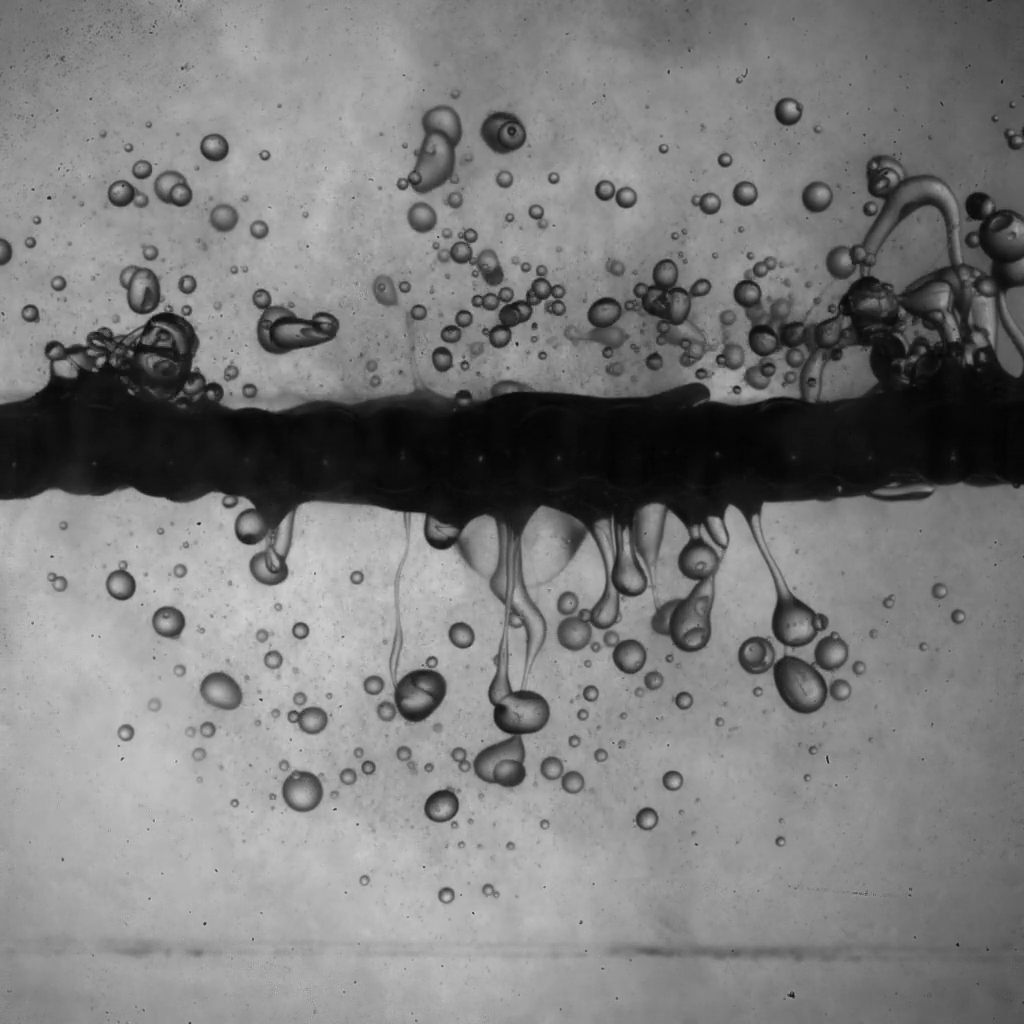
Spraying a surface clean with a jet of fluid can be an energy-intensive operation. But a recent experiment shows that pulsed flow — which creates vortex rings — could be a viable cleaning alternative. Here we see vortex rings impacting a porous, beaded surface that’s covered in oil. Vortex rings with lots of rotation actually pass through the beads, knocking oil off both the front and back surfaces (Image 1). Even with a lower rotation rate, a vortex ring can still help clean the upper surface (Image 2). (Image and research credit: S. Jain et al.; via APS Physics)
Category: Research

Sandgrouse Soak in Water
Desert-dwelling sandgrouse resemble pigeons or doves, but they have a very different superpower: males can soak in and hold 25 milliliters of water in their feathers, which they carry tens of kilometers back to their chicks. The key to this ability is the microstructure of the bird’s breast feathers. Unlike other species, where feathers have hooks and grooves that “zip” them together, the sandgrouse’s specialized feathers have tiny barbules with varying bending stresses. When dipped in water, their curled shape unwinds, allowing water to soak in through capillary action. Barbules at the tips curl inward, holding the water in place so that the sandgrouse can fly home with it.
Studying nature’s solutions for water-carrying will help engineers design better materials for human use, whether that’s a water bottle that avoids sloshing or a medical swab that’s better at absorbing and releasing fluids. (Image and video credit: Johns Hopkins; research credit: J. Mueller and L. Gibson; via Forbes; submitted by Kam-Yung Soh)

Overcoming Turbulence
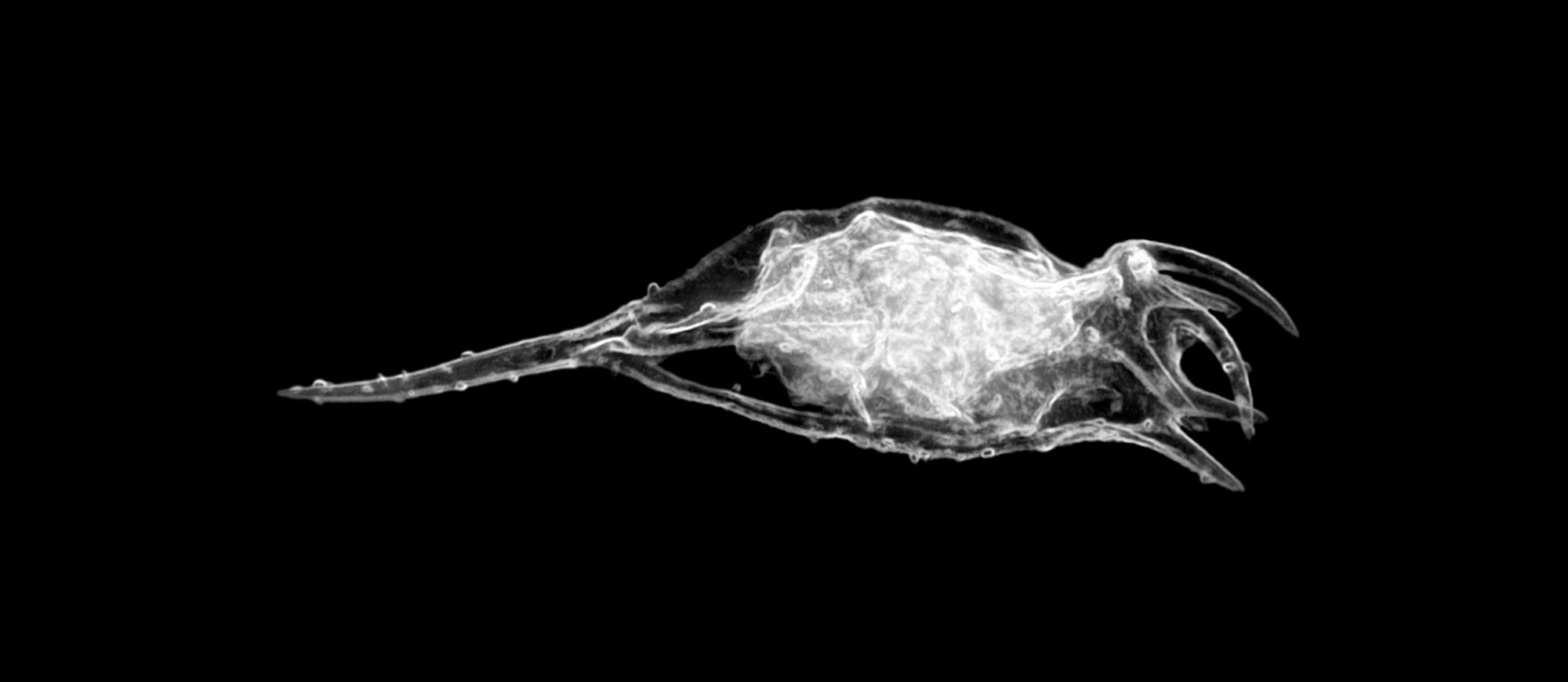
Despite their microscopic size, many plankton undertake a daily migration that covers tens of meters in depth. As they journey, they must contend with currents, turbulence, and other flows that could knock them off-course. And, increasingly, research shows that a plankton’s shape makes a big difference in these flows.
Spherical plankton tend to cluster in areas of flow moving opposite to their direction of travel. But more elongated plankton can resist — or even reverse — this tendency, helping them stay on track. In turbulence, elongated swimmers are also better at keeping their thrust oriented in the desired direction of travel. So both nature and engineers should favor elongated microswimmers when contending with turbulence and potential crossflows. (Image credit: Picturepest/Flickr; research credit: R. Bearon and W. Durham)

How Hagfish Slime Clogs
When attacked, the eel-like hagfish slimes its predator, clogging the fish’s gills so that it can escape. A recent study looks at just what makes the slime so effective. There are two main (non-seawater) components to hagfish slime: mucus and threads. The team’s experiments showed that the slime’s clogging is due almost entirely to the mucus; the clogging power of full slime and mucus-only slime is almost identical.
So what are the threads for? They make it harder for the mucus to get washed away. Mucus alone isn’t able to clog as effectively after a single rinse, but, with the threads included, the slime hardly budges. That staying power makes it all the harder for a predator to clear its gills once slimed. In fact, it’s still unclear to scientists whether a slimed fish can free itself from the clogging. After all, the attacker can’t use the hagfish’s trick to free itself from slime. (Image credit: dirtsailor2003/Flickr; research credit: L. Taylor et al.)

Predicting Heat Waves
The United States, Europe, and Russia have all seen deadly, record-breaking heat waves in recent years, largely in areas that are ill-equipped for sustained high temperatures. A new paper presents a theory that predicts how hot these heat waves can get and what mechanism ultimately breaks the hot streak.
Heat waves start when an area of high-pressure air forms over land, with an anticyclone circulating around it. Air at the center of the zone warms and rises, and if the anticylone can’t move, temperatures will just keep rising. Despite the heat, there is still moisture in the rising air of a heat wave. The authors found that if that moist air can reach an altitude where the atmospheric pressure is 500 hPa (a typical altitude of 5-7 km), then the maximum daily temperature will stop rising. At that altitude, the moist air can condense into rain, and, even if that rain evaporates before reaching the ground, it is enough to cool temperatures.
The key variable in the theory is the atmospheric temperature at 500 hPa, something that meteorological models are able to predict well up to three weeks in advance. That means this theory should enable meteorologists to give advanced warning of high temperatures, helping communities prepare. (Image credit: T. Baginski; research credit: Y. Zhang and W. Boos; via APS Physics)

Bending in the Stream
Nature is full of cilia, hairs, and similar flexible structures. Unsurprisingly, flows interact with these structures very differently than with smooth surfaces. Here, researchers investigate flow in a channel lined with flexible, hair-like plates. Initially, the channel is filled with oil and dark particles that help visualize the flow. Then, they pump water into the setup.
As the water intrudes, it forms an interface with the oil. That interface is powerful enough to bend individual hairs in the system. When the hair bends far enough, it can touch its neighbor, sealing the oil inside the gap between them. Along the length of the channel, this behavior leads to trapped pockets of oil that never drain, no matter how much water flows by. (Image and research credit: C. Ushay et al.)

Hawaiian Magma Complex
Few volcanoes are as well-studied as those of the Big Island of Hawai’i. With a host of seismic monitors and frequent eruptions, scientists know the near-surface region of Hawai’i well. But a recent study looked at nearly 200,000 seismic events after the 2018 collapse of Kilauea’s crater and found hints of what goes on much deeper.
Mapping out earthquakes beneath the island revealed a cluster of activity near a village named Pahala. These earthquakes took place 36 to 43 kilometers below the surface and seem to be connected to magma filling a sill complex there. From that deep reservoir, the team was also able to map seismic activity leading upwards to both Kilauea and Mauna Loa volcanoes. Despite the 34 kilometers between those two volcanoes, they appear to be fed through the same web of magma! (Image credit: top – USGS, illustration – J. Wilding et al.; research credit: J. Wilding et al.; via Physics Today)

This cartoon illustrates the web of magma linking Kilauea and Mauna Loa deep underground. 
Hunting By Whisker
Seals and sea lions often hunt fish in waters too dark or turbid to rely on eyesight. Instead, they follow their whiskers, using the turbulence generated by a fish’s wake. The vortices shed by the fish cause the seal’s whiskers to vibrate, giving them sensory information. To better understand what a seal can derive from this, a recent experiment looked at what a thin whisker can pick up from an upstream cylinder.
As expected, the strength of the whisker’s vibration fell off the farther away the cylinder was. But the researchers found that, if they moved the cylinder quickly — like a fish trying to dart away — the vibration of the whisker was stronger. They also found that the whisker was sensitive to misalignment. If the cylinder was placed ahead and to the side of the whisker, the whisker would still vibrate but would do so around a different equilibrium position. That result implies that a seal can get information both about the fish’s speed and direction, simply from the twitch of its whiskers. (Image credit: seal – K. Luke, illustration – P. Gong et al.; research credit: P. Gong et al.; via APS Physics)

Illustration of a seal following a fish versus the experiment, a whisker following a cylinder’s wake. 
A Toad’s Sticky Saliva
Frogs and toads shoot out their tongues to capture and envelop their prey in a fraction of a second. They owe their success in this area to two features: the squishiness of their tongues and the stickiness of their saliva. The super squishy toad tongue deforms to touch as much of the insect as possible. That shape-changing helps deliver the saliva, which is an impressively fast-acting, shear-thinning fluid. Under normal circumstances, the saliva is sticky and about as viscous as honey. But the shear from the tongue’s impact makes the saliva flow like water, spreading across the insect’s body. Then it morphs back into its viscous, sticky self, providing enough adhesive power that the insect can’t escape the toad pulling its tongue back in. (Video credit: Deep Look/KQED; research credit: A. Noel et al.)

Swimming With Corkscrews
For many microswimmers, like bacteria or spermatozoa, swimming through common fluids is like moving through mud. Unless they can produce enough thrust to overcome a fluid’s yield-stress, they are effectively stuck in a solid. A recent study breaks down exactly what a microswimmer has to manage, assuming they use a helical, corkscrew-like tail for propulsion.
The first barrier is creating enough force to be able to rotate in the fluid, but that alone is not enough to ensure forward motion. Once rotating, the swimmer’s thrust has to be large enough to deform the fluid around it. Without that, the swimmer is stuck. And, finally, once they’re moving, the swimmer’s tail pitch determines how fast they can move and whether the fluid’s characteristics slow it down.
The researchers hope their work can shed light on propulsion for bacteria in the body, as well as larger creatures like burrowing earthworms and fruit-invading parasites. (Image credit: SwedishStockPhotos; research credit: F. Nazari et al.; via APS Physics)