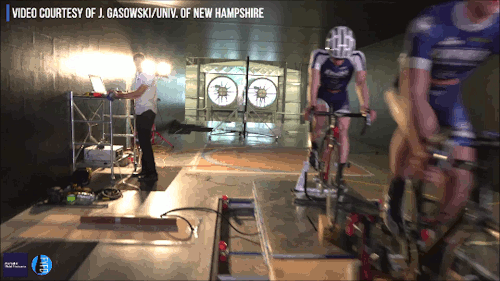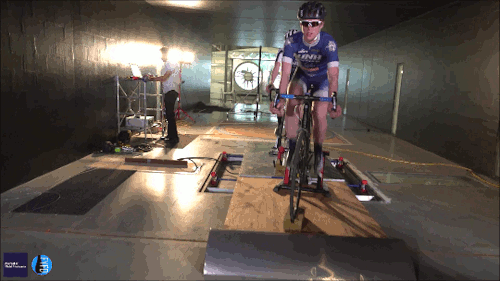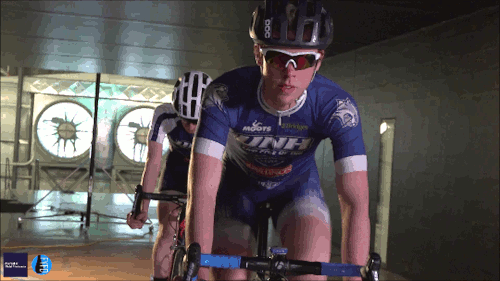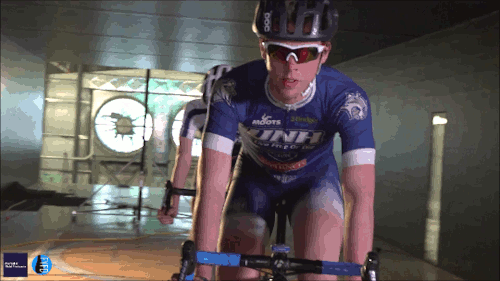There are many ways to make a droplet oscillate in a star-shape – like vibrating its surface or using acoustic waves to excite it – but these methods involve externally forcing the droplet’s oscillation. Leidenfrost drops – liquids levitating on a film of their own vapor caused by the extremely hot surface below – turn themselves into stars. It all starts with the constant evaporation driven by the heat below. This creates a thin, fast-moving layer of vapor flowing beneath the drop. That vapor shears the drop, causing capillary waves – essentially ripples – that travel through the drop in a characteristic way. Those ripples in turn cause pressure oscillations in the vapor layer, alternately squeezing and releasing it. Feedback from the vapor layer then drives the droplet into star-shaped oscillations. Under the right conditions, water drops can form stars with as many as 13 points! (Image and research credit: X. Ma and J. Burton, source)
Tag: physics

Swirling Blooms
Every summer, as the ice melts, the waters of the Chukchi Sea off the Alaskan coast come alive with phytoplankton blooms. In satellite images like this one, they can look like abstract paintings formed from swirling colors. In the Chukchi Sea, two main currents collide. One, water from the Bering Sea, is cold, salty, and nutrient-rich. This is the preferred home to phytoplankton known as diatoms, which are responsible for some of the greenish hues seen here.
Coccolithophores, another variety of phytoplankton, prefer the warmer, less salty Alaskan coastal waters. Despite a relative lack of nutrients, the coccolithophores thrive, creating the milky turquoise color seen in the image. Knowing these characteristics of the phytoplankton, observing the growth of blooms over time may tell scientists about how the flows in these areas shift and change from year to year. (Image credit: NASA; via NASA Earth Observatory)
Pyrocumulus on the Horizon
The Cranston wildfire in California is intense enough that it’s creating its own weather. This timelapse video shows the formation and growth of a pyrocumulus cloud, also associated with volcanoes, over the wildfire. In both instances, the extreme heat causes a massive column of hot, turbulent air to rise. Because ash and smoke are carried upward as well, there are many places for any moisture in the atmosphere to nucleate, forming the cloud we see. In timelapse, the roiling nature of the air’s motion is especially apparent. This turbulence can be dangerous, as it may contribute to high winds and even lightning, both of which can spread the fire further. (Video credit: J. Morris; via James H.)

What Keeps a Foam Intact
Beer, soda, soap, meringue – foams are everywhere in our lives. But have you ever wondered why some foams disappear so quickly while whipped egg whites stick around? That’s the subject of this Gastrofisica video, which is in Spanish but has English captions.
Foams form when air gets introduced into a liquid, but for those bubbles to stick around, they need a certain special something. With soapy water, that ingredient is surfactants, molecules with both hydrophobic (water-fearing) and hydrophilic (water-loving) ends, which line up at the interface of the foam and help hold it together. But surfactants are relatively weak, especially compared to to the albumin proteins in an egg white. By whipping egg whites, you’re effectively untangling those proteins, and, like surfactants, they line up at the interface of the foam so that their hydrophobic and hydrophilic parts can hang out in their preferred mediums. With so many similar molecules crowded together, the proteins coagulate, adding extra strength and stiffness to your whipped egg whites. (Video and image credit: Tippe Top Physics; h/t to MinutePhysics)


A Burst of Microdroplets
If you hold a bubbly beverage like champagne or soda near your face, you’ll feel a light mist of tiny, nearly invisible droplets.These droplets form when bubbles reach the surface and pop, generating a tiny jet that ejects an even tinier droplet, as shown in the animation above. This process is remarkably common; its occurrence in the ocean results in billions of tons of sea salt entering our atmosphere each year. Since these tiny microdroplets stay aloft for far longer than their larger brethren, understanding how they form and just how small they can be is vital for understanding their impact on climate, pathogen spreading, and other topics. A new study suggests that the minimum size for an ejected droplet is just 1% of the size of the bubble that births it. (Image and research credit: C. F. Brasz et al., source)

Swimming, Cycling, and Sailing
Summer brings with it lots of great sports, and whether you love riding a bike, sailing a boat, or just hanging out at the pool, our latest FYFD/JFM video has something for you. Want even more sports physics? Check out the Olympic series we did for the London and Rio games. And if you’re looking for more of the latest fluids research, don’t miss the rest of our video series. (Video and image credit: N. Sharp and T. Crawford)

“Le Temps”
Thomas Blanchard is back with another beautiful music video. This one features ink cascading over various shapes underwater. Lots of tiny mushroom-shaped Rayleigh-Taylor instabilities here caused by the ink’s greater density compared to the surrounding water. There are also some lovely examples of transitional flow, especially around the spheres. Initially, flow over the spheres looks completely smooth and laminar. But, on the latter half of the sphere, where the flow is under increasing pressure, you see disturbances growing until little fingers of ink break away entirely. Be sure to watch the whole video; you don’t want to miss this! (Video and image credit: T. Blanchard)

Convection Without Heat
We typically think of convection in terms of temperature differences, but the real driver is density. In the animations above, cream sitting atop a liqueur is undergoing solutal convection – no temperature difference needed! The alcohol in the liqueur mixes with the cream to form a lighter mixture that rises to the surface. The lower surface tension of the alcohol is also good at breaking up the cream, forming little cells. As the alcohol in those cells evaporates, the cream gets heavier and sinks down into the liqueur, where it can pick up more alcohol, rise back to the surface, and begin the cycle again. (Image credit: J. Monahan et al., source)

A Viscous Splash
The splash of a drop may be commonplace, but it is still a mesmerizing and fertile phenomenon. When it comes to splashing, scientists are still learning how to predict the outcome. Here a drop of silicon oil impacts a film of silicon oil with an even higher viscosity. The momentum of that impact creates a crater and a splash curtain that rises and expands from the initial point of impact. Because the film viscosity is higher than the drop’s, the evolution of the corona slows down. Eventually, surface tension and gravity start pulling the splash curtain back down as the crater collapses. Meanwhile at the top of the splash, capillary forces pull fluid into the rim, which becomes unstable and grows cusps that eventually eject a cloud of smaller droplets. (Image and research credit: H. Kittel et al., source)

The Kaye Effect
Allow a stream of shampoo to fall into a pile and you’ll catch a glimpse of the bizarre Kaye effect. A jet of shampoo will briefly rise up before becoming chaotic and falling. The key to this behavior is the shear-thinning of the shampoo. When the shampoo is just sitting on a surface, it’s quite viscous, but slide your hand across it, and the shampoo will become much less resistant to flowing.
When the jet of falling shampoo hits the pile, it creates a little dimple. Sometimes the incoming jet hits that dimple and slips along it, thanks to a sudden decrease in viscosity. That can send an outgoing jet of shampoo riding off the dimple like a ramp. As the dimple deepens, the outgoing streamer rises up until it hits the incoming jet and becomes unstable. The shampoo streamer collapses, only to be restarted when a new dimple forms. (Image and video credit: S. Mould; h/t to Guillaume D.)