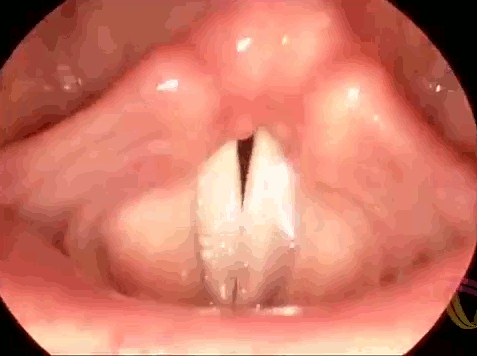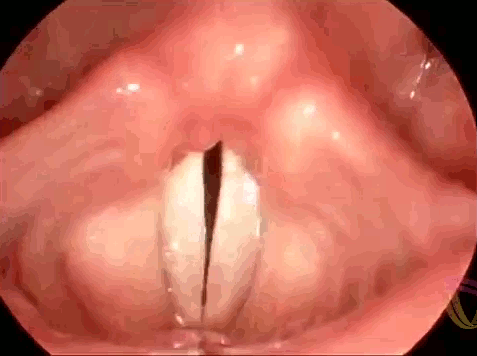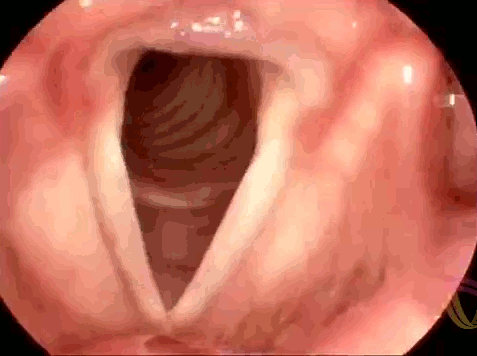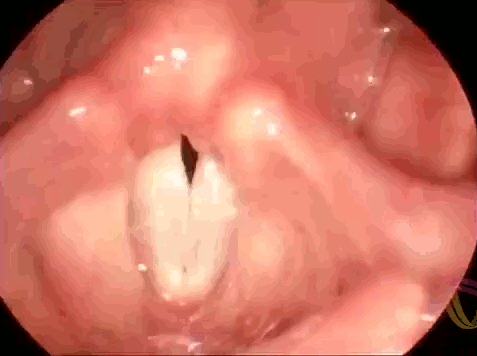
These are the vocal folds of a woman singing. Human speech (and song) results from interactions between elastic muscles and aerodynamics. As we exhale, the vocal folds are initially pushed apart, then the flow of air moving past creates low pressure (via the Bernoulli effect) that helps pull the folds together. As the folds close, high pressure again forms to force them open. This sets a cycle of oscillation or vibration that produces sound. To change the pitch of the sounds we create, we can lengthen or shorten the vocal folds or change their tension. In this respect, they behave somewhat similarly to the strings of a musical instrument. If you’d like to admire more vocal folds in action, check out this endoscopic video for four singers performing together. (Image credit: LinguaHealth, source)
Search results for: “art”

Boulder Sorting on Asteroid Itokawa
Itokawa is a small asteroid visited by the Japanese Hayabusa probe in 2005. Photographs of the asteroid revealed a surface covered in large boulders at high elevations and small pebbles in the valleys. The Brazil nut effect is often invoked to explain size separation in particle mixtures, but Itokawa is so small that any shaking sufficient to sort particles would likely exceed the asteroid’s meager escape velocity. Instead, researchers have suggested an alternative size sorting mechanism: ballistic sorting.
The idea of ballistic sorting is that pebbles that strike boulders will impact and bounce a long way, whereas pebbles that strike other pebbles are likely to rebound only a short way. In both experiments and simulations, the researchers found that this was the case and that mixtures of large and small particles tended to separate just as on the asteroid. The effect is possible on Earth as well, but Itokawa’s small gravitational acceleration makes for more effective size sorting. (Image credit: JAXA; research credit: T. Shinbrot et al.)

Using Jets to Find Food
Archer fish are well-known for their ability to hit aerial targets with perfectly aimed jets of water, as we’ve discussed previously. But a new study shows they use a similar technique to form underwater jets that help them uncover food. The researchers found that the fish altered the timing of their jet formation based on the type of substrate – fine sand, course sand, or mud – that the food pellet was hidden in. A great next step in this research would be using a technique like particle image velociometry (PIV) to measure the flow field directly and see to what extent the fish’s actions are altering the jet they produce. (Image and research credit: J. Dewenter et al.; GIF source: freshphotons)

Titan’s Bubbly Islands
Titan, Saturn’s largest moon, is a fascinating world with remarkable similarities to our own. It is the only other world we know of with stable bodies of liquid at its surface. Unlike Earth, frigid Titan’s lakes and seas are filled with methane and ethane. Radar data from the Cassini mission has shown oddly changing shorelines on Titan, above, with islands that seem to magically appear and disappear over time.
Researchers at NASA’s Jet Propulsion Laboratory now think these islands may, in fact, be bubbles. As Titan’s lakes cool, they’re better able to absorb nitrogen gas, but when temperatures warm up, that gas comes out of solution and floats to the surface, much like the bubbles of carbon dioxide in a soda. If this hypothesis holds up, there are some interesting implications for Titan’s atmosphere. Here on Earth, bubbles popping in the ocean are a major source of aerosol particles. It’s possible migrating rafts of bubbles could behave similarly on Titan. (Photo credit: NASA/JPL-Caltech/ASI/Cornell; submitted by jpshoer)
I’m excited to announce I will be visiting JPL later this month (March 30th), where I have the honor of giving a Women’s History Month talk. If there are any JPLers who are FYFD fans, I hope to see you there. Be sure to RSVP to the ACW luncheon by the March 24th deadline.
Reader Question: Image Credits
kermitsstickylittlefingers asks:
Dear Nicole, thanks a lot for your amazing blog which I have been following for years. My name is Julian and I am a PhD candidate working on tree frog attachment at Wageningen University in the Netherlands. Inspired by FYFD, I decided to start a science blog on bioadhesion and biomimetics. With respect to that, I would like to ask a question: How do you handle sources and referring to image credits and how do you always know to whom to refer? Thanks for the answer, Julian
Hi Julian,
That is an awesome topic you’re studying and sharing. I hope you keep up the great work.
I do my best to give appropriate credit when sourcing materials, but sometimes it can be quite hard to track down an original source. I’ve had to pass up great visuals on occasion because I simply could not determine whose work they originally were.
Fortunately, there is an invaluable tool out there for tracking down sources: Google Image Search. That service lets you upload a photo and it will show you other versions of that picture on other pages, which often helps in tracking down the proper original source. If you use the Chrome browser, you can simply right-click on a photo and select “Search Google for image” to automatically do this search.
Kudos to you for trying to do right by creators and credit them for their work. Good luck in your blogging and your research! (I <3 tree frogs.)

Sloshing in Space
Last month, French astronaut Thomas Pesquet posted a video of some experiments he did on the International Space Station exploring the movement of fluids in microgravity. He filmed the experiments as part of the SPHERES Slosh project. Sloshing is the technical term for how liquids respond to the motion of their container, and it’s a tough problem whether you’re carrying a full coffee mug on Earth or dealing with a partially-emptied fuel canister in orbit.
Here on Earth, gravitational forces dominate how fluids respond, but in microgravity, surface tension is a more powerful player. Pesquet’s demonstrations help scientists here on Earth better understand and model how liquids respond to movement in space. One major application for this is in spacecraft fuel tanks, which engineers must be able to design so that they empty themselves consistently with or without the added complications of spinning, maneuvering, or impulsive kicks of acceleration. (Video and image credit: ESA; submitted by gdurey)


Vertical Axis Wind Turbines
Most people are familiar with the propeller-like shape of conventional wind turbines. These turbines can be more than 100m tall and can generate several megawatts apiece, but placing them in arrays requires a lot of open space because flow downstream of one turbine will interfere with the efficiency of the next. Vertical axis wind turbines (VAWT), like those shown in the photo above, are smaller and produce a fraction of the power of their larger horizontal-axis counterparts, but VAWTs can be placed much closer together. In fact, putting them in closely spaced arrays can actually increase their output through flow synergies. Researchers hope that eventually VAWT arrays will be able to produce significantly more power per land area than conventional wind turbines. (Image credit: Texas A&M Corpus Christi)

Breaking the Wave Speed Limit
Whirligig beetles are small surface swimming insects. As they race across the water surface, they create both visible and unnoticeable waves on the water. These waves are the result of both surface tension and gravity. Typically, it’s the wavelength of the gravity waves that limit a swimmer or boat’s speed. When the wavelength of the gravity waves a swimmer creates meets the size of the swimmer, the waves generated ahead of the swimmer start to reinforce the waves forming at the back of the swimmer. This traps the swimmer (or boat) in a trough between its bow and stern waves and limits the max speed of the swimmer since overcoming this critical hull speed requires excessive amounts of power.
The tiny whirligig beetle overcomes this natural speed limit cleverly. It is smaller than the shortest possible gravity wave in water. Thus, it can never be trapped between its bow and stern waves! This allows the tiny swimmer to zip across the water’s surface at speeds above 0.5 m/s. That’s over 30 beetle body lengths per second! (Image credit: H. L. Drake, source; research credit: V. Tucker; submitted by Marc A.)

How Jet Engines Work
Jet engines are a major part of aviation today, and this great video from the new LIB LAB project breaks down how jet engines operate. It focuses especially on the subject of combustion, in which fuel-air mixtures are burned to generate power and thrust. By breaking fuels down into simpler compounds, jet engines are able to accelerate exhaust gases, which creates thrust. They even provide instructions for an effervescence-driven bubble rocket so that kids can (safely!) experiment with propulsion at home. (Video credit: LIB LAB/Corvallis-Benton County Public Library)

Supporting Bubbles
Surface tension holds small droplets in a partial sphere known as a spherical cap. But when droplets become larger, they flatten out into puddles due to the influence of gravity. In contrast, soap bubbles remain spherical to much larger sizes. The bubble pictured above, for example, is more than 1 meter in radius and nearly 1 meter in height.
There is a maximum height for a soap bubble, though, and it’s set by the physical chemistry of the surfactants used in the soap. To support itself, the bubble requires a difference in surface tension between the top and bottom of the bubble. A higher surface tension is necessary at the top of the bubble to help prevent fluid from draining away. The difference in surface tension between the top and bottom of the bubble can never be greater than the difference in surface tension between pure water and the soap mixture – thus those values set a maximum height for a bubble. The researchers found their bubbles maxed out at a height of about 2 meters, consistent with their theoretical predictions. (Image credit: C. Cohen et al.; via freshphotons)