Beautiful colors, subtle flows, and sudden fractals animate Thomas Blanchard’s “Sensations,” which, like his other short films, is entirely CGI-free. It’s a lovely exploration of droplets, liquid lenses, Marangoni effects, and fingering instabilities. (Video and image credit: T. Blanchard)
Search results for: “droplet”

Forming Vesicles on Titan
Scientists are still debating exactly what shifts nature from chemical and physical reactions to living cells. But vesicles — small membrane-bound pockets of fluid carrying critical molecules — are a commonly cited ingredient. Vesicles help cluster important organic molecules together, increasing their chances of combining in the ways needed for life. Now scientists are suggesting that Titan, Saturn’s moon, could form vesicles of its own.
On Earth, molecules known as amphiphiles feature a hydrophilic (water-loving) end and a hydrophobic (water-fearing) one. When dispersed in water, amphiphiles crowd at the surface, placing their hydrophilic end in the water and their hydrophobic end outward toward the air. On Titan, the Cassini mission revealed organic nitrile molecules that behave similarly with methane rather than water.
Their two-sided structure means that these molecules — like Earth’s amphiphiles — will gather at the surface of Titan’s liquids. When methane rain falls on the Titan’s seas, the impact creates aerosol droplets that slowly settle back to the liquid surface. When that happens, the droplet’s molecular monolayer and the lake’s monolayer meet, enclosing the droplet’s contents in a double-layer of molecules that prevent contact between the droplet and the lake.
Within that newly-formed vesicle, all kinds of molecules can bump shoulders, creating new opportunities for complex chemistry. (Image credit: Titan – ESA/NASA/JPL/University of Arizona, illustration – C. Mayer and C. Nixon; research credit: C. Mayer and C. Nixon; via Gizmodo)

Crown Splash
When a falling drop hits a thin layer of water, the impact sends up a thin, crown-shaped splash. This research poster shows a numerical simulation of such a splash in the throes of various instabilities. The crown’s thick edges are undergoing a Rayleigh-Plateau instability, breaking into droplets much the way a dripping faucet does. On the far side, the crown has rapidly expanding holes that pull back and collide. The still-intact liquid sheet at the base of the crown shows some waviness, as well, hinting at a growing instability there. (Image credit: L. Kahouadji et al.)

Evaporating Off Butterfly Scales
This award-winning macro video shows scattered water droplets evaporating off a butterfly‘s wing. At first glance, it’s hard to see any motion outside of the camera’s sweep, but if you focus on one drop at a time, you’ll see them shrinking. For most of their lifetime, these tiny drops are nearly spherical; that’s due to the hydrophobic, water-shedding nature of the wing. But as the drops get smaller and less spherical, you may notice how the drop distorts the scales it adheres to. Wherever the drop touches, the wing scales are pulled up, and, when the drop is gone, the scales settle back down. This is a subtle but neat demonstration of the water’s adhesive power. (Video and image credit: J. McClellan; via Nikon Small World in Motion)

Water droplets evaporate from the wing of a peacock butterfly. 
Penguin Poo Seeds Antarctic Clouds
Forming clouds requires more than just water vapor; every droplet in a cloud forms around a tiny aerosol particle that serves as a seed that vapor can condense onto. Without these aerosols, there are no clouds. In most regions of the world, aerosols are plentiful — produced by vegetation, dust, sea salt, and other sources. But in the Antarctic, aerosol sources are few. But a new study shows that penguins help create aerosols with their feces.
Penguin feces is ammonia-rich, and that ammonia, when combined with sulfur compounds from marine phytoplankton, triggers chemistry that releases new aerosol particles. The researchers measured ammonia carried on the wind from nearby penguin colonies and found that the birds are a large ammonia source, producing 100 to 1000 times the region’s baseline ammonia levels. In combination with another ingredient in penguin guano, the researchers found the penguins boosted aerosol production 10,000-fold. That means penguins can actually influence their environment, helping to create clouds that keep Antarctica cooler. (Image credit: H. Neufeld; research credit: M. Boyer et al.; via Eos)

“Soap Bubble Bonanza
This video offers an artistic look at a soap bubble bursting. The process is captured with high-speed video combined with schlieren photography, a technique that makes visible subtle density variations in the air. The bubbles all pop spontaneously, once enough of their cap drains or evaporates away for a hole to form. That hole retracts quickly; the acceleration of the liquid around the bubble’s spherical shape makes the retracting film break into droplets, seen as falling streaks near the bottom of the bubble. The retraction also affects air inside the bubble, making the air that touched the film curl up on itself, creating turbulence. Then, as the film completes its retraction, it pushes a plume of the once-interior air upward, as if the interior of the bubble is turning itself inside out. (Video and image credit: D. van Gils)

The Hidden Beauty in the Mundane
Physicist Sidney Nagel has spent his career on topics that are somewhat unexpected: how coffee stains form, how droplets splash — or don’t, and how fluid flows into viscous fingers. Often this means looking at the mechanics of everyday occurrences that we otherwise take for granted. Instead, Nagel probes carefully at things like a coffee stain, asking why it’s darker at the edges and what he could do to keep that from happening — all to ultimately uncover the forces and mechanisms at play. Quanta has a great little interview with him on this and other topics. Check it out here. (Image credit: S. Nagel and K. Norman; via Quanta)



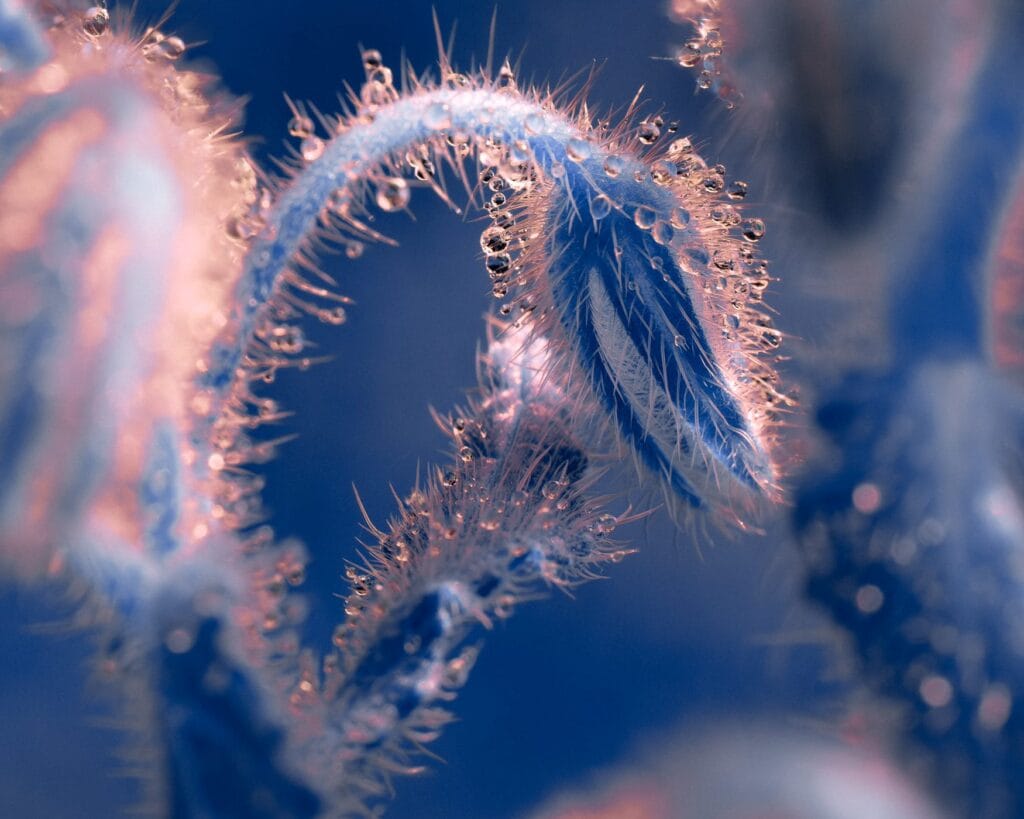
“Spines”
Water droplets cling to spine-covered plant life in this series from photographer Tom Leighton. The hairs are hydrophobic — notice how spherical the drops appear. Many plants make parts of their leaves and stems hydrophobic in order to redirect water toward their roots, where it can be taken in. Others use hair-like awns to collect and draw in dew that supplements their water capture. (Image credit: T. Leighton; via Colossal)

Charged Drops Don’t Splash
When a droplet falls on a surface, it spreads itself horizontally into a thin lamella. Sometimes — depending on factors like viscosity, impact speed, and air pressure — that drop splashes, breaking up along its edge into myriad smaller droplets. But a new study finds that a small electrical charge is enough to suppress a drop’s splash, as seen below.

The drop’s electrical charge builds up along the drop’s surface, providing an attraction that acts somewhat like surface tension. As a result, charged drops don’t lift off the surface as much and they spread less overall; both factors inhibit splashing.* The effect could increase our control of droplets in ink jet printing, allowing for higher resolution printing. (Image and research credit: F. Yu et al.; via APS News)
*Note that this only works for non-conductive surfaces. If the surface is electrically conductive, the charge simply dissipates, allowing the splash to occur as normal.

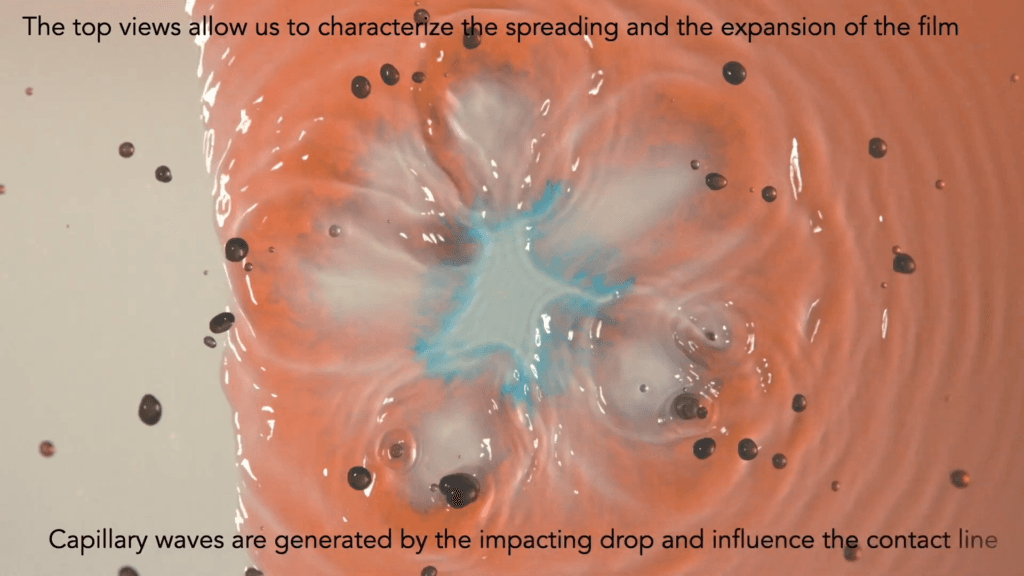
Drops on the Edge
Drops impacting a dry hydrophilic surface flatten into a film. Drops that impact a wet film throw up a crown-shaped splash. But what happens when a drop hits the edge of a wet surface? That’s the situation explored in this video, where blue-dyed drops interact with a red-dyed film. From every angle, the impact is complex — sending up partial crown splashes, generating capillary waves that shift the contact line, and chaotically mixing the drop and film’s liquids. (Video and image credit: A. Sauret et al.)