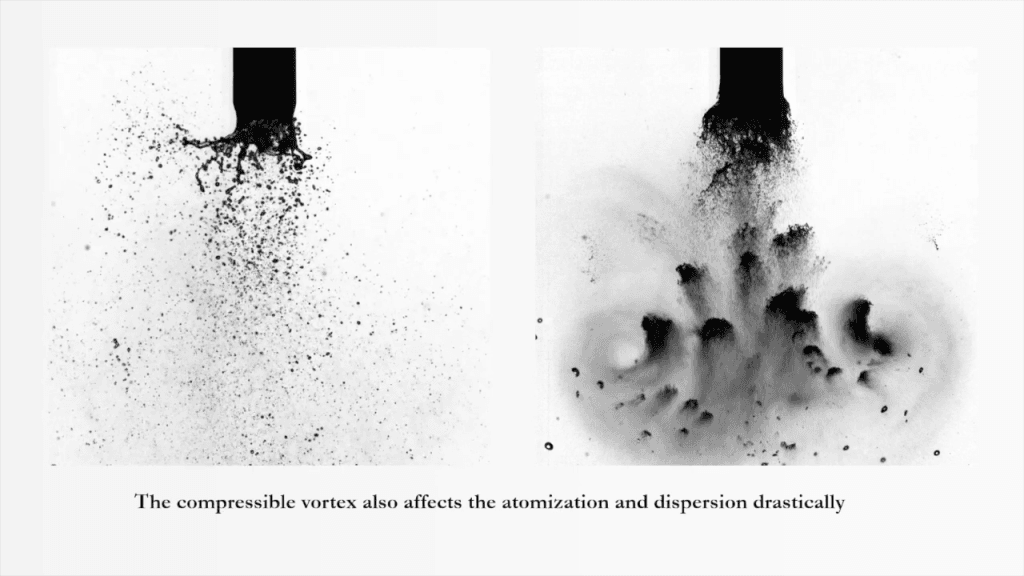
Many industrial processes break a fluid jet into droplets, like spray painting and ink-jet printing. Here, researchers examine an effervescent fluid jet made up of both liquid and gas. Like a fluid-only jet, this fizzy jet forms sheets, bags, ligaments, and droplets. As it breaks down, it creates a range of droplet sizes–both large and small. But when a shock wave passes, the jet and its droplets get atomized into even tinier droplets. (Video and image credit: S. Rao et al.)
Tag: fluid dynamics

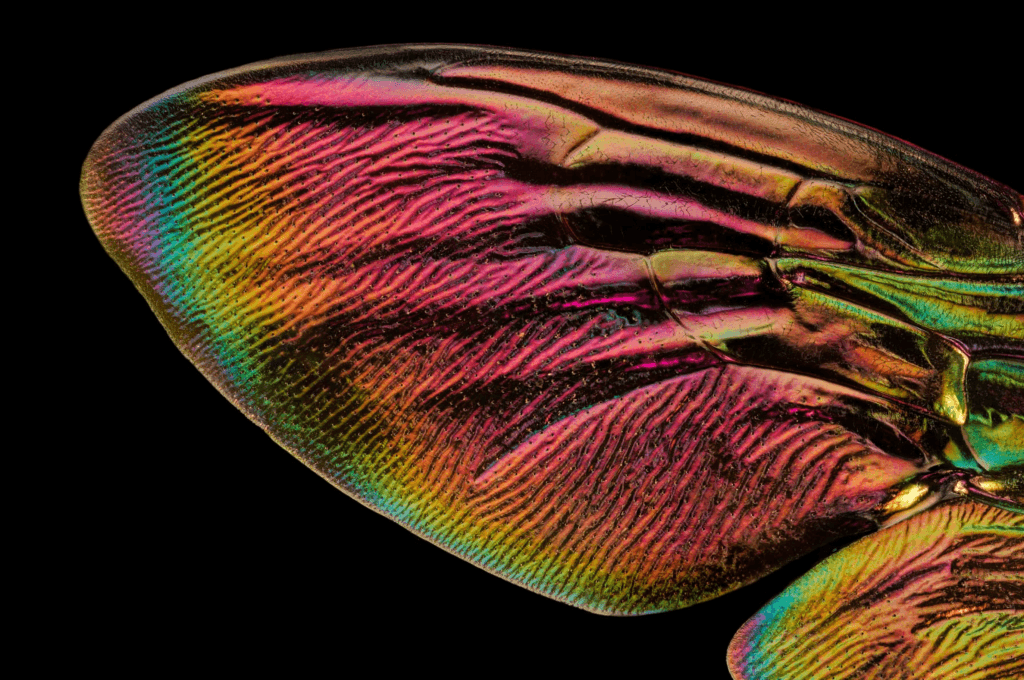
Insect Wings in Extreme Macro
Photographer Chris Perani is fascinated by the microstructures of insect wings, which he captures in “extreme macro” through focus stacking–letting us see wings in glorious micron-scale detail. In addition to giving insects their brilliant colors and irridescence, these structures serve another key role: they help insects stay dry. In a world where contact with water is unavoidable, insects have instead evolved to trap air in the gaps of their wings, letting water slide off instead of sticking. (Image credit: C. Perani; via Colossal)

Melting Can Propel Icebergs
Icebergs have long served as a metaphor for not knowing what’s going on beneath the surface. Studies like today’s are a reminder of why that is. Researchers found that asymmetric icebergs–shaped, in this case, like a right triangular prism–can self-propel as they melt. Their shape forces cold, dense meltwater to slide down the surface, generating a sinking plume that propels the ice as a whole. The team demonstrated this effect in both fresh- and saltwater. For icebergs wandering into warm waters, the effect is particularly strong and may reach levels about 10% of the magnitude of dominant propulsive forces like wind. (Image and research credit: M. Berhanu et al.; via APS)


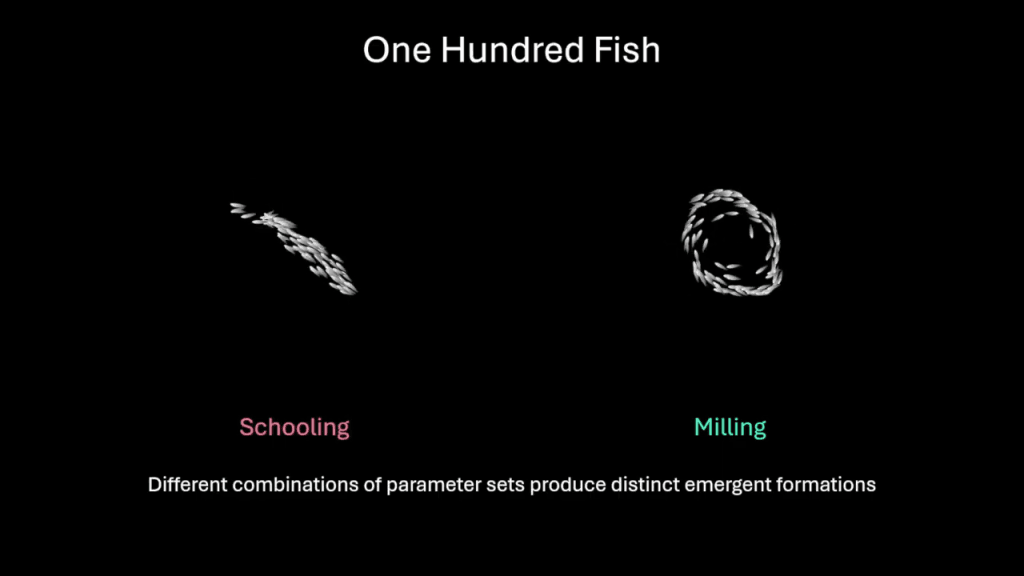
Schooling at Scale
Relatively simple visual and hydrodynamic signals are enough to make digital fish school in ways that resemble living ones. Here, researchers look at what happens when well-behaved schools of fish get too big. The researchers first demonstrate that their schools behave reasonably at one hundred members, either in a schooling configuration or a group milling around a central region.
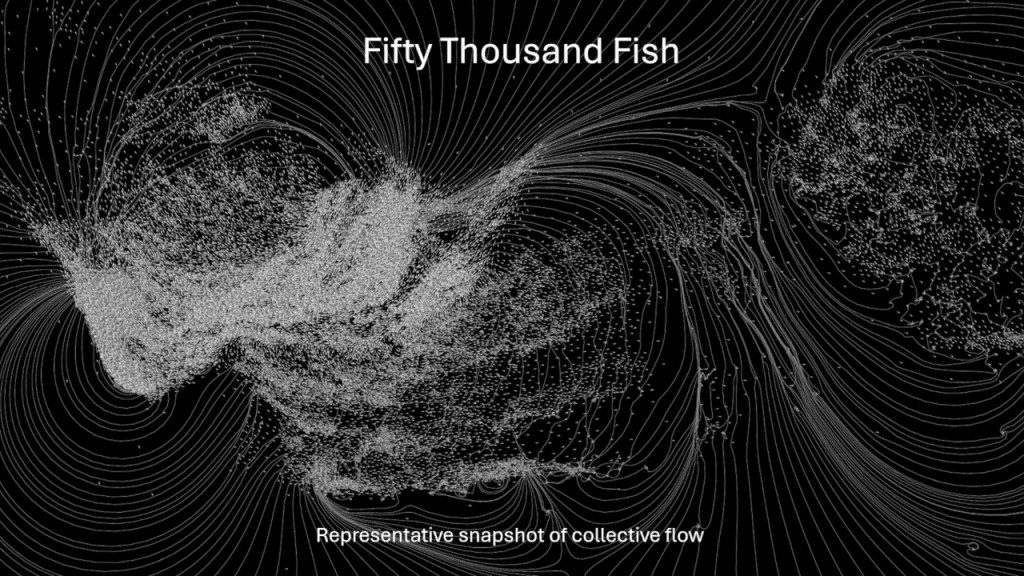
At one thousand fish, the schools are still reasonably coherent and sensible. But at fifty thousand fish, the picture is drastically different. Neither schooling nor milling groups are able to remain together. They fracture and scatter into smaller groupings. (Video and image credit: H. Hang et al.)

Scrubbing Bubbles
Cleaning produce helps fruits and vegetables last longer and reduces the chances for foodborne illness. But it can be a difficult feat with soft, delicate foods like tomatoes, berries, or greens. Current methods often combine ultrasonic cleaning and chemicals like chlorine. Instead, researchers are looking to boost the cleaning power of bubbles themselves by giving them an acoustic pick-me-up.

Stop-and-go. A bubble slides along an inclined surface in a pronounced stop-and-go motion when vibrated near its frequency for translational resonance. The team combined a bubble-filled bath with sound at low (sub-cavitation) frequencies. They found that driving sound waves at the right frequency could vibrate the bubbles in a way that made them slide in a stop-and-go motion along inclined surfaces. This swaying significantly boosted their cleaning power; getting surfaces 90% cleaner than non-resonating bubbles did. (Image credit: S. Hok/Cornell University; video and research credit: Y. Lin et al.; via Gizmodo)

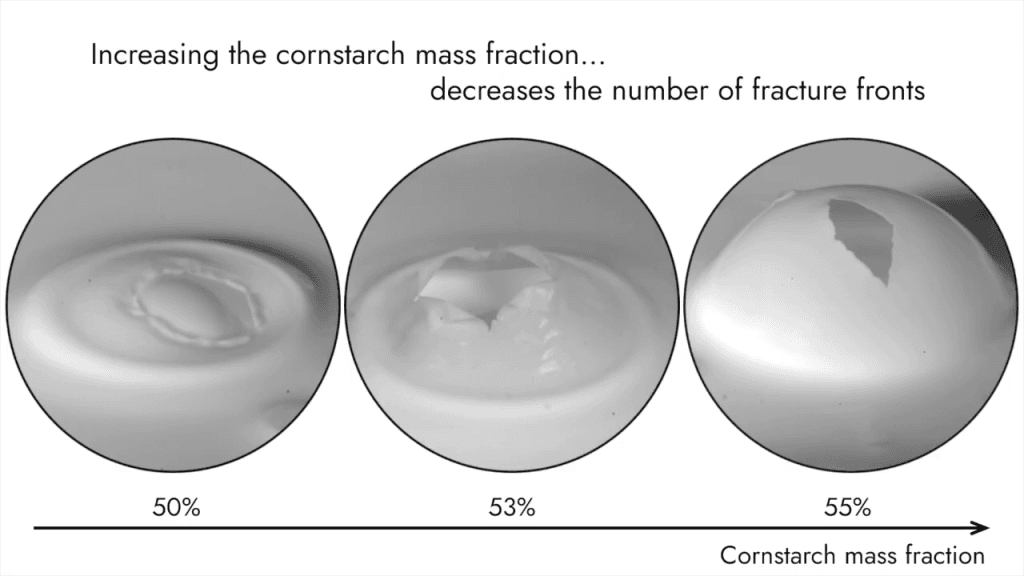
Bursting an Oobleck Bubble
When soap bubbles burst, the hole grows as an expanding circle. But not every fluid bursts this same way. Here, researchers let air rise through oobleck–a fluid made from cornstarch suspended in water–to form a bubble. In time, as with all bubbles, the oobleck bubble bursts. But–in keeping with oobleck’s solid-like properties–the film tears open and fractures. As it sinks back into the liquid, it wrinkles before it slowly relaxes back into fluid form. (Video and image credit: X. Zhang et al.)

Frog Kick
A toad swims across a pond in this award-winning image from photographer Paul Hobson. The shot was actually captured from below the water, with the camera kept dry in a glass housing. Although the frog appears to be mid-leap, the light-distorting ripples around its feet hint at the flow its kick generated. It’s reminiscent of the vortices left by water striders as they move. (Image credit: P. Hobson/BWPA; via Colossal)


Fluid Flows Break Up Microswimmer Clumps
The field of active matter looks at the collective motion of particles and organisms–how birds flock and fish school. In systems of “dry” squirmers–those that have no hydrodynamic interactions with one another–clumps of squirmers can form with empty spaces in between them. This is known as motility-induced phase separation, or MIPS. Researchers wondered whether microswimmers in a fluid–which do produce hydrodynamic forces that can affect one another–would also show MIPS.
In a new study, researchers show, instead, that hydrodynamic interactions between swimmers will prevent (or destroy) these clumps. Through a combination of theoretical work and simulation, the authors found that translational flows between swimmers swept the swimmers out of clumps as they formed. Rotational flows between swimmers made them able to change direction faster, which also kept stable clumps from forming. (Image and research credit: T. Zhou and J. Brady; via APS)

Hydrodynamic interactions destroy clumps of microswimmers. This simulation shows microswimmers that are initially in a clumped formation before hydrodynamic interactions are “turned on”. Once the swimmers can affect one another through the flows their motion creates, the clumps quickly break apart. 
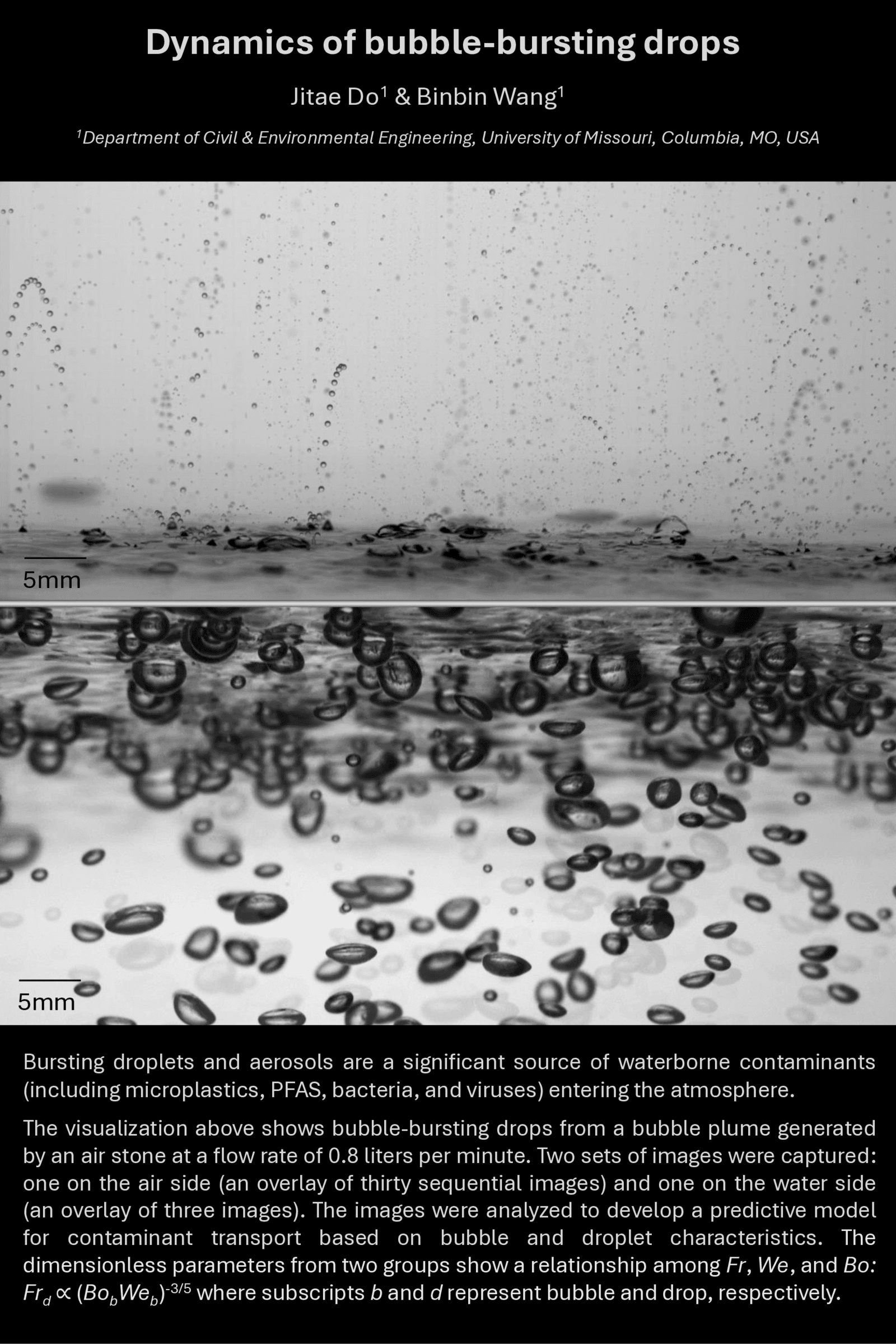
Bursting Bubbles
When air bubbles rise through a liquid, they scavenge dust, viruses, microplastics, and other impurities as they go. Once at the surface, these contaminant-covered bubbles thin and burst, generating many tiny droplets that arc through the air above. You’re likely familiar with the sight and sensation from a glass of champagne or soda.
Here, researchers have stacked two sets of sequential images to illustrate this complicated flowscape. Under the surface, a trio of photos are stacked to show bubbles rising and gathering at the surface. In the air, the researchers have stacked thirty sequential images, which together trace out the parabolic arcs of droplets sprayed by the bursting bubbles. (Image credit: J. Do and B. Wang)


Building Triboelectric Charge
In volcanic eruptions, collisions between ash particles can sometimes build up enough electric charge for lightning to arc through the plume. Scientists have long debated how this happens–it’s not obvious that insulating materials like oxides would build up electric charges through contact, especially when dealing with substances of the same material. It’s not like rubbing a balloon against your hair, where each material–and its tendency to hold a charge–differs.

A 500-micron silica sphere acoustically levitated above a silica plate in the experiment. To test how charges build on identical materials, a team of scientists used acoustic levitation to repeatedly bounce a silica bead against an identically treated silica plate, observing their charge build-up. Then they would take one of the pieces–either the sphere or the plate–and treat it to strip away the film of molecules that naturally adsorb onto the surface over time. Then they bounced the treated and untreated surfaces off one another again.
The result was–pardon the pun–striking. Whichever surface had been treated to remove adsorbates charged more negatively the second time around. Looking more closely at what they were removing, the team found their surfaces were mostly adsorbing carbon molecules. And if they iteratively removed the carbon from both the sphere and plate, they could no longer charge the two through collision. It seems that the key to charging two oxides off one another is actually the difference between the incidental amounts of carbon on their surfaces! (Image credit: volcano – M. Szeglat, experiment – G. Grosjean et al.; research credit: G. Grosjean et al.; via Gizmodo)