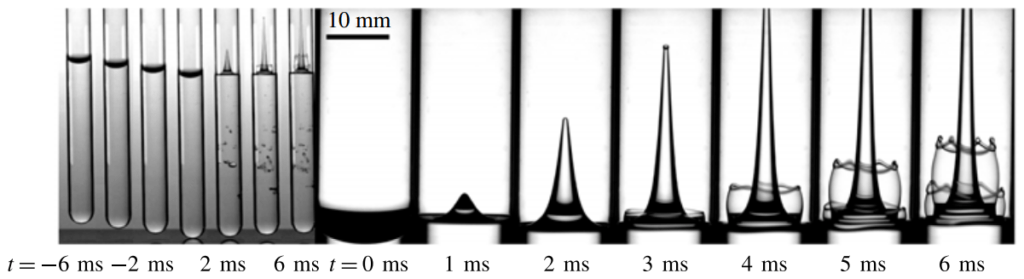
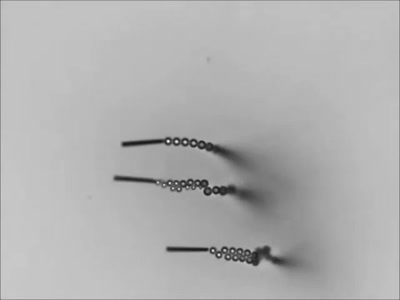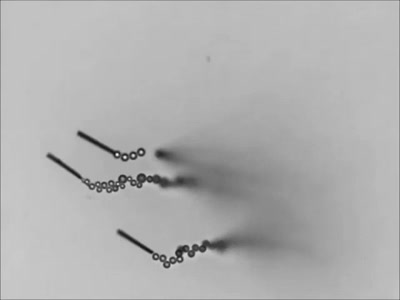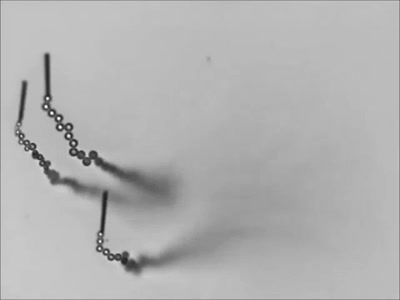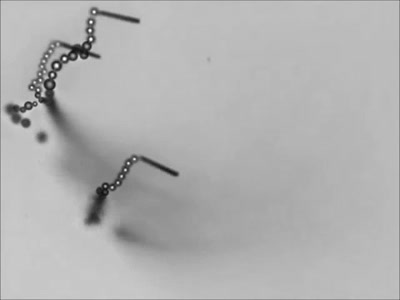
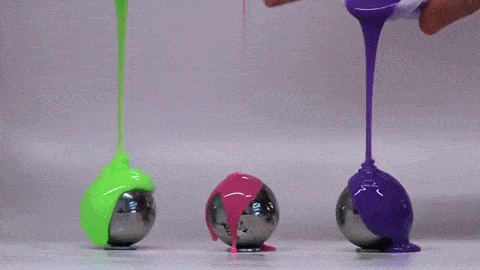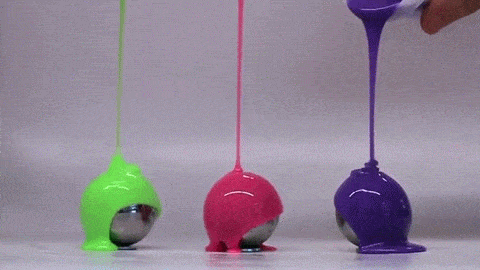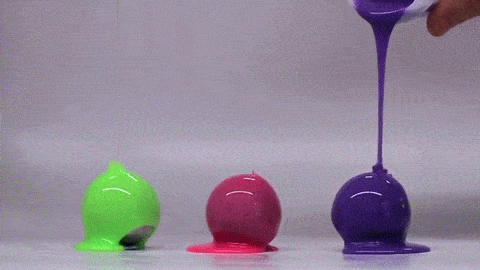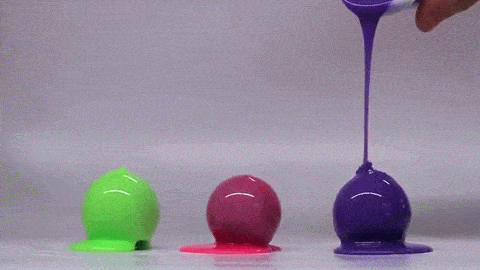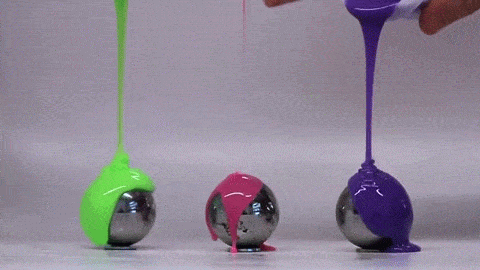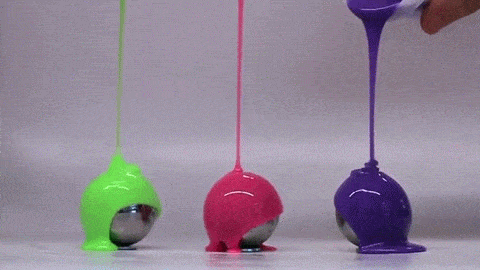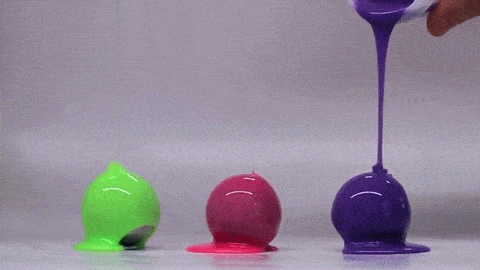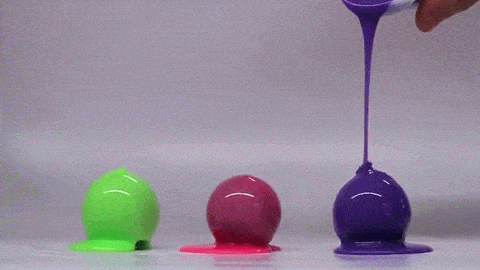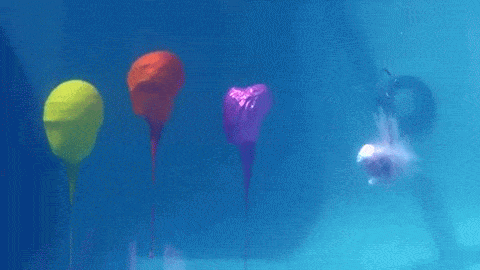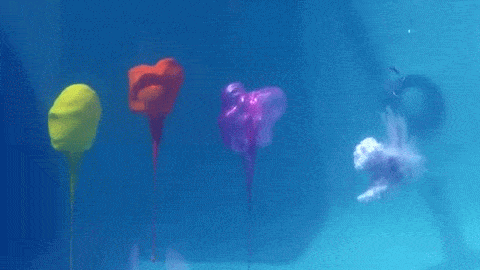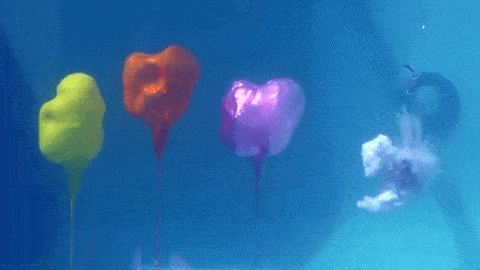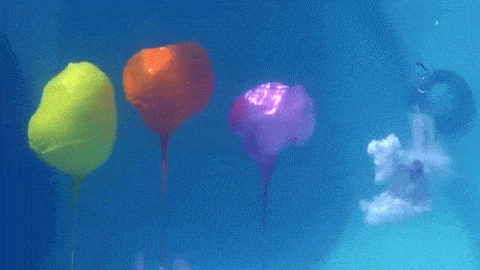Dropping a partially-filled test tube of water against a table makes the meniscus at the air-water interface invert into a jet of liquid. In some cases, the impact is strong enough to generate splashing crowns of water around the base of the jet. These crowns come in two forms – one with many splashes layered upon one another and the other with only a few splashes and a faster jet.
The many-layered splash crowns come from the pressure wave that reflects back and forth from the bottom of the tube to the surface and back. This pressure wave moves at the speed of sound and vibrates the water surface, creating the many splashes. The same reflected pressure wave occurs in the second type of splash crown, but it gets disrupted by cavitation bubbles that form in the water (visible in the lower left image). Instead the splash crowns form from the shock waves generated when the cavitation bubbles collapse. (Image credits: A. Kiyama et al.)