As red ants scout their way to food, the terrain can sometimes get in the way. Here a leading scout has made their body into a bridge that their fellows can use to cross the watery gap. Take a close look at the water’s surface and you’ll see that the meniscus curves up to meet the rocks. That’s a clue that this image is really very small! For water on Earth, that curvature only occurs at lengths below a couple of millimeters, where surface tension has the power to overcome gravity’s efforts to flatten the surface. The ants’ bridge is only possible because the red ant is small enough and light enough for surface tension to support it. Learn more about the amazing interactions of ants and water in some of my previous posts. (Image credit: Chin Leong Teo; via Colossal)
Search results for: “art”

Box Closing Physics
My fellow board game aficionados (and anyone else who regularly opens and closes lidded boxes) have probably noticed the way a lid drops slowly onto its box once aligned. The weight of the lid pressurizes air inside the box, driving a flow through the narrow gap between the walls of the box and the lid. Researchers found that the time it takes for a box to slide closed is closely related to the size and shape of the gap between the walls. Despite gaps of less than 1 millimeter, air moving out of the box typically flows at about 1 meter per second!
With their mathematical model of the flow from a closing box, the group was also able to determine the optimal shape for a fast-closing box, something that may be of interest to manufacturers as well as fans of board games. (Image credit: N. Sharp; research credit: J. de Ruiter et al.; via APS Physics)

“ColorLover”
“ColorLover,” a short film by artist Rus Khasanov, is a delightful liquid rainbow. The video’s ingredients seem to be ink, paint, oil, and a bit of superhydrophobic coating primed to reveal a heart. I love that latter touch; it’s a cool way to use regular materials in a way that some might assume involved digital effects! (Video credit: R. Khasanov)

“Reverent”
Today, enjoy this moody black-and-white short film of storm timelapses. Photographer Mike Olbinski is a master of this subject. I never tire of watching his towering convective supercell thunderstorms or his picturesque microbursts. The lightning-lit clouds in the latter half of the film are particularly spectacular (assuming you do not have sensitivities to flashing lights). And there are a few haboobs and a tornado in there for good measure, too. (Image and video credit: M. Olbinski)

Within the Bubble’s Pop
To our eyes, a soap bubble appears to pop instantly, but when observed in high-speed video, the process is far more complex. In this video, the Slow Mo Guys pop human-sized bubbles, giving us an opportunity to appreciate the rupture process at speeds up to 50,000 frames per second.
Once the rupture starts, the hole spreads very symmetrically. But as the hole grows, the remaining soap film starts distorting. As Gav and Dan observe, the far side of the bubble actually wrinkles up before the rupture front arrives and tears the remaining fluid into droplets! (Image and video credit: The Slow Mo Guys)

Mixing the Immiscible
Immiscible liquids — like oil and water — do not combine easily. Typically, with enough effort, you can create an emulsion — a mixture formed from droplets of one liquid suspended in the other — like the one above. But a team of researchers have taken mixing immiscible liquids to a new level using their Vortex Fluid Device (VFD).
Longtime readers may remember the group from their Ig-Nobel-winning demonstration of unboiling an egg, but this time the team is used the VFD to mix and de-mix immiscible liquids. As shown in the video below, the VFD is essentially a fast-spinning tube tilted at a 45-degree angle. As it spins, the liquids inside are forced into thin films with very high shear rates — high enough that immiscible liquids like water and toluene are forced together without forming an emulsion. Essentially, the mechanical forces mixing the liquids are strong enough to overcome the chemistry that typically keeps them apart.
Impressively, the device manages this without using harsh surfactants or catalysts that other methods rely on. As a result, the technique offers a greener method for mixing chemicals for pharmaceuticals, cosmetics, food processing, and more. (Image credit: pisauikan; research credit: M. Jellicoe et al.; video credit: Flinders University; submitted by Marc A.)

Sunrise Cloudscape
With the low sun angle of dawn, the details of this cloudscape stand out. Captured by an external camera on the International Space Station, this image shows cloud formations over the northwest Atlantic. In the foreground, towering cumuli mark rising plumes of warm, moist air evaporating from the ocean. Beyond those clouds, a flat anvil cloud spreads horizontally after a temperature inversion prevented it from rising any further. (Image credit: NASA; via NASA Earth Observatory)

Coronal Heating
Compared to its interior, the surface of our sun is a cool 6,000 degrees Celsius. But beyond the surface, the sun’s corona heats up dramatically through interactions between plasma and strong magnetic fields. The exact mechanisms of this interaction have been mostly theoretical thus far, but a recent laboratory experiment has validated a part of that theory.
One explanation for coronal heating posits that the strong magnetic fields can accelerate magnetohydrodynamic waves called Alfvén waves to speeds faster than sound, and that at this crossover point, changes occur in the waves’ behavior. Using liquid rubidium, researchers were able to observe this crossover under laboratory conditions, confirming that the Alfvén waves change at the speed of sound in exactly the manner predicted by theory. (Image credit: NASA SDO; research credit: F. Stefani et al.; via Physics World)
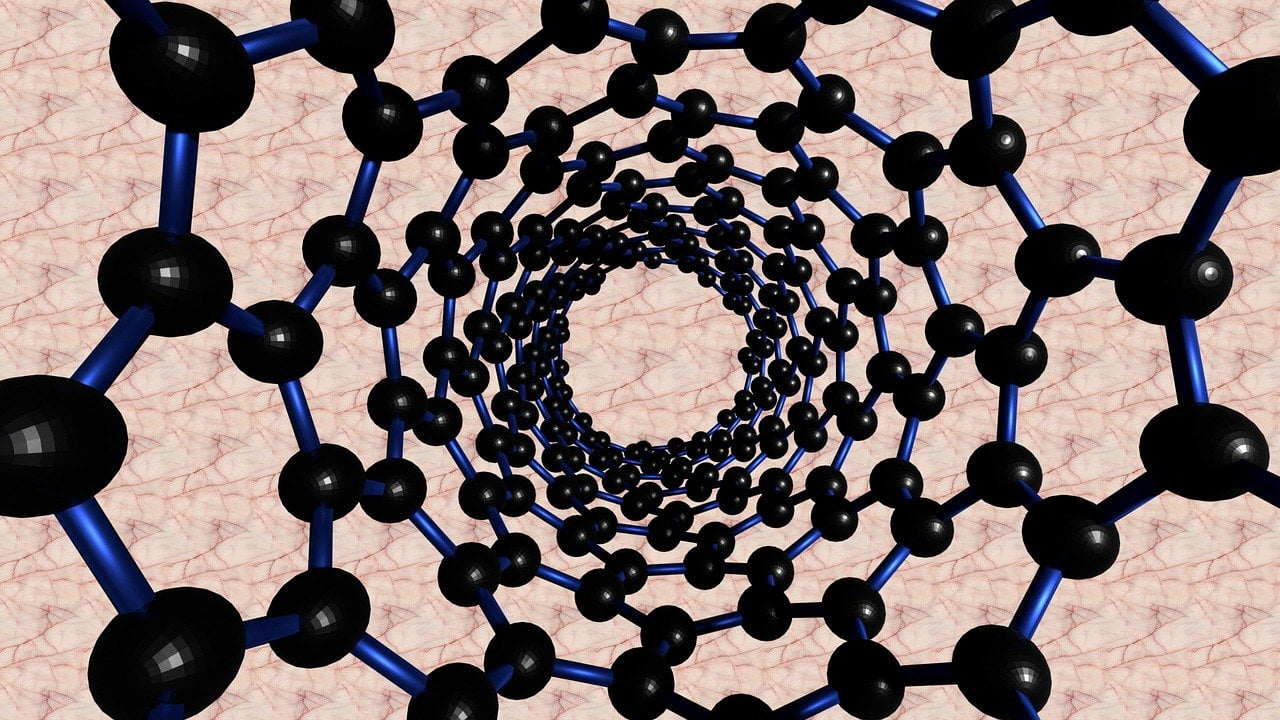
Electronic Friction
Years ago, physicists discovered that water flows with surprisingly little friction through narrow carbon nanotubes. At our scale, flow behavior is typically the opposite: there’s greater friction (and, thus, slower flow) in a narrower pipe. To unravel the mystery, researchers had to delve into quantum mechanics and model the interactions between the atoms of a water molecule and the electrons of the carbon atom. Essentially, this meant building a quantum picture of the liquid-solid interface inside the nanotube.
The team found that the electrons of the nanotube exert a drag-like force on the water molecules, creating friction that slows the flow. Since narrow nanotubes have fewer electrons than larger tubes, there is less friction on the flow and the water flows faster! (Image credit: cintersimone; research credit: N. Kavokine et al.; via SciAm; submitted by Kam-Yung Soh)

Watery Salt Flats
Unusually high rainfall in Bolivia’s Salar de Uyuni turned the world’s largest salt flat into a shallow salt lake. These natural-color satellite images show the area in late January 2022. If you zoom in on the full resolution image, there are incredible detailed swirls in the water. It’s like peering at an abstract or Impressionist painting. The many colors are attributable to several sources, including volcanic sediments, runoff, and a variety of microbes and algae thriving in the mineral-filled waters. (Image credit: L. Dauphin; via NASA Earth Observatory)













