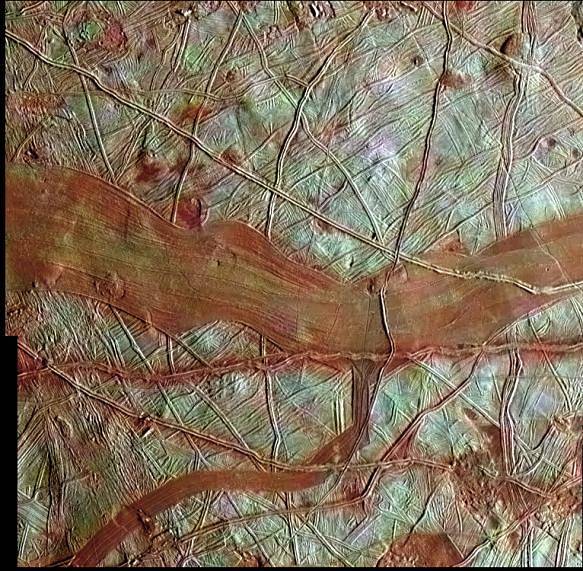
Jupiter’s icy moons, Europa and Ganymede, are home to subsurface oceans. These moons also experience strong tidal forces from their parent planet and sibling moons that squeeze and deform them over time. A new study focuses on the bands, seen in red in the top image of Europa, that form as a result of these deformations. By simulating (bottom image) both the convective currents within the Europan ocean and the deformation of the ice over time, scientists are able to study how these geological surface features may have formed. Over the course of about a million years, material from the interior ocean works its way up into the center of a band. Because this process takes so long, the researchers point out that any attempt to collect material from the bands will yield “fossil” ocean material – essentially a glimpse of Europa’s ocean as it existed a million years ago rather than how it exists today! (Image credit: NASA; image and research credit: S. Howell and R. Pappalardo, source; submitted by Kam-Yung Soh)
Tag: convection

Tea Physics
Tea is a popular beverage around the world, and nearly everyone has their own method for making the perfect cup. Perhaps unsurprisingly, scientists have studied tea physics as well. One such study used both experiments and numerical simulations to study tea infusion from teabags. The authors looked at round, two-dimensional teabags in two configurations – one in which the bag was left still during infusion and one in which the bag was dunked up and down in the water.
In the static case, as the hot water leeches solutes out of the tea leaves, it forms a buoyant convection current. In this case, the convection is driven by solute concentration, not temperature. The convection creates a re-circulation in the cup that helps slowly distribute the tea solutes.
The dunking method, unsurprisingly, distributes tea solutes much faster. In addition to stirring the cup’s contents, dunking helps drive flow through the tea leaves, releasing solutes faster. Although the authors study the two methods in detail, they decline to pass judgement on what method is “the best”. (Photo credit: T. Foster, source; research credit: G. Lian and C. Astill; submitted by Marc A.)

Martian Mantle Convection
Over geological timescales – on the order of millions of years – even hard substances like rock can flow like a fluid. Heat from the Earth’s core drives convection inside our mantle, and that fluid motion ultimately drives the plate tectonics we experience here at the surface. But most other planetary bodies, including those with mantle convection similar to ours, don’t have a surface that shifts like our tectonic plates. Mars and Venus, for example, have solid, unmoving surfaces. The images above provide a peek at what goes on beneath. The upper image shows a simulation of mantle convection inside Mars over millions of years. The lower image is a timelapse of dye convecting through a layer of glucose syrup being heated from below. Notice how both examples show evidence of convective cells and plumes that help circulate warm fluid up and colder fluid downward. (Image credit: Mars simulation – C. Hüttig et al, source; N. Tosi et al., source; submitted by Nicola T.)

Castle-like Clouds
An astronaut captured this towering cloud over Andros Island from orbit aboard the ISS. This is a cumulus castellanus cloud, named for the castle-like crenelations at its top. Castellanus clouds form in areas with strong vertical updrafts, often due to cloud-level atmospheric instabilities rather than heating at the Earth’s surface. These clouds frequently proceed rain or even thunderstorms. What distinguishes castellanus from other types of cumulonimbus clouds is their shape: castellanus clouds have protrusions that are taller than they are wide – like the castles for which they are named. (Image credit: NASA / Expedition 48; via NASA Earth Observatory)

Layered Latte Physics
Latte lovers may be familiar with the layered latte, a beverage with distinctive horizontal layers mixing espresso and milk, but you may not have taken the time to wonder how these layers form. Like many layering phenomena in our oceans, the layered latte is the result of double-diffusive convection. This means that there are two variables that both affect density in the fluid mixture and that they act at different rates.
In the latte, those factors are 1) the different densities of the milk and espresso and 2) density changes caused as the latte cools to room temperature. A layered latte forms when the lighter espresso is poured into denser milk. If it’s poured quickly enough, the momentum of the pour forces some of the espresso down into the milk, despite the buoyant force that tries to keep the espresso on top. So that initial pour sets up a density gradient that runs from pure espresso at the top to pure milk on the bottom, with varying mixtures of the two in between.
The distinct layers won’t form until the latte begins cooling off. Along the walls of the container, heat is lost more quickly, causing fluid to cool and start sinking. But a specific bit of fluid can only sink until the fluid surrounding it is the same density. That can carry a cooler bit of latte to the bottom of a layer, but not into the denser layer below. At this point, our bit of latte moves inward, starts to warm up, and circulates up through the center of its layer. As when it sank, the fluid can only move up until it encounters a layer with equal or lesser density, at which point it must move horizontally instead. This thermal convection, combined with the density gradient formed by the initial pour, sets up the distinctive layers of the latte. The layers are quite stable – neither gentle stirring nor taking a sip will disrupt them for long – provided the drink remains warmer than the surrounding air. (Image credits: kopeattugu/Instagram, N. Xue et al.; research credit: N. Xue et al.; via NYTimes; submitted by Kam-Yung Soh)

Resisting Coalescence
When a droplet falls on a pool, we expect it to coalesce. There are exceptions, like bouncing droplets, but in general a droplet only sticks around for a split second before being engulfed. And yet, from morning coffee (top image) to walks in the woods, we frequently see millimeter-sized droplets sticking around for far longer than it seems like they should. New research offers a clue as to why: it’s thanks to a temperature difference.
When there’s an appreciable temperature difference between the drop and the pool, it causes rotating convective vortices (bottom image) in both the drop and the pool. When the temperature difference is large, the vortices are strong enough that their motion recirculates air inside the tiny gap between the drop and the pool. This supports the weight of the drop and keeps the two liquids separate. But the convection also redistributes heat, and eventually the drop and pool become similar enough in temperature that the circulation dies out, the air gap drains, and the two coalesce. (Image and research credit: M. Geri et al.; via MIT News; submitted by Antony B.)

Flow Inside Convection Cells
Looking at convective cells, it’s easy to think that they are still and unmoving. But when you add particles, their inner flow becomes obvious. Warm, light fluid moves up through the center of each cell, skims along the surface, and then sinks at the edges of the cell after losing its heat at the cooling surface. Below, the fluid moves back toward the cell center, getting warmer as it’s heated by the lower surface. Once it reaches the middle of the cell, it’s light enough to rise up and start the process again. Convective cells like these are typical in cooking – watch for them forming in your miso soup or hot chocolate – but they can also be found on the sun and even in situations without heating! (Image credit: G. Kelemen, source)

Convection
Blue paint in alcohol forms an array of polygonal convection cells. We’re accustomed to associating convection with temperature differences; patterns like the one above are seen in hot cooking oil, cocoa, and even on Pluto. In all of those cases, temperature differences are a defining feature, but they are not the fundamental driver of the fluid behavior. The most important factors – both in those cases and the present one – are density and surface tension variations. Changing temperature affects both of these factors, which is why its so often seen in Benard-Marangoni convection.
For the paint-in-alcohol, density and surface tension differences are inherent to the two fluids. Because alcohol is volatile and evaporates quickly, its concentration is constantly changing, which in turn changes the local surface tension. Areas of higher surface tension pull on those of lower surface tension; this draws fluid from the center of each cell toward the perimeter. At the same time, alcohol evaporating at the surface changes the density of the fluid. As it loses alcohol and becomes denser, it sinks at the edges of the cell. Below the surface, it will absorb more alcohol, become lighter, and eventually rise at the cell center, continuing the convective process. (Image credit: Beauty of Science, source)

Inside Earth’s Core
Without our magnetic field, life as we know it could not exist on Earth. Instead, our atmosphere would be stripped away and the surface would be bombarded by charged particles in the solar wind. Relatively little is known about the dynamo process that governs our magnetic field, though it’s thought to be the result of liquid iron moving in the Earth’s outer core. The video above shows a slice of a recent 3D simulation of this liquid iron segment of our core. The colors show variations in the temperature, revealing vigorous convection in the core. This motion, combined with the spinning of the Earth, is the likely source of our magnetic field. Researchers hope that simulations like these can help us understand features we observe in our magnetic field – like local variations in field strength and the pole reversals in our geological record. (Video credit: N. Schaeffer et al.; CNRS via Gizmodo)

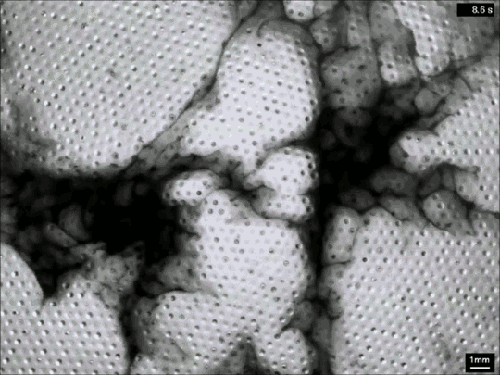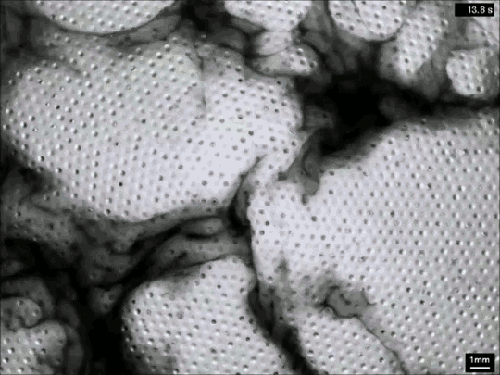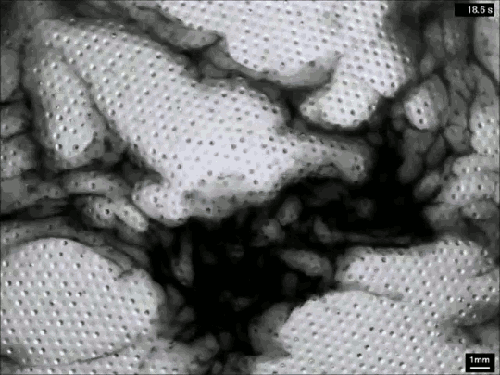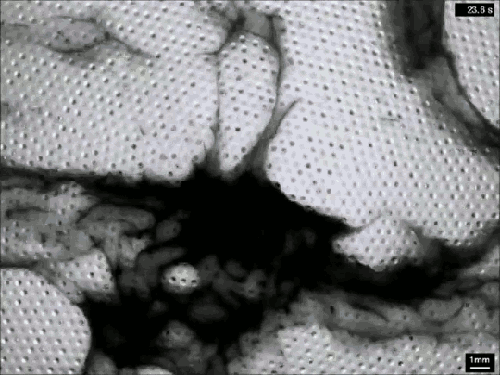
Creating Clouds
What you see here is the formation of clouds and rain – but it’s not quite what you’re used to seeing outside. This is an experiment using a mixture of sulfur hexafluoride and helium to create clouds in a laboratory. Everything is contained in a cell between two transparent plates. Liquid sulfur hexafluoride takes up about half of the cell, and when the lower plate is heated, that liquid begins evaporating and rising in the bright regions. When it reaches the cooled top plate, the liquid condenses into droplets inside the dimples on the plate, eventually growing large enough to fall back as rain. The dark wisps you see are areas where cold sulfur hexafluoride is sinking, much like in the water clouds we are used to. Setups like this one allow scientists to study the effects of turbulence on cloud physics and the formation of droplets. (Image credit: E. Bodenschatz et al., source)
Boston-area folks! I’ll be taking part in the Improbable Research show Saturday evening at 8 pm at the Sheraton Boston. Come hear about the Boston Molasses Flood and other bizarre research!