The Martian atmosphere is scant compared to Earth’s, but its winds still sculpt and change the surface regularly. The average atmospheric pressure on Mars is only 0.6% of Earth’s, and the density is similarly low at 1.7% of Earth’s. Despite this thinness, Martian winds are still substantial enough to shift sands on a daily basis, as shown above. These two images were taken one Martian day apart, showing how sand ripples moved and how the Curiosity rover’s tracks can be quickly obscured. Part of the reason Mars’ scant atmosphere is still so good at moving sand is that Martian gravity is roughly one-third of ours; if the sand is lighter, it doesn’t take as much force to move! (Image credit: NASA/JPL-CALTECH/MSSS)
Search results for: “art”

Break-Up of the Chelyabinsk Meteor
In 2013, a meteor about 20-meters in diameter broke up over Chelyabinsk, Russia in a dramatic display that damaged buildings within 100 km and injured more than 1200 people. To better understand the threat presented by such objects, NASA has been conducting 3D, hypersonic simulations like the one shown here. The meteor material is shown in gray and black. Brighter colors like red and yellow indicate the hot, high-pressure shock wave caused when the meteor slams into the atmosphere. Aerodynamic effects quickly erode the meteor, ripping it into pieces that disperse energy explosively in the atmosphere. While you might think the meteor breaking up is good for us, it’s actually the blast waves from its break-up that cause the most damage. (Image and video credit: NASA, source; via Gizmodo)
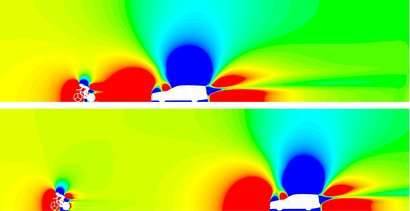
Reader Question: Drafting in Time Trials

In a comment on this recent post regarding drafting advantages to a leader, reader fey-ruz asks:
in cycling, team follow cars are required to maintain a minimum distance from their riders during time trials for this very reason (although i imagine the effects in that context are much smaller and dependent on the conditions, esp the wind speed, direction, and strength). FYFD, is there a simple way to understand where this upstream influence comes from? or a specific term in the navier-stokes equations that it results from?
Cars following riders during a time trial can actually make a huge difference! One study from a couple of years ago estimated that a car following a rider in a short (13.8 km) time trial could take 6 seconds off the rider’s time. The images up top show a simulation from that study with a car following at 5 meters versus 10 meters. The colors indicate the pressure field around the car and rider. Red is high pressure, blue is low pressure. Both the car and the rider have high pressure in front of them; you can think of this as a result of them pushing the air in front of them.
A large part of the rider’s drag comes from the difference in pressure ahead and behind them. (For a look at flow around a cyclist that focuses on velocity instead, check out my video on cycling aerodynamics.) When a car drives close behind a cyclist, it’s essentially pushing air ahead of it and into the cyclist’s wake. This actually reduces the difference in pressure between the cyclist’s front and back sides, thereby reducing his drag. Because cars are large, they have an oversized effect in this regard, but having a motorbike or another rider nearby also helps the lead cyclist aerodynamically.
As for the Navier-Stokes equation – this effect isn’t one that you can really pin down to a single term since it’s a consequence of the flow overall. (Image credits: TU Eindhoven; K. Ramon)

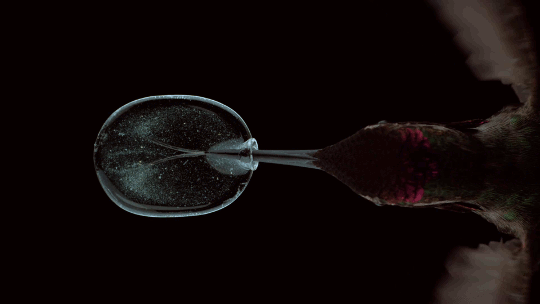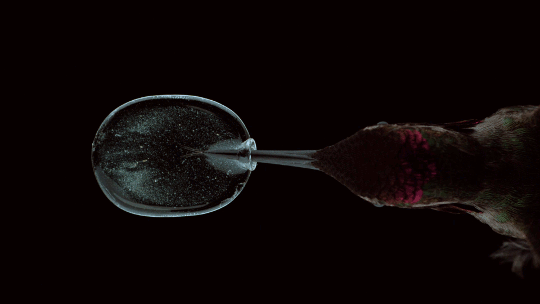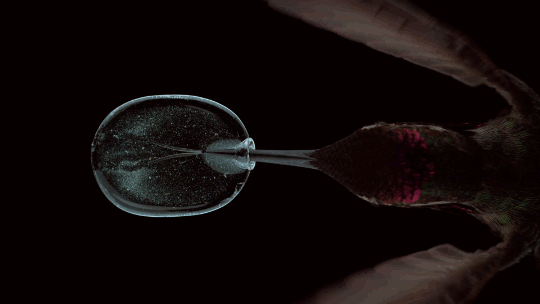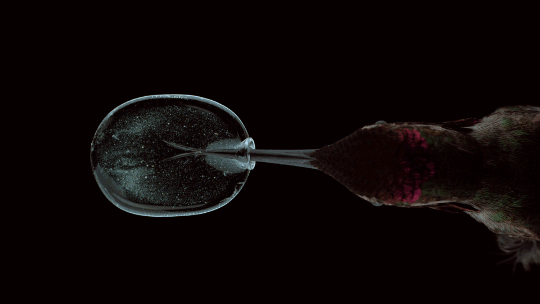
How Hummingbirds Drink
Hummingbirds are incredible acrobatic fliers, capable of hovering for more than 30 seconds at a time, even in windy conditions. Their feeding habits are equally impressive. Many species of hummingbirds have a forked tongue, each half of which curls over like a partial straw. As the bird extends its tongue, its beak compresses the space inside the tongue’s curls. Once in the nectar, both halves of the tongue re-expand, pulling liquid in along the full length of the tongue. For the birds, this is a much faster technique than simply sucking the nectar up like a straw. Hummingbirds can lick nectar more than ten times a second this way. For more gorgeous imagery of hummingbirds, be sure to check out National Geographic’s full feature. (Image credit: A. Varma, source; via Aarthi S.)

Reader Question: Resonating Bottles
Reader shoebill-san asks:
why does it make that weird sound when i blow over a bottle? i did a science experiment in college where we looked at the resonance in a beaker at different water levels, is it like that? related?
Blowing across the top of a bottle creates what’s called Helmholtz resonance, where air inside the neck of the bottle actually vibrates up and down, like you see in the animation above. The stream of air from your mouth creates low pressure just outside the bottle, pulling some of the air out. That air will tend to overshoot, ultimately causing pressure in the bottle neck to drop lower. That vacuum will pull air back into the bottle, at which point the low pressure your blowing supplies pulls it back out, and so on. The actual sound you hear comes from those puffs of moving air. In reality, they move too fast to see; the animation comes from a high-speed video, and I highly recommend watching the full vid.
From your description, I’m not 100% sure what the experiment you did in college was, but I’m guessing it was some variation of the glass harp, where you rub a partially-filled glass and get an eerie sound that varies depending on how much water is in the glass. Like the bottle example above, that’s an example of resonance, but the two are different. In the bottle, it’s the air that’s resonating. For the glass harp, it’s the glass walls themselves that are resonating. The liquid inside just changes the pitch by slowing down the speed at which the glass’s walls vibrate. For a full and fantastic explanation of how that works, check out this video by Dan Quinn. (Image credit: N. Moore, source)

Cycling Skinsuits and Vortex Generators
It didn’t take long for an aerodynamic controversy to crop up in this year’s Tour de France. At the 14km individual time trial, riders from Team Sky wore custom Castelli skinsuits with integrated dot-like patterns on their upper arms (shown above). By the next day, a sports scientist with a competing team cried foul play, claiming that these fabrics could have given Team Sky as much as 25 seconds’ advantage over other riders. The Sky team finished with 4 out of the top 10 places on the time trial, and their leader, three-time Tour winner Chris Froome, finished some 35 seconds ahead of his expected competitors for the yellow jersey.
Vortex generators explained
So how could a few dots make a measurable difference? These protrusions are vortex generators meant to modify flow around a cyclist. Humans are not aerodynamic and what typically happens when air flows over a cyclist’s arms is shown in the flow visualization above: the air follows the curve of the arm part way, then it separates from the body, leaving a region of recirculation that increases drag. Vortex generators can help prevent or delay that drag-inducing flow separation by adding extra energy and turbulence to the air near the arm’s surface. Because turbulent boundary layers can follow a curve longer before separating, this helps reduce the drag by reducing the recirculation zone.
About that time savings
Aerodynamically speaking, those vortex generators can make a difference, but the question is, how much? In his complaint, Grappe cites a 2016 paper by L. Brownlie et al. that wind-tunnel tested different vortex generator patterns for use in running apparel. The speeds tested included those relevant to cycling. The specific numbers Grappe quotes aren’t directly relevant, however:
As noted above, race garments that contain VG provide reductions in Fd of between 3.7 and 6.8% compared to equivalent
advanced race apparel developed for the 2012 London Olympics which in turn provided substantially lower drag than
conventional race apparel.the effectiveness of 5, 10 and 15 cm wide strips of VG applied to each flank of a sleeveless singlet revealed that the 5 cm wide
strips provided between 3.1 and 7.1% less Fd than the 10 cm wide strips and between 1.9 and 4.3% less Fd than the 15 cm wide
strips.Here Brownlie et al. are specifically describing the savings for running apparel, which uses vortex generators in very different places than you would on a cyclist. Note the second quote even refers to a sleeveless singlet, so the vortex generators measured are definitely not in the same place as these skinsuits!
The bottom line
I fully expect that vortex generators give a marginal aerodynamic edge, which is why Sky and other teams have already been using them in competition. But I hesitate to declare that the savings is as high as 5-7%, and I have no way to verify Grappe’s subsequent claims that this translates to 18-25 seconds in the time trial. Those are numbers he gives without citing what model is being used to translate drag gains into time.
In the end, what is needed is clarification of the rules. As they stand, one rule seems to allow the skinsuits because the vortex generators are integrated into the fabric, whereas another states clothing is forbidden “to influence the performances of a rider such as reducing air resistance”. Those two stances seems contradictory, and, for now, the race officials’ verdict to allow the suits stands.
If you want to learn more about aerodynamics and cycling, be sure to check out my latest FYFD video. (Image credits: B. Tessier/Reuters; Getty Images; L. Brownlie et al. 2009; h/t to W. Küper)

When Walls Chirp
If you’ve ever clapped near a wall with a corrugated surface, you may have noticed some strange echoes. Surfaces like these can cause a chirping sound to observers. The reason, as Nick Moore explains in the video above, is that the original sound reflects off the corrugations at different times and travels back to the observer such that the first reflections to arrive are closely spaced (and thus higher pitched) while the later reflections are spread further out. This creates a chirp that starts at a high pitch and then falls to lower ones. Have you ever come across structures that do this? (Video credit: N. Moore)

“Fractal”
Timelapses are a wonderful way to capture the power and majesty of storms like the supercell thunderstorms featured in Chad Cowan’s “Fractal”. The video contains snapshots from six years’ worth of storms over the US’s Great Plains. The highlights include some spectacular mammatus clouds (0:30) and excellent billowing cloud formation (1:27) with turbulence every bit as towering as that of a volcanic plume. June is one of the best months for amazing storms in the Great Plains, largely thanks to the atmospheric mixing that occurs over the Rocky Mountains. If you have the opportunity to witness these amazing natural displays, enjoy it, but be safe! (Video credit: C. Cowan; image via Colossal)


Watching Radiation
We’re used to radiation being invisible. With a Geiger counter, it gets turned into audible clicks. What you see above, though, is radiation’s effects made visible in a cloud chamber. In the center hangs a chunk of radioactive uranium, spitting out alpha and beta particles. The chamber also has a reservoir of alcohol and a floor cooled to -40 degrees Celsius. This generates a supersaturated cloud of alcohol vapor. When the uranium spits out a particle, it zips through the vapor, colliding with atoms and ionizing them. Those now-charged ions serve as nuclei for the vapor, which condenses into droplets that reveal the path of the particle. The characteristics of the trails are distinct to the type of decay particle that created them. In fact, both the positron and muon were first discovered in cloud chambers! (Image credit: Cloudylabs, source)

Tendrils of Fog
Fog snakes its way from the ocean into the Strait of Juan de Fuca in this animation constructed from satellite imagery. The strait lies between Vancouver Island and the Olympic Peninsula in the Pacific Northwest. Fogs like this form when skies are clearer and heat from the surface is able to escape upward. The surface air then cools and condenses into fog. Steady winds pushed fog into the strait over the course of about 9 hours. There’s a remarkable level of detail in the satellite images, taken by the new GOES-16 satellite that launched in late 2016. Notice the ragged wave front as the fog stretches eastward and the shock-wave-like lines behind it in the strait. Both result from interactions between the fog cloud and the shape of the land masses it’s encountered. (Image credit: NASA Earth Observatory)