Ruslan Khasanov’s “Space Iris” explores the similarities between nebulae and eyes. Made entirely with common fluids like paint, soap, and alcohol, the film shows off the gorgeous possibilities of surface-tension- and density-driven instabilities. Marangoni flows abound! I even see some hints of solutal convection, perhaps? (Video and image credit: R. Khasanov; via Colossal)
Tag: surface tension

Getting Water Out of Your Ear
Swimming often results in water getting stuck in our ear canals. The narrow space, combined with the waxy surface, is excellent at trapping small amounts of water. If left in place, that excess fluid distorts hearing, can cause pain, and may eventually lead to an ear infection. So most people’s common response is to tilt their head sideways and shake it or jump to knock the water out. This recent study looks at just how much acceleration is needed to dislodge that water.

An acceleration of 7.8g isn’t enough to remove the water from this artificial ear canal. The team built an artificial ear based on the shape of a human’s ear canal and observed how much acceleration was needed to knock the water out. The answer? Quite a bit. As seen above, nearly 8g of acceleration was enough to distort the interface of the water in the ear canal, but it didn’t move the water out.
At higher accelerations — above 20 times the acceleration due to gravity – the air-water interface distorts enough to get the water to flow. But accelerations that large are enough to potentially damage brain tissues.

At over 24g, the acceleration is enough to dislodge the water from this artificial ear canal. But accelerations this high can cause brain damage. The problem is worse for children and babies, whose tiny ear canals necessitate even larger accelerations. For them, shaking hard enough to remove water could cause real damage. Instead, a couple drops of vinegar or alcohol in the ear will lower the surface tension and make the fluid easier to remove. (Image credit: top – J. Flavia, others – S. Kim et al.; research credit: S. Kim et al.; submitted by Sunny J.)

Liquid Lens Rupture
A blob of sunflower oil floating on soapy water forms a disk known as a liquid lens. But add some dyed ethanol and things take a turn. The lens rapidly expands and distorts as the ethanol and soapy water meet. These surface flows are driven by the imbalance of surface tension between the different liquids. The liquid lens deforms and abruptly ruptures, releasing dye and ethanol before rebounding into a stable lens again. Adding more ethanol to the lens will repeat the cycle. (Image credit: C. Kalelkar and P. Dey; research credit: D. Maity et al.)

Bending in the Stream
Nature is full of cilia, hairs, and similar flexible structures. Unsurprisingly, flows interact with these structures very differently than with smooth surfaces. Here, researchers investigate flow in a channel lined with flexible, hair-like plates. Initially, the channel is filled with oil and dark particles that help visualize the flow. Then, they pump water into the setup.
As the water intrudes, it forms an interface with the oil. That interface is powerful enough to bend individual hairs in the system. When the hair bends far enough, it can touch its neighbor, sealing the oil inside the gap between them. Along the length of the channel, this behavior leads to trapped pockets of oil that never drain, no matter how much water flows by. (Image and research credit: C. Ushay et al.)

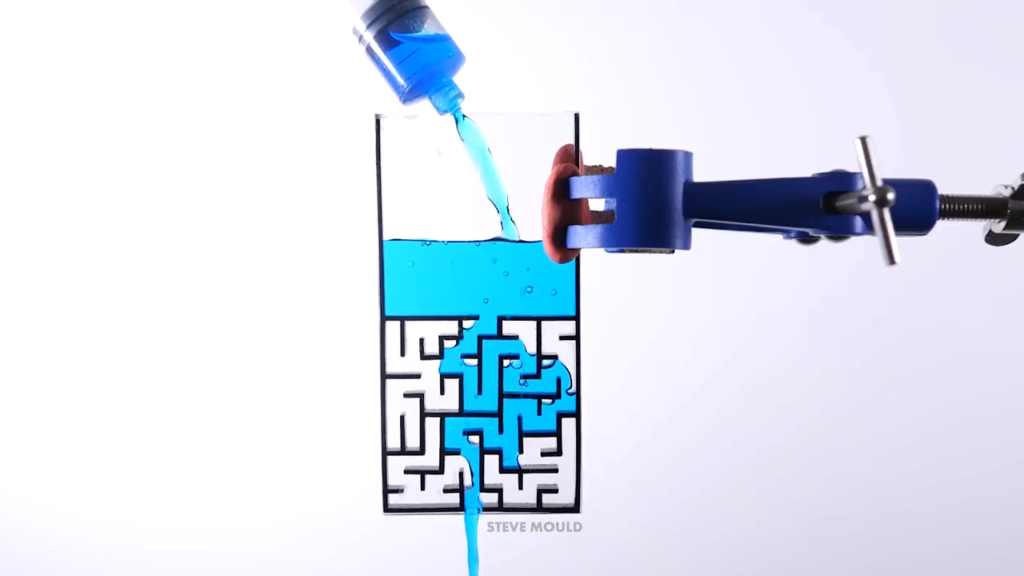
Can Water Solve a Maze?
Inspired by a simulation, Steve Mould asks a great question in this video: can water solve a maze? Yes — with some caveats. Steve makes two different maze patterns — a simple and a complex path — in two different sizes. With the small, simple-path version, the water immediately follows the correct path without taking any wrong turns. What keeps it on the right path seems to be a combination of air pressure and surface tension. In the dead-end passages, the air has nowhere to go in order to allow the water in. So the pressure of the trapped air and the narrowness of the passages (which allows surface tension to help hold the water in place) keeps the water out of the false paths.
With the larger mazes, the water is able to take some false turns as it seeks the lowest possible path. But after awhile the incorrect region fills and the water takes the next lowest path available, which eventually leads it to the outlet.
Toward the end of the video, Steve notes that the large mazes sometimes stop flowing, even though water is still in the reservoir. I’ll quibble slightly here with his explanation, though; I don’t think surface tension is playing as much of a role in this stoppage as friction. The water is basically being driven through a long, narrow pipe, which means quite a lot of friction between it and the walls. Just as you need a certain driving pressure to keep water in a pipe flowing, the maze needs a high enough driving pressure to keep the water going. The point at which drainage stops is the point where the upstream pressure (caused by the depth of the reservoir above the maze) is equal to the pressure lost due to friction in the pipe. All in all, it’s a very cool experiment and a video well-worth watching! (Video and image credit: S. Mould)

Long-Lived Bubbles
Without surfactants to stabilize them, bubbles don’t last long at room temperature. But adding a little heat changes the picture. When heated, the bubbles get stabilized by a thermal gradient that lifts fluid toward the bubble’s peak, where it cools and gathers. Eventually, the cold fluid grows heavy enough to sink down the side of the bubble (in either a constant stream or occasional drips); with warm fluid getting pulled up to replace it (via the Marangoni effect), the process repeats and the bubble lives on. (Video credit: S. Nath et al.; see also)

Mermaid Cereal
In the Cheerios effect, floating objects can fall into one another due to capillary attraction — just like Cheerios link up in a cereal bowl. Here researchers play with that effect by adding repulsive magnets to their “cereal” pieces. They find that their so-called mermaid cereal falls into preferential spacing, with pieces pairing up but never touching. Adding lots of these pieces in a confined space creates interesting crystalline and striped patterns, as seen later in the video. (Video credit: A. Hooshanginejad et al.)

Pee-Flinging Sharpshooters
The tiny glassy-winged sharpshooter feeds exclusively on nutrient-poor sap from plant xylem. Since the sap is 95% water, the insects have to consume massive amounts, necessitating lots of urination — up to 300 times their body weight each day! With so much urine to get rid of and so little energy to spare, the sharpshooter has developed an ingenious, low-energy method to expel its waste. The insect forms a droplet on its anal stylus and flings it. A recent study reveals just how clever the insect’s method is.
Researchers found that sharpshooters fling their droplets 40% faster than their stylus moves. This superpropulsion is only possible because the stylus’s motion is finely tuned to the droplet’s elasticity. Essentially, the insects achieve single-shot resonance with every throw. The energy-savings for the insects is substantial; researchers estimate that making a jet of urine instead would cost four to eight more times energy. (Video credit: Georgia Tech; image and research credit: E. Challita et al.; via Ars Technica; submitted by Kam-Yung Soh)

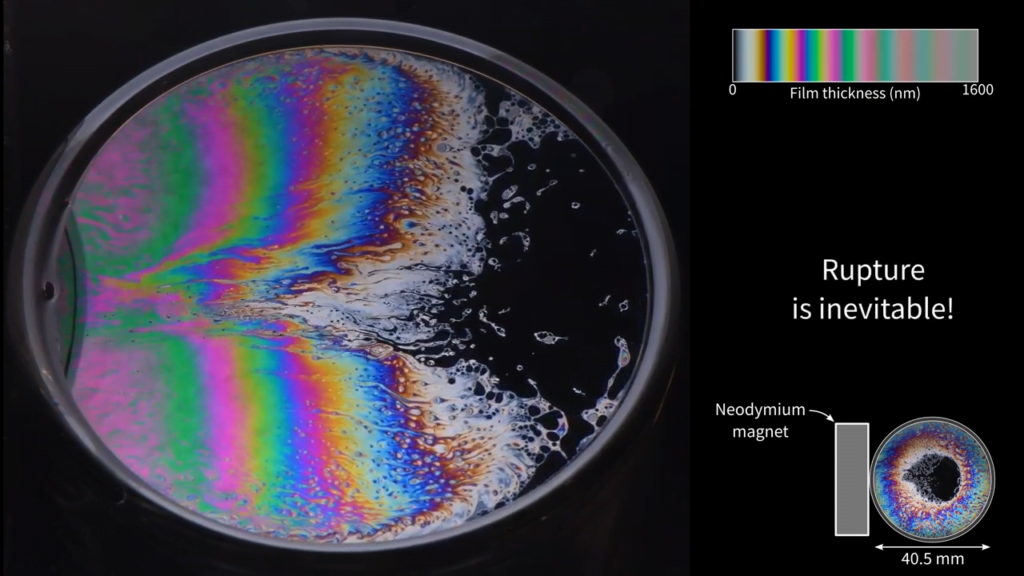
Magnetic Soap Films
Soap films naturally thin over time as fluid evaporates and differences in film thickness cause surface-tension-driven flows. In this video, researchers experiment with adding magnetic nanoparticles to the soap film. In the second image, the white structures near the center of the film contain nanoparticles, and they’re drawn toward the magnet that sits (out-of-frame) to the left of the film. With more nanoparticles and a stronger magnetic field (Image 3), the entire soap film takes on a distinctive profile that thins from left to right. The effect is so strong that the film quickly thins to the point of rupture. (Image and video credit: N. Lalli et al.)

Surfactants and Waves
In the ocean, waves often curl over and trap air, becoming plunging breakers. How do surfactants like soap or oil affect this process? That’s the question behind this video, where researchers visualize breaking waves with differing amounts of added surfactant. In the case of pure water, the wave forms a smooth jet that curls over and traps air when the wave breaks. As more and more surfactant gets added, the shape of that jet and cavity change. In one case, they become irregular. In another, they disappear entirely, and with the most surfactant added, the wave suddenly looks just like the water-only case.
The key to these behaviors, it turns out, is not how much surfactant there is, but how much the concentration of surfactant varies along the length of the wave. When there are significant changes in the surfactant concentration along the wave, local Marangoni flows try to even out the surface tension, causing the wave to break up in an irregular fashion. (Image and video credit: M. Erinin et al.)