Cavitation happens when the local pressure in a liquid drops below its vapor pressure. A low-pressure bubble forms, typically very briefly, when this occurs. These bubbles are spherical unless they form near a surface. In that case, the bubbles take on a flatter, oblong shape. As they collapse, the bubbles form a jet, like the one seen inside the bubble above. The jet extends through the bubble and stretches into a funnel shaped protrusion on the bubble’s far side. Eventually, the whole shape becomes unstable and breaks into many smaller bubbles. Shock waves can be generated in the collapse, too; often the jet generates at least two in addition to the ones created when the bubble reaches its minimum size. This is part of why cavitation can be so destructive near a surface. (Image credit: L. Crum)
Search results for: “art”

“Galaxy Gates”
Viewing fluids through a macro lens makes for an incredible playground. In “Galaxy Gates”, Thomas Blanchard and the artists of Oilhack explore a colorful and dynamic landscape of paint, oil, and glitter. The nucleation of holes and the breakdown of sheets to filaments and droplets plays a major role in the visuals. The surface layer is constantly peeling away to reveal what’s going on underneath. In many cases this initial motion settles into a field of oil-rimmed droplets floating like planets against a colorful galactic backdrop. Watch carefully in the second half of the video, and you can even catch a few instances of a stretched ligament of fluid breaking into a string of satellite drops, like at 1:51. Check out some of Blanchard’s previous work here and here. (Video credit: Oilhack and T. Blanchard; GIFs and h/t to Colossal)

Flow in a Turbine
Fluid flows are complex, complicated, and ever-changing. Researchers use many techniques to visualize parts of a flow, which can help make what’s happening clearer. One technique, shown above, uses oil and dye to visualize flow at the surface. The vertical, black, airfoil-shaped pieces are stators, stationary parts within a turbine that help direct flow. After painting the stator mount surface with a uniform layer of oil, the model can be placed in a wind tunnel (or turbine) and exposed to flow. Air moving around the stators drags some of the oil with it, creating the darker and lighter streaks seen here. Notice how the lines of oil turn sharply around the front of the stator and bunch up near its widest point. Those crowded flow lines tell researchers that the air moves quickly around this corner. (Image credit: D. Klaubert et al., source)

The Surge in the Hourglass

When we watch sands running through an hourglass, we think their flow rate is constant. In other words, the same number of grains falls through the neck at the beginning and the end. In many practical granular flows, like those through industrial hoppers (left), this is not the case. Instead, emptying those containers involves a surge near the end where the discharge rate is higher.
The surge is related to the interstitial fluid – the air, water, or other fluid in the space between the grains. On the right, you see an experiment in which brown grains submerged in green-dyed water are emptied. The dark layer is dyed water initially at the top of the grains. As the container drains, that dyed layer moves down more rapidly than the grains; this indicates that the interstitial fluid is actually being pumped by the draining of the grains. Researchers think this is an important factor affecting the final surge. (Image credits: hopper – T. Cizauskas; discharge graph – J. Koivisto and D. Durian, source; research credit: J. Koivisto and D. Durian; submitted by Marc A)
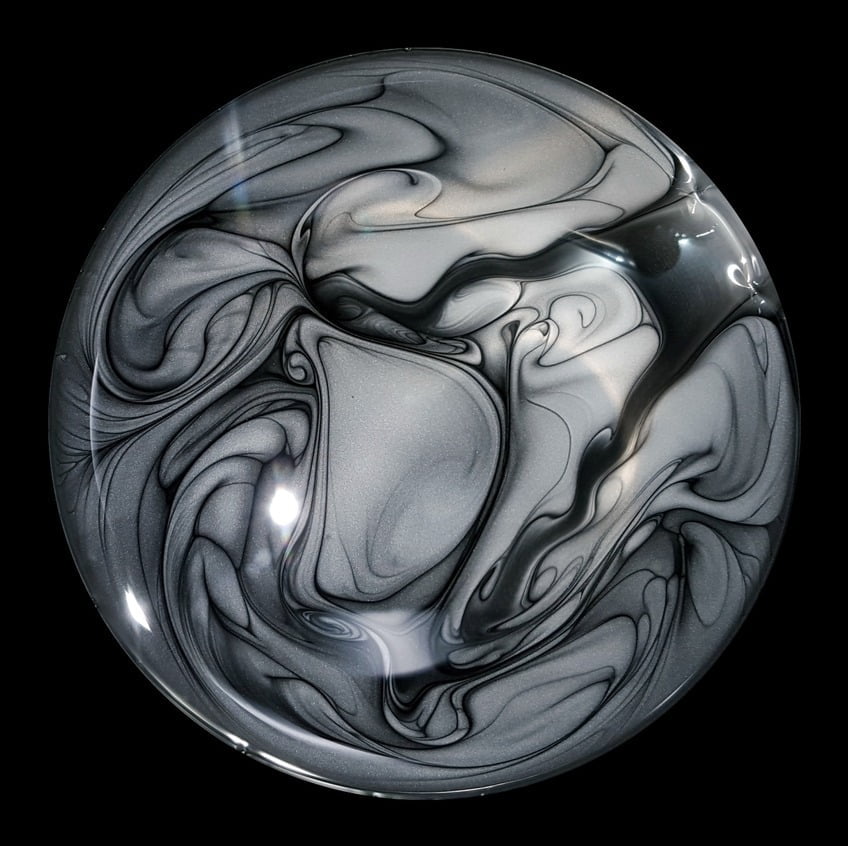
Graphene Swirls
Graphene powder swirls in alcohol in this prize-winning photo from this year’s Engineering and Physical Sciences Research Council photography competition in the UK. The image was captured while producing graphene ink that can print circuits directly onto paper. According to the researcher’s description, this ink is forced through micrometer-sized capillaries at high pressure to rip the layers apart and produce a smooth, conductive ink in solution. In this photo, we seem to see more conventional mixing driven by the powder’s injection and the variations in surface tension due to the alcohol and its evaporation. The graphene leaves behind beautiful streaklines that highlight its path as it mixes. (Image credit: J. Macleod; via Discover)

Reconnecting
Vortices are a common feature of many flows. Here we see a helical vortex tube spinning in a swirling flow. The vortex itself is visible thanks to air trapped in its low-pressure core. As the vortex spins, two sections of it come together. This results in what’s known as vortex reconnection: the vortex lines break apart and rejoin in a new configuration – as a small independent vortex ring and a shorter section of helical vortex. Events like this are common but usually hard to observe directly. They’ve been previously visualized using vortex knots and have even been sighted in the quantum vortices of superfluid helium. (Image credit: S. Skripkin, source; research credit: S. Alekseenko et al., pdf)

The Japanese Pufferfish
[original media no longer available]
If you’ve ever dived or snorkeled over a sandy lake or ocean bottom, you’ve probably seen some neat patterns there. But it’s hard to compete with the Japanese pufferfish for pure artistry. This small fish creates enormous and elaborate designs in the sand in order to attract a mate. The male fish moves the sand into place by flapping his fins very close to the surface. Above a critical flapping velocity, his fins generate vortices capable of picking up sand, as seen below. With repeated passes, the fish is able to excavate the trough that is key to his creation. It’s a constant fight against the current, though.
Puffers aren’t the only ones who flap their fins to move the sands. Rays and flounders use this technique to bury themselves and hide (Video credit: BBC Earth; image credit: A. Sauret, source; research credit: A. Sauret et al.)

A Real Tatooine

Since at least the release of “Star Wars”, we have wondered what life would be like on a circumbinary planet – a planet orbiting two stars. In the past few decades, we have discovered several such planets, but we are still in the early days of modeling the climate of these worlds. One recent study uses the stars of the Kepler 35 system, which are only slightly less luminous than our sun, to explore the climate of an Earth-like water planet.
According to the study, this fictional planet would maintain Earth-like habitability at a distance of 1.165–1.195 astronomical units from its suns’ center of gravity – just a little further out than our own orbital distance. Variables like the planet’s mean global surface temperature and precipitation vary with two distinct periods – the time required for the stars to orbit one another and the time it takes for the planet to orbit its stars. Both factors affect how much sunlight the planet receives. The planet’s climate response to these changes is complex and varies depending on location, but the overall variations observed in the climate are small. It does show, however, that places like Tatooine don’t have to be desert planets! (Image credit: Tatooine – Star Wars; Kepler 35 system – L. Cook; research credit: M. Popp and S. Eggl)

Sorting by Bubble

Microfluidic devices, also known as labs-on-a-chip, require clever techniques for processes like sorting particles by size. One such technique uses an oscillating bubble to sort particles. When the bubble vibrates back and forth (left) it creates what’s known as a streaming flow – large regions of recirculation (shown as gray ellipses in the right image). If the bubble is placed inside a channel, we say that two flows have been superposed; the device combines both the left-to-right flow of the channel and the recirculating streaming flow.
Introduce a micron-sized particle into this combined flow, and it will get carried to the bubble and then bounced around by its effects (left). In fact, the larger the particle is, the more the bubble deflects it relative to the flow. You can see this in the image on the right as well. Here the frame rate has been matched to the bubble’s vibration, so the bubble appears stationary, and the particle paths look smooth. The gray lines show the fluid’s path, and individual solid particles are introduced at the left. The largest particle gets strongly deflected as it passes the bubble and exits at the top-right. A fainter, smaller particle follows after it. Being smaller, the bubble’s deflection on it is weaker, and this second particle exits along a path to the center-right. The result is a fast and simple method for particle sorting. (Image and research credit: R. Thameem et al., source)

Capillary Action in Microgravity
On Earth, gravity dominates over many fluid effects, but in microgravity a different picture emerges. This animation shows a two-channel apparatus partially filled with silicone oil being dropped. While in free-fall, the liquid experiences microgravity conditions and the height of the fluid in the two connected channels changes. The oil meniscus climbs up the walls of the tubes thanks to capillary action. This is the result of intermolecular forces between the liquid and solid walls. Capillary action is most effective in narrow tubes where surface tension and the adhesion between the liquid and solid can actually propel liquid up the walls, as seen here. On Earth we mostly ignore capillary action, except in very small spaces, but for space systems, it is a major force to reckon with in designing flows. (Image credit: NASA Glenn Research Center, source)



