebound by preventing it from wetting the surface. This video shows almost the opposite: a water droplet hitting solid carbon dioxide (-79 degrees C). Upon contact, the solid carbon dioxide sublimates, creating a thin layer of gas that separates the droplet from the surface. You can also see the vortex ring that accompanies the drop’s impact. Water vapor near the carbon dioxide surface has condensed into tiny airborne droplets that act as tracer particles that reveal the vortex’s formation and the rebounding droplet’s wake. (Video credit: C. Antonini et al.; Research paper)
Tag: science

Navigating the Interface
Walking on water may be the stuff of legend at human scales, but it’s a fact of everyday life for many smaller species. Waxy, hydrophobic coatings typically make such insects’ points of contact (feet, legs, etc.) water-repellent, and their light weight can be supported by surface tension. Navigating the interface between air and water is more complicated, though, and these creatures have evolved several mechanisms to help. Some, like water striders, use appendages they insert below the surface for propulsion. At 0:49 in the montage above, you can see flow visualization of the vortices generated by a stroke. Other insects release a chemical in their wake that lowers the local surface tension and drives them away via the Marangoni effect. For more, see here and especially this Physics Today article. (Video credit: D. Hu and J. Bush)

Fluids Round-up – 11 August 2013
Time for another fluids round-up! Here are your links:
- Back in January 1919, a five-story-high metal tank full of molasses broke and released a wave of viscous non-Newtonian fluid through Boston’s North End. Scientific American examines the physics of the Great Molasses Flood, including how to swim in molasses. If you can imagine what it’s like to swim in molasses, you’ll know something of the struggle microbes experience to move through any fluid. They also discusses some of the strange ways tiny creatures swim.
- In sandy desert environments, helicopter blades can light up the night with so-called helicopter halos. The effect is similar to what causes sparks from a grinding wheel. Learn more about this Kopp-Etchells effect.
- Check out this ominous footage of a tornadic cell passing through Colorado last week.
- If you want more of a science-y look to your drinkware, you should check out the Periodic TableWare collection over on Kickstarter.
- Finally, wingsuits really take the idea of gliding flight to some crazy extremes. Check out this video of in-flight footage. Watch for the guy’s wingtip vortices at 3:16 (screencap above)! (submitted by Jason C)
(Photo credit: Squirrel)

Contaminants Flowing Uphill
Here’s an example of some baffling fluid dynamics. Researchers have found that, when pouring a fluid from one container into a lower one containing a fluid with floating particulates, it’s possible for the floating particles to travel upstream against gravity and the flow. The phenomenon is driven by surface tension. The particulates floating in the lower container decrease the fluid’s surface tension relative to the pure fluid pouring in from above. This creates a gradient in surface tension that, via the Marangoni effect, drives a small flow upstream, in the direction of the greater surface tension. In the video above, this flow takes the form of two recirculating vortices in the pouring channel, oriented such that their upstream velocities run along the outside of the channel. Occasionally this flow draws particulates up the waterfall and into the recirculating zones, creating upstream contamination. We reported this previously, but the researchers have now released videos demonstrating the effect, including in pipettes and a water flume. Usually it’s taken for granted that matter cannot move upstream, so this could be a game-changer, especially at small scales where surface tension already dominates. For more, see their paper. (Video credit: S. Bianchini et al.)

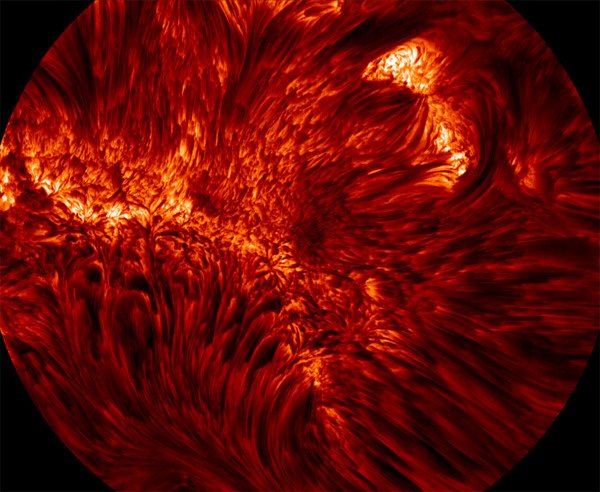
Convection on the Sun

New photographs showing ultra-fine structure in the sun’s chromosphere and photosphere have been released. They offer a fascinating view into the magnetohydrodynamics of the sun, where the fluid behaviors of plasma are constantly modified by the sun’s magnetic field. The left image shows fine-scale magnetic loops rooted in the photosphere, while the right image shows our clearest photo yet of a sunspot. The dark central portion is the umbra, where magnetic field lines are almost vertical; it’s surrounded by the penumbra, where field lines are more inclined. Further out, we see the regular convective cell structure of the sun. (Photo credit: Big Bear Solar Observatory/NJIT; via io9 and cnet)

Bubbles With Tails
In water and other Newtonian fluids, a rising bubble is typically spherical, but for non-Newtonian fluids things are a different story. In non-Newtonian fluids the viscosity–the fluid’s resistance to deformation–is dependent on the shear rate and history–how and how much deformation is being applied. For rising bubbles, this can mean a teardrop shape or even a long tail that breaks up into fishbone-like ligaments. The patterns shown here vary with the bubble’s volume, which affects the velocity at which it rises (due to buoyancy) and thus the shear force the bubble and surrounding non-Newtonian fluid experience. (Video credit: E. Soto, R. Zenit, and O. Manero)

Bouncing in Lockstep

Droplets of silicone oil bounce on a pool of the same thanks to the vibration provided by a loudspeaker. Each droplet’s bounce causes ripples in the pool and the interference between these ripples fixes the droplets in lockstep with one another. As long as the vibration continues to feed the thin layer of air that separates the droplets from the pool during each bounce and no impurities break the surface tension at the interface, the droplets will bounce indefinitely on their liquid trampoline. Such systems can be used to observe quantum-mechanical behavior like wave-particle duality on a macro-scale. (Photo credit: A. Labuda and J. Belina)

Flow Over a Delta Wing
Fluorescent dye illuminated by laser light shows the formation and structure of vortices on a delta wing. A vortex rolls up along each leading edge, helping to generate lift on the triangular wing. As the vortices leave the wing, their structure becomes even more complicated, full of lacy wisps of vorticity that interact. Note how, by the right side of the photo, the vortices are beginning to draw closer together. This is an early part of the large-wavelength Crow instability. Much further downstream, the two vortices will reconnect and break down into a series of large rings. (Photo credit: G. Miller and C. Williamson)

Self-Assembling Ferrofluids
Ferrofluids–colloidal suspensions made up of ferromagnetic nanoparticles and a carrier liquid–are known for their interesting and sometimes bizarre behaviors due to magnetic fields. The video above shows how, when subjected to an increasing magnetic field, a single droplet of a ferrofluid on a superhydrophobic surface will split into several droplets. The process is called static self-assembly, and it results from the ferrofluid seeking a minimum energy state relative to the force supplied by the magnetic field. Change the magnetic field and the droplets shift to the next energy minimum. But what happens when you change the magnetic field continuously and too quickly for the droplets to respond? A whole different set of structures and behaviors are observed (video link). This is dynamic self-assembly, a different ordered state only achieved when the ferrofluid is forceably kept away from the energy minima seen in the first video. For more, see the additional videos and the original paper. (Video credit: J. Timonen et al.; via io9)

“Perpetual Puddle Vortex Experiment”
Anthony Hall’s “Perpetual Puddle Vortex Experiment” is an intriguing display of several physical mechanisms. What looks like a puddle is actually a vortex constantly sucking fluid down a hole in the table. The liquid is re-circulated into the puddle so it never disappears. The table itself is treated to be hydrophobic, causing the distinctive curvature and large contact angle of the puddle’s rim. The oils mixed in float on top, creating patterns of foam that visualize the swirling motions of the fluid as the vortex pulls it in. (Video credit and submission by: A. Hall)