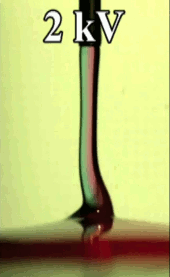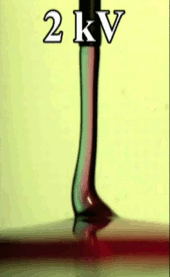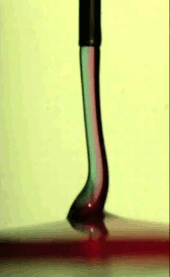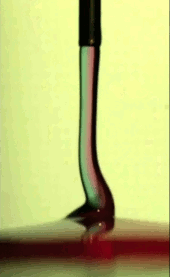Major volcanic eruptions can be accompanied by pyroclastic flows, a mixture of rock and hot gases capable of burying entire cities, as happened in Pompeii when Mt. Vesuvius erupted in 79 C.E. For even larger eruptions, such as the one at Peach Spring Caldera some 18.8 million years ago, the pyroclastic flow can be powerful enough to move half-meter-sized blocks of rock more than 150 km from the epicenter. Through observations of these deposits, experiments like the one above, and modeling, researchers were able to deduce that the Peach Spring pyroclastic flow must have been quite dense and flowed at speeds between 5 – 20 m/s for 2.5 – 10 hours! Dense, relatively slow-moving pyroclastic flows can pick up large rocks (simulated in the experiment with large metal beads) both through shear and because their speed generates low pressure that lifts the rocks so that they get swept along by the current. (Image credit: O. Roche et al., source)
Category: Research

Rogue Wave Recreated

If you look online, the term “rogue wave” gets thrown around a lot – a whole lot. And most of the videos you see of “rogue waves”, “freak waves”, and “monster waves” are just, in fact, big waves. What makes a deep-water ocean wave a rogue, scientifically speaking, is that it is extreme compared to its surroundings. One definition requires that a rogue wave be more than twice as tall as the height of average large waves in the area – like the rogue that takes out the Lego boat above. Outside the lab, this is a rare event – fortunately – because a true rogue wave has tremendous destructive power and seems to appear out of the blue.
This seemingly unpredictable behavior is thought to arise from nonlinear interactions between waves. Essentially, under the right conditions, a rogue wave grows monstrously large by sucking energy out of other surrounding waves. One way to try and predict rogue waves is to measure all the waves nearby and simulate their potential nonlinear interactions computationally – but this is time-consuming and requires a lot of computing power.
Instead, researchers have developed an alternative method, illustrated in the time series above. Instead of considering the rogue potential for all waves, they identify waves with characteristics that make them more likely to go rogue and focus on simulating those waves. In the animation, the wave packets are colored from green to red based on their increasing likelihood of turning into rogue waves. The algorithm is simple enough to run quickly on a laptop and can provide a couple minutes of warning to a ship’s crew – enough time to batten down before the wave hits. (Image credits: simulation – T. Sapsis et al., source; experiment: N. Ahkmediev et al., source; via The Economist and MIT News; submitted by 1307phaezr)

Turbulent Convection
These golden lines reveal the complexity of turbulent convective flow. They come from a numerical simulation of turbulent Rayleigh-Benard convection, a situation in which fluid trapped between two plates is heated from below and cooled from above. This situation would typically create convection cells similar to those seen in clouds or when cooking. Inside these cells, warm fluid rises to the top, cools, and sinks down along the sides. With large enough temperature differences, instabilities will occur and cause the flow to become turbulent so that the clear structure of convection cells breaks down into something more chaotic. Such is the case in this simulation. This visualization shows skin friction on the bottom (heated) plate in a flow of turbulently convecting liquid mercury. The bright lines are areas with large velocity changes at the wall, an indication of high shear stress and vigorous convective flow. (Image credit: J. Scheel et al.; via Gizmodo)

Humans Running on Water
How fast does a speedster like The Flash or Dash Parr from The Incredibles have to go to run on water? As we saw from other water-walkers like the basilisk lizard and the western and Clark’s grebes, any large creature wanting to run on water needs to generate the necessary vertical impulse by hitting the water hard, pushing off against the cavity that creates, and pulling their foot up before the cavity collapses around it.
Using basilisk lizards as our guide, we can build a simplified hydrodynamic model (following Glasheen and McMahon and Minetti et al.) to describe this process and predict a speedster’s necessary speed. If we assume our runner removes their foot before the cavity collapses, we have a relatively simple relation to satisfy, namely: the vertical impulse from the slap combined with the vertical impulse from the push, or stroke, must equal or exceed the impulse from the runner’s weight:
(Impulse from slap) + (Impulse from stroke) >= Impulse from runner’s weight
The impulse from the runner’s weight is relatively straightforward. It depends on the runner’s mass, gravity, and the time it takes the runner to complete a step. The other two terms are a bit more complicated and require some approximations. One is that we’ll treat the runner’s foot like a circular disk – this makes it easier to figure out the drag while the runner pushes against the water. Ultimately, the model requires five variables (four, if we assume that we’re on Earth):
– the runner’s mass
– the area of the runner’s foot
– the depth the runner’s foot reaches underwater
– the time it takes the runner to take one step
– the acceleration due to gravityI will spare you the math, but I’ve created an online calculator (now with English or metric versions) with the model, so you can follow along with my math or play around with your own numbers.
Click through to see how fast a human has to go to run on water.
So how fast would The Flash have to run? Barry Allen is grown man, roughly 75 kg in mass, with a foot area of about 314 cm^2. We can assume that he pushes his leg about 0.15 m into the water with each step. The best human sprinters run with a step time of 0.2 – 0.26 seconds, but Barry’s a metahuman, so we’ll give him the benefit of the doubt and say that he can take a step in 0.15 seconds. (Let’s be honest, he’s probably capable of faster than that!)
To keep from sinking, The Flash would have to strike his feet against the water at about 37 m/s. It’s a little tough to say exactly how that would translate into forward speed. Both basilisks and grebes strike the water at a higher speed than their forward velocity. Since their feet are parallel to the surface when they strike, the slap phase only gives them vertical impulse. Their forward velocity comes from the stroke phase where they can push off against the water. This suggests that a runner who generates a lot of their vertical impulse during the slap phase will be able to get more forward velocity out of the stroke phase because they can afford to push forward off the cavity instead of mostly up. That’s consistent with what we observe in the lizards and grebes; the grebe gets more of its impulse from the slap and its forward velocity is a larger percentage of its foot impact velocity compared to the basilisk.
Using the lizards and birds as our guide, we can estimate that The Flash, who gets about 45% of his necessary vertical impulse from slapping, will have a forward velocity of about 27 m/s or 98 kph. That’s a lot faster than any human has ever run – Usain Bolt has managed about 44.7 kph – but it’s not that fast. In CW’s The Flash TV show, his team estimates that he must run 650 miles per hour, or 1050 kph, to run on water. That is way faster than necessary!
How about Dash Parr, though? Dash is about 10 years old, so he’s a lot smaller than Barry. That means he has less mass to keep afloat (about 32 kg), but it also means that he has smaller feet (154 cm^2) and shorter legs (0.1 m foot depth). For the same stride rate, that means that Dash has to hit the water at 47 m/s, about 25% faster than Barry. It also means that Dash gets a tad more oomph from his slap (~46%) and runs across the water at 128 kph, about 30% faster than Barry has to go.
That’s totally doable for a superhero, but what about us regular humans? Sadly, our large mass and small feet won’t let us run on water like The Flash or Dash, but there are ways to bend the rules. One is to reduce gravity – this was the subject of an Ig Nobel prize-winning study by Minetti et al. The researchers put fins on volunteers, suspended them from a harness to reduce their effective weight, and got them to run in place in a pool. They found that fin-augmented humans could run on water in gravity about 20% of Earth’s.

Another technique is to increase a runner’s effective foot area without making them bother to lift the foot out of the water. In essence, a human can run on water across over-sized lily pads. In their study, Lothman and Ruina accomplished this with plywood pads laid out in a pool. The pads were buoyant enough to stay afloat at the water surface but would sink if a person stood still on them. But by running quickly from one to the next, their test subject was able to successfully run across water.
So, there you have it: speedsters can run on water without breaking autobahn speed limits and the rest of us can cheat. Be sure to check out the online calculator and play with the model yourself. And join me tomorrow for my special webcast with Professor Tadd Truscott and Randy Hurd, who inspired this foray into water-walking!
ETA: I updated the calculator so that there is now an English unit version as well as a metric unit version.

Research credits:
– Glasheen and McMahon, “A hydrodynamic model of locomotion in the basilisk lizard”, 1996.
– Minetti et al., “Humans running in place on water at simulated reduced gravity,” 2012.
– Lothman and Ruina, “Humans can run on water using big instantly-changable shoes,” 2012.
(Image credits: Disney, CW/DC Comics, Lothman and Ruina, source, and The Splash Lab)

Jumping Off Water

Many insects and arachnids can walk on water by virtue of their hydrophobicity and small size. With their light weight and skinny legs, these invertebrates curve the air-water interface like a trampoline, with surface tension providing the elasticity that keeps them afloat. What’s truly incredible, though, is that many of these creatures, like water striders, can actually jump off the water surface.
The top animation shows high-speed video footage of a water strider leaping off the water. Notice how it distorts the air-water interface but doesn’t break the surface – it makes no splash.
The key is not to push too hard. If the insect exerts a force exceeding the limits of what surface tension can withstand, then its legs will break the water surface and it will lose energy to drag and viscous forces. The insect must generate its jumping force without exceeding a hard limit.
The water strider achieves this feat not by pushing downward but by rotating its middle and hind legs. Rotating its legs allows the insect to maintain contact with the water surface longer and continue deforming the interface as it jumps. This maximizes the momentum it transfers to the water, which, in turn, increases the insect’s take-off velocity. By studying and then emulating this mechanism, scientists were able to successfully create a tiny 68-mg water-jumping robot. (Image credits: J. Koh et al., sources, PDF)
This week FYFD is exploring the physics of walking on water, all leading up to a special webcast March 5th with guests from The Splash Lab.

Grebe Rushing Physics
As capable a water-runner as the common basilisk is, the western and Clark’s grebe is even more impressive. Not only do these birds weigh up to three times as much as an adult basilisk, but they start their water-walking from inside the water, which requires overcoming much more hydrodynamic force.
Like the lizards, grebes must slap the water with their feet to generate upward forces capable of supporting their weight above water. The birds take as many as 20 steps a second – an incredible and unmatched stride rate for a creature their size. Their feet impact the water at up at 4.5 m/s, which generates an impulse equivalent to 30-55% of the grebe’s weight. The rest of the necessary impulse comes from the stroke phase, where the bird pushes its foot down against the water.
When retracting its foot, the grebe extracts the foot with a sideways motion through the water – unlike the basilisk which pulls its foot out through the air cavity its stroke created. In order to reduce drag, the grebe’s foot collapses into a more streamlined shape as it gets pulled from the water, letting the bird set up for the next step. (Image/video credit: B. Struck, source; research credit: G. Clifton et al.)
This week FYFD is exploring the physics of walking on water, all leading up to a special webcast on March 5th with guests from The Splash Lab. The live webcast will be open to all FYFD patrons, so be sure to sign up if you want to tune in.

Surface-Tension Supported Walkers

Nature’s smallest water-walkers use surface tension to keep themselves afloat. This includes hundreds of species of invertebrates like insects and spiders as well as the occasional extremely tiny vertebrate, like the 2-4 cm long pygmy gecko shown above. These animals typically have very thin parts of themselves touching the water – like the spindly legs of the water strider. These skinny appendages curve the air-water interface and that curvature, along with the water’s surface tension, generates the force supporting the animal.
Staying afloat on surface tension does little good if a raindrop or passing splash submerges these tiny water-walkers. To avoid that fate, these animals are also hydrophobic or water repellent. This adaptation keeps them from drowning and helps them enhance the curvature where their feet meet the water.
Those tiny indentations can also be important for the animal’s propulsion. Water striders, for example, use their long middle legs like oars to propel themselves. Any rower will tell you that sticks make poor paddles – they’re just not good at transferring momentum to the water. But curving the surface and then pushing off that curvature works remarkably well. It’s how the water strider creates the vortices in its wake in the image above.
For more on water strider propulsion, I recommend this Science Friday video. If you’d like to see the gecko in action, check out BBC Life’s “Reptiles and Amphibians” episode, which is available on Netflix in the U.S. (Image credits: pygmy gecko, BBC; water strider, J. Bush et al.)
This week FYFD is exploring the physics of walking on water, all leading up to a special webcast on March 5th with guests from The Splash Lab. You don’t want to miss it!

The Basilisk Lizard
One of the most famous water-walking creatures is the common basilisk lizard. These South American reptiles are far too large to be kept aloft by surface tension and other interfacial effects. They generate the vertical force necessary to stay above water by slapping the water hard and fast. There are three phases to a basilisk’s water running gait: the slap, the stroke, and the retraction.
In the slap phase, the lizard slams its foot flat against the water surface at a peak velocity of about 3.75 m/s. The impact pushes water down and generates an upward force on the lizard that accounts for between 15-30% of the lizard’s body weight, depending on the size of the lizard. The rest of the upward force comes from the stroke phase, where the lizard pushes its foot downward in the water, causing an air cavity to form.
The air cavity is vital for the last phase of the lizard’s step. The basilisk must pull its foot out and prepare for the next slap, ideally doing so without generating too much drag. The lizard does this by pulling its foot through the air cavity before it seals. Doing so through air is much easier than through water.
Water-walking this way requires fast reflexes. Basilisks take up to 20 steps per second when running across water and reach speeds of about 1.6 m/s. Although both juvenile and adult basilisks can run on water, the smaller lizards do better because they can generate more than enough impulse to overcome their weight. (Image credit: T. Hsieh/Lauder Laboratory, source; video credit: BBC; research credits: J. Glasheen and T. McMahon, G. Clifton et al.)
This week FYFD is exploring the physics of walking on water, all leading up a special webcast March 5th with guests from The Splash Lab.

Watching a Sneeze
What does a sneeze look like? You might imagine it as a violent burst of air and a cloud of tiny droplets. But this high-speed video shows, that’s only part of the story. The liquid leaving a sneezer’s mouth and nose is a mixture of saliva and mucus, and in the few hundred milliseconds it takes to expel this air/mucosaliva mixture, there’s not enough time for the liquid to break into droplets. Instead, liquid leaves the mouth as a fluid sheet that breaks into long ligaments.
Because mucosaliva is viscoelastic and non-Newtonian, it does not break down into droplets as quickly as water. Instead, when stretched, the proteins inside the fluid tend to pull back, causing large droplets to form with skinny strands between them – the beads-on-a-string instability. The end result when the ligaments do finally break is more large droplets than one would expect from a fluid like water. Understanding this break-up process and the final distribution of droplet sizes is vital for better understanding the spread of diseases and pathogens. (Video credit: Bourouiba Research Group; research paper: B. Scharfman et al., PDF)

Electric Coiling
A falling jet of viscous fluid–like honey or syrup–will often coil. This happens when the jet falls quickly enough that it gets skinnier and buckles near the impact point. Triggering this coiling typically requires a jet to drop many centimeters before it will buckle. In many manufacturing situations, though, one might want a fluid to coil after a shorter drop, and that’s possible if one applies an electric field! Charging the fluid and applying an electric field accelerates the falling jet and induces coiling in a controllable manner.
An especially neat application for this technique is mixing two viscous fluids. If you’ve ever tried to mix, say, food coloring into corn syrup, you’ve probably discovered how tough it is to mix viscous substances. But by feeding two viscous fluids through a nozzle and coiling the resulting jet, researchers found that they could create a pool with concentric rings of the two liquids (see Figure C above). If you make the jet coil a lot, the space between rings becomes very small, meaning that very little molecular motion is necessary to finish mixing the fluids. (Image credits: T. Kong et al., source; via KeSimpulan)