Unusually high rainfall in Bolivia’s Salar de Uyuni turned the world’s largest salt flat into a shallow salt lake. These natural-color satellite images show the area in late January 2022. If you zoom in on the full resolution image, there are incredible detailed swirls in the water. It’s like peering at an abstract or Impressionist painting. The many colors are attributable to several sources, including volcanic sediments, runoff, and a variety of microbes and algae thriving in the mineral-filled waters. (Image credit: L. Dauphin; via NASA Earth Observatory)
Year: 2022

Parametric Resonance
At first glance, Steve Mould’s video on parametric resonance has nothing whatsoever to do with fluid dynamics. He uses a pendulum suspended on a spring to demonstrate how driving a system at a frequency that’s a multiple of the system’s natural frequency can add energy through resonance. Although his examples don’t use fluids, this phenomenon happens there, too, especially in vibrated fluid systems. Take, for example, this droplet bouncing on a vibrating pool. Depending on the amplitude of the vibrations driving the system, the droplet may bounce in time with the vibration, in time with the waves, or at a frequency twice that of the vibration. (Image and video credit: S. Mould)

By pulling on the string each time the mass swings through its lowest point (i.e., twice per swing cycle), Steve adds energy to the system, which is reflected in the increasing amplitude of the pendulum’s swing. This is an example of parametric resonance. 
Leidenfrost On Ice
We’ve seen many forms of Leidenfrost effect — that wild, near-frictionless glide that liquid droplets make on a very hot surface — over the years, but here’s a new one: the three-phase Leidenfrost effect. Researchers found that they could generate a Leidenfrost effect using an ice disk placed on an extremely hot surface. During the effect, the ice and its melting layer of water glide on vapor, hence the name.
The team found that getting a three-phase Leidenfrost effect requires a much, much higher temperature than the regular Leidenfrost effect. Water will get its glide on at 150 degrees Celsius. Getting ice to glide on the same surface required a stunning 550 degrees Celsius! Why the big difference? The challenge is that water layer, which, by definition, has a 100-degree difference between its boiling side and its frozen boundary. It takes so much heat to maintain that layer that there’s little energy left over for evaporation; that’s why it takes so much more energy to get the three-phase Leidenfrost effect. (Image and research credit: M. Edalatpour et al.; via Ars Technica; submitted by Kam-Yung Soh)

Spinning Tops
What does the flow look like around a spinning top? Here, researchers used dye to visualize what happens in a Newtonian fluid (like air or water) as well as a viscoelastic fluid. The Newtonian fluid (upper images) divides into two circulating zones, one below the top and one above. They both take the shape of a toroidal, or donut-shaped, vortex, visible here in cross-section.
The long molecules of the viscoelastic fluid lend it elasticity to resist stretching. The result is a very different flow field. Beneath the top, there’s still a toroidal vortex, though it appears tighter. But around the upper part of the top, there’s a butterfly-like region of recirculation! (Image credit: B. Keshavarz and M. Geri)

Superhydrophobic Drag
Using air or bubbles to reduce drag on boats is a popular idea, whether using supercavitation, the Leidenfrost effect, or superhydrophobic coatings. But most of the experiments done thus far use spheres rather than realisitic boat shapes. In this study, the researchers used two model boats — one with a hydrofoil and the other in a conventional motorboat shape — and applied superhydrophobic coatings to different parts of the model to see how superhydrophobicity affected the overall drag.
Perhaps surprisingly, they found that superhydrophobic coatings can actually increase the drag! The effect was particularly stark for the hydrofoil boat (Image 2), where the surface jets (lower half) caused by the superhydrophobic coating slowed the boat by 30% compared to its unmodified speed (upper half).
For the speedboat, a superhydrophobic hull made no overall difference in its drag, though it changed how water splashed in its wake. And coating the boat’s propeller was particularly detrimental, resulting in a speed up to three times slower. Overall, the study suggests that superhydrophobic coatings may be useful in some circumstances, but they have to be applied carefully, as they can have negative impacts, too. (Image credits: top – S. Anghan, others and research credit: I. Vakarelski et al.)

Fagradalsfjall Volcano
We’ve seen a lot of drone photography from volcanic eruptions in the last few years, but this footage from Iceland Aerials seems even more daredevil than usual. In this video, you can cruise over fountains of lava and watch as it cascades downhill. The perspective on some of these shots is absolutely unreal; it almost seems like it would have to be CGI. (Video credit: Iceland Aerials; via Colossal)

Eruption in a Box
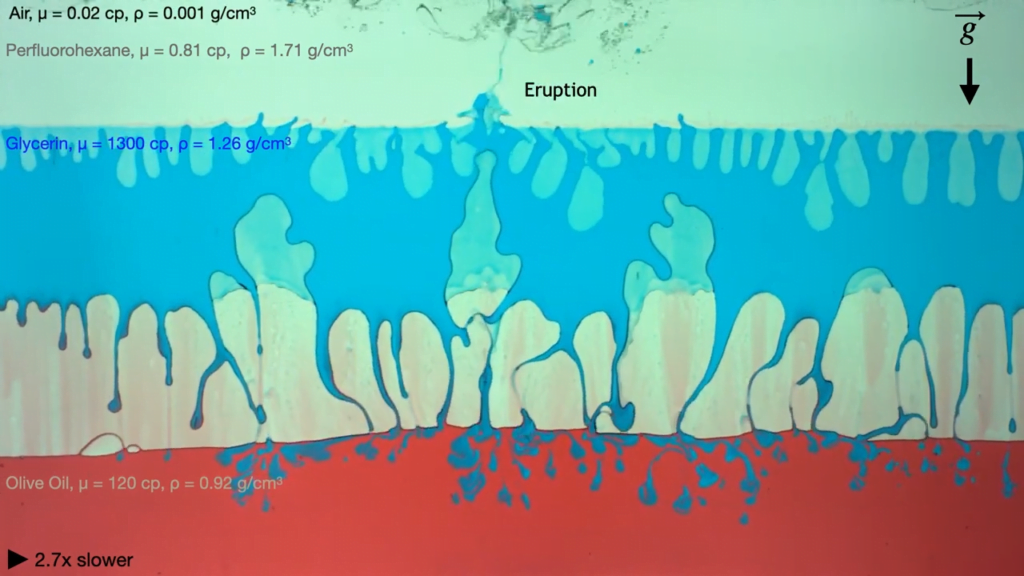
In layers of viscous fluids, lighter and less viscous fluids can displace heavier, more viscous liquids. Here, researchers demonstrate this using four fluids sandwiched between layers of glass and mounted in a rotating frame. (Think of those liquid-air-sand art frames found in museums but bigger!)
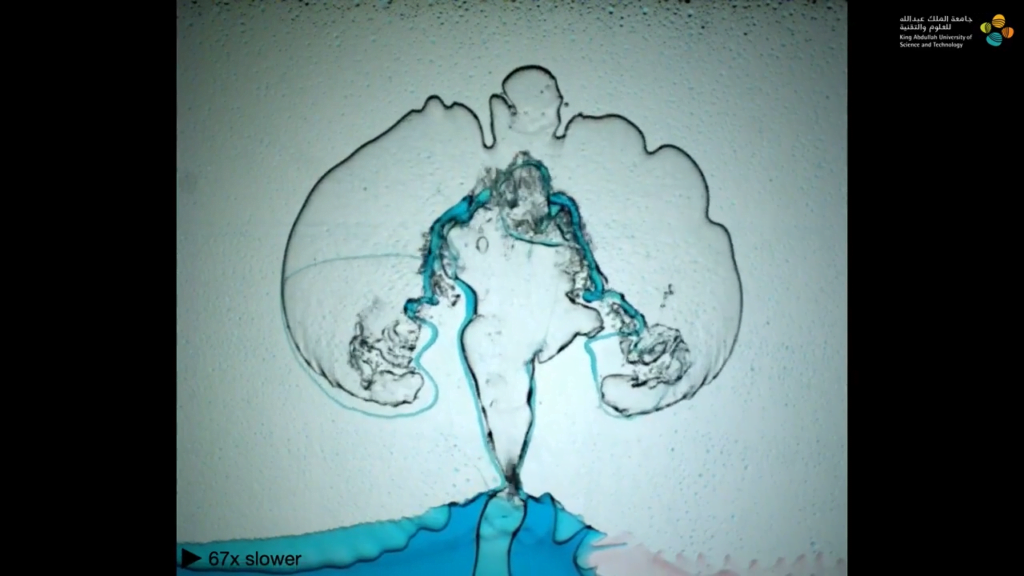
In their first example, each layer of fluid is denser than the one beneath it, so buoyancy forces the lowest layer — air — to rise. The air pushes its way through the more viscous layer of olive oil, then slowly makes its way through the even more viscous glycerin before bursting through the last layer in an eruption. As the team varies the viscosity and miscibility of the layers, the movement of the buoyant fluids through the viscous layers changes dramatically. (Image and video credit: A. Albrahim et. al.)

Bendable Ice
Ice — as we typically encounter it — is extremely brittle and easily broken. That’s due to defects in the ice, places where atoms have settled into a spot that does not match the perfect crystalline alignment. Because tiny defect-free threads of ice made by researchers turn out to be wildly flexible!
To make these perfect ice strands, each of which is a tiny fraction of the thickness of a human hair, researchers applied an electric voltage to a needle in a water-vapor-filled chamber. The technique condensed ice microfibers with perfect crystal structures in a matter of seconds. When bent, the microfibers actually shift from one crystalline arrangement to another in order to carry stress, and once the force is removed, the thread reverts back to its initial straight form. (Image and research credit: P. Xu et al.; via Science News; submitted by Kam-Yung Soh)

Columbia Glacier’s Retreat
In southeastern Alaska, the Columbia Glacier once stretched as far as Heather Island in Prince William Sound. After a long period of stability, the glacier began retreating in 1980 and currently sits more than 15 miles from its previous extent. This video explores the glacier’s evolution through false-color satellite imagery, which allows researchers to distinguish the glacier from sea ice, open water, exposed rocks, and nearby vegetation. Though rapid overall, the glacier’s retreat takes place in fits and starts, due to a combination of influences including climate change, sea and ice interactions, and the effects of local topography. (Video and image credit: NASA Earth Observatory)

False-color animation showing the retreat of Alaska’s Columbia Glacier since 1980. 
Dissolving Pinnacles
Limestone and other water-soluble rocks sometimes form sharp stone pinnacles like the ones seen here in Borneo. Scientists have recreated these structures in the laboratory simply by immersing water-soluble substances (essentially blocks of candy) into water. Without any background flow, the blocks will slowly form these pinnacle forests as material dissolves into the nearby water, creating a heavy solute-rich fluid that sinks down the exterior of the block. The convection generated by this dissolution drives the material into these sharp shapes, as shown mathematically in this recent study. (Image credit: N. Naim; research credit: J. Huang and N. Moore; via APS Physics)