If you’ve drizzled viscous liquids like honey or syrup, you’ve no doubt witnessed their ability to coil. Combine that coiling with a moving platform and you form a system known as the fluid dynamical sewing machine, which creates different consistent patterns of loops and curves depending on the speed at which the liquid falls and the velocity of the moving platform. The predictability of these patterns makes them especially useful for 3D printing. Previously a group at MIT developed a glass printer that could use the instability, and here a group from Montreal demonstrates how they can build solid coils at various scales. Their video also explores what the structural properties of such coils are after they solidify. (Image, video, and research credit: R. Passieux et al.)
Tag: viscous flow

Bees, Squid, and Oil Plumes
It’s time for another JFM/FYFD collab video! April’s video brings us a taste of spring with research on how bees carry pollen, squid-inspired robotics, and understanding the physics of underwater plumes like the one that occurred in the Deepwater Horizons spill eight years ago. Check it all out in the video below. (Image and video credit: T. Crawford and N. Sharp)

Sunset Flow
Day and night mix in this flow visualization of watercolor pigments and ferrofluid. The former, as suggested by their name, are water-based, whereas ferrofluids typically contain an oil base. This means the two fluids are immiscible. Like oil and vinegar in salad dressing, the only way to mix them is to break one into tiny droplets floating in the other. This is what happens near their boundary, where brightly-colored paint droplets float in a network of dark channels. To the right, the paint and ferrofluid have been swirled around to create viscous mixing patterns among the paint colors with occasional intrusions of thin ferrofluid fingers. (Image credit: G. Elbert)

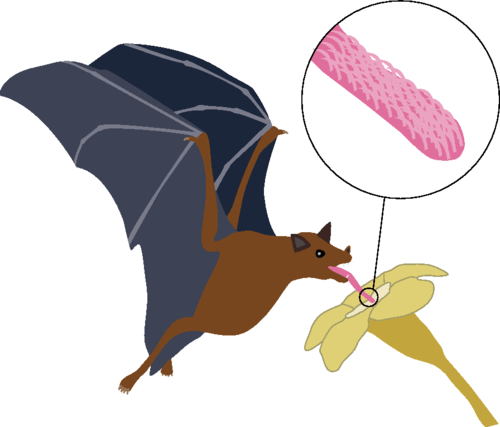
Hairy Tongues Help Bats Drink
Nectar-drinking bats, honey possums, and honeybees all use hair-like protrusions on their tongues to help them drink. In bats, these papillae have blood vessels that swell when drinking, stiffening the hairs. To investigate this drinking mechanism, researchers built their own version of a bat tongue by fabricating hairy surfaces and testing how well they trapped viscous oil when dipped and withdrawn. Through a combination of experiment and mathematical modeling, the researchers found that the optimal fluid uptake depended on the density of hairs, fluid viscosity, and the withdrawal speed. When they compared their results to actual bats, honey possums, and honeybees, they found that those animals’ tongues have hair densities very close to the predicted optimal value, suggesting that their model is capturing the important physical mechanisms that have driven evolutionary advantages for these species. (Image and research credit: A. Nasto et al.; submitted by Kam-Yung Soh)

Swimming Like a Balloon
For humans, swimming is relatively easy. Kick your legs, wheel your arms, and you’ll move forward. But for microswimmers, swimming can be more complicated. For them, the world is a viscous place, and the rules that we swim by can’t help them get around. In a highly viscous world, flows are reversible. Kick one limb down and you might move forward, but when you pull the limb up, you’ll be sucked right back to where you started. So microswimmers must use asymmetry in their swimming. In other words, their recovery stroke cannot be the mirror-image of their power stroke.
A new study suggests that simple elastic spheres could make good microswimmers through cyclic inflation and deflation. When the sphere deflates, it buckles, making a shape unlike its inflating one. This difference in shape change is enough to propel the sphere a little with each cycle. Right now the test system is a macroscale one, but the researchers hope to continue miniaturizing. (Image and research credit: A. Djellouli et al.; via APS Physics; submitted by Kam-Yung Soh)

Convection Without Heat
Glycerol is a sweet, highly viscous fluid that’s very good at absorbing moisture from the ambient air. That’s why a drop of pure glycerol in laboratory conditions quickly develops convection cells – even when upside-down, as shown above. This is not the picture of Bénard-Marangoni convection we’re used to. There’s no temperature or density change involved; in fact, there’s no buoyancy involved at all! This convection is driven entirely by surface tension. As glycerol at the surface absorbs moisture, its surface tension decreases. This generates flow from the center of a cell toward its exterior, where the surface tension is higher. Conservation of mass, also known as continuity, requires that fresh, undiluted glycerol get pulled up in the wake of this flow. It, too, absorbs moisture and the process continues. (Image credit: S. Shin et al., pdf)

When Fire Ants are a Fluid
Substances don’t have to be a liquid or a gas to behave like a fluid. Swarms of fire ants display viscoelastic properties, meaning they can act like both a liquid and a solid. Like a spring, a ball of fire ants is elastic, bouncing back after being squished (top image). But the group can also act like a viscous liquid. A ball of ants can flow and diffuse outward (middle image). The ants are excellent at linking with one another, which allows them to survive floods by forming rafts and to escape containers by building towers.
Researchers found the key characteristic is that ants will only maintain links with nearby ants as long as they themselves experience no more than 3 times their own weight in load. In practice, the ants can easily withstand 100 times that load without injury, but that lower threshold describes the transition point between ants as a solid and ants as a fluid. If an ant in a structure is loaded with more force, she’ll let go of her neighbors and start moving around.
When they’re linked, the fire ants are close enough together to be water-repellent. Even if an ant raft gets submerged (bottom image), the space between ants is small enough that water can’t get in and the air around them can’t get out. This coats the submerged ants in their own little bubble, which the ants use to breathe while they float out a flood. For more, check out the video below and the full (fun and readable!) research paper linked in the credits. (Video and image credits: Vox/Georgia Tech; research credit: S. Phonekeo et al., pdf; submitted by Joyce S., Rebecca S., and possibly others)
ETA: Updated after senoritafish rightfully pointed out that worker ants are females, not males.

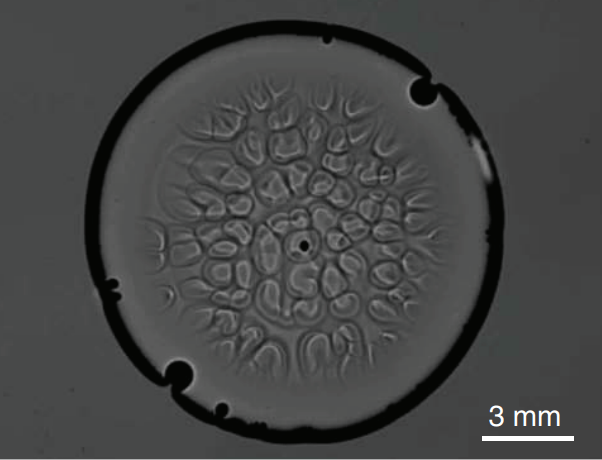
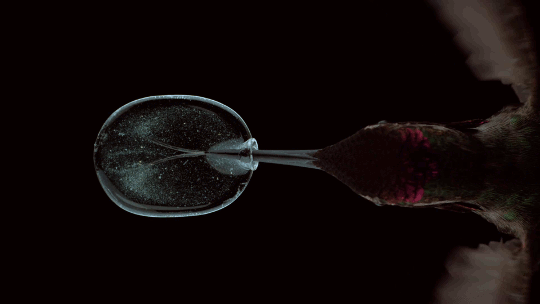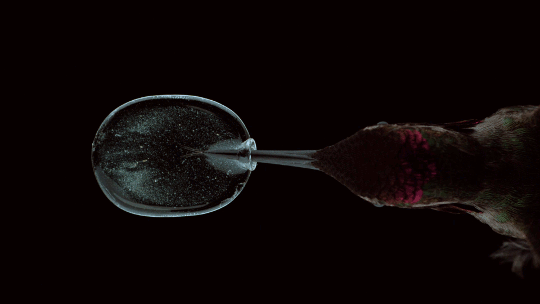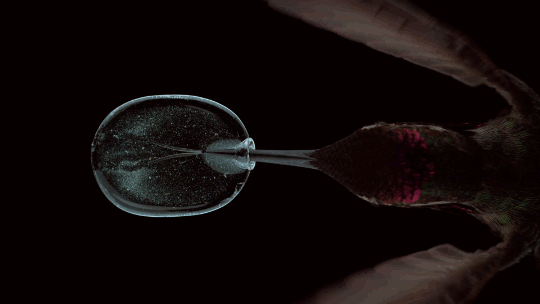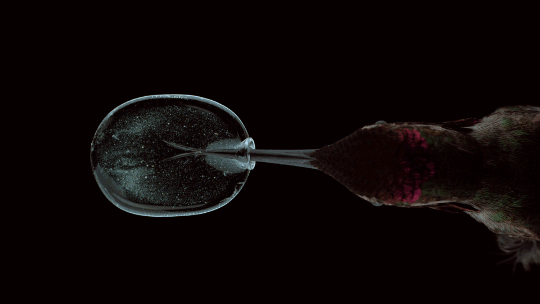
How Hummingbirds Drink
Hummingbirds are incredible acrobatic fliers, capable of hovering for more than 30 seconds at a time, even in windy conditions. Their feeding habits are equally impressive. Many species of hummingbirds have a forked tongue, each half of which curls over like a partial straw. As the bird extends its tongue, its beak compresses the space inside the tongue’s curls. Once in the nectar, both halves of the tongue re-expand, pulling liquid in along the full length of the tongue. For the birds, this is a much faster technique than simply sucking the nectar up like a straw. Hummingbirds can lick nectar more than ten times a second this way. For more gorgeous imagery of hummingbirds, be sure to check out National Geographic’s full feature. (Image credit: A. Varma, source; via Aarthi S.)

Shark Tooth Instability
Imagine that you partially fill a horizontal cylinder with a viscous fluid, like corn syrup or honey. If that cylinder is still, the fluid will simply pool along the bottom. On the opposite extreme, if you spin it very fast, that cylinder will become coated in an even layer of fluid that rotates along with the cylinder thanks to centrifugal force. Between those two extremes in rotational velocity, some interesting fluid behaviors occur. Start spinning the cylinder and some of the pooled fluid will be pulled up the sides, eventually forming a thicker film with a straight front along the bottom of the cylinder. Spin faster and that straight front starts to break down, forming sharper cusp-like waves known as shark teeth. (Image credit: S. Morris et al., source; research credit: S. Thoroddsen and L. Mahadevan)

Daily Fluids, Part 3
A lot of the fluid dynamics in our daily lives centers around the preparation and consumption of food. (And in its digestion afterward, but that’s another story!) Here are a few examples of fluid dynamics you might not have realized you’re an expert on:

Low Reynolds Number Flows
This is a fancy way of discussing the motion of syrup, honey, and other thick and viscous fluids we interact with in our lives. These flows are typically slow moving and exhibit some neat properties like coiling or being possible to unstir.
Immiscible Fluids
Oil and water don’t mix, a fact anyone familiar with salad dressings or marinades is well aware of. The way around this is to shake them up! This disperses droplets of the oil within the water (or vinegar or whatever) to create an emulsion. While not truly mixed, it does make for more pleasant eating.
Multiphase Flows
Multiphase flows are ones containing both liquid and gaseous states. Boiling is an example we often see in our daily lives, though carbonated beverages, water sprayers, and sneezes are other common ones.
Leidenfrost Effect
The Leidenfrost effect occurs when liquid is introduced to a surface that is much, much hotter than its boiling point. Part of the liquid instantly vaporizes, leaving droplets to skitter around on a thin vapor layer. This is most often seen around the stove and in skillets. (And, yes, it does qualify as a multiphase flow!)Tune in all week for more examples of fluid dynamics in daily life. (Image credit: S. Reckinger et al., source)
P.S. – I’m at VidCon (@vidconblr) this year! If you are, too, come say hi and get an FYFD sticker 😀