The Winter Olympics are underway in Sochi, Russia, and here at FYFD, I am busy preparing a special series of posts on fluid dynamics in the Winter Games. Look for the first of those starting on Monday. In the meantime, you can check out some of FYFD’s previous themed series now compiled into a special archive. (Photo credit: B. Armangue)
Tag: science

Convective Impressionism
Buoyant convection, driven by temperature-dependent changes in density, is a major force here on Earth. It’s responsible for mixing in the oceans, governs the shape of flames, and drives weather patterns. The images above show flow patterns caused by buoyant convection. The colors come from liquid crystal beads immersed in the fluid; red indicates cooler fluid and blue indicates warmer fluid. You can see plumes of warmer fluid rising in some of the photos. At the same time, though, the images are beautiful simply as art and are strongly reminiscent of works by Vincent van Gogh. (Image credit: J. Zhang et al.)

The Reynolds Experiment
One of the most famous and enduring of all fluid dynamics experiments is Osborne Reynolds’ pipe flow experiment, first published in 1883 and recreated in the video above. At the time, it was understood that flows could be laminar or turbulent, though Reynolds’ terminology of direct or sinuous is somewhat more poetic:
Again, the internal motion of water assumes one or other of two broadly distinguishable forms-either the elements of the fluid follow one another along lines of motion which lead in the most direct manner to their destination, or they eddy about in sinuous paths the most indirect possible. #
There had, however, been no direct evidence of these eddies in a pipe. Reynolds built an apparatus that allowed him to control the velocity of flow through a clear pipe and simultaneously introduce a line of dye into the flow. He carefully varied the velocity and temperature (and thus viscosity) in his apparatus and not only documented both laminar and turbulent flow but found that the transition from one to another could be described by a dimensionless number he derived from the Navier-Stokes equation. This number was dependent on the fluid’s velocity and kinematic viscosity as well as the diameter of the pipe. This was the birth of the Reynolds number, one of the most important parameters in all of fluid dynamics. (Video credit: S. dos Santos; research credit: O. Reynolds)

Wind and Waves Visualized

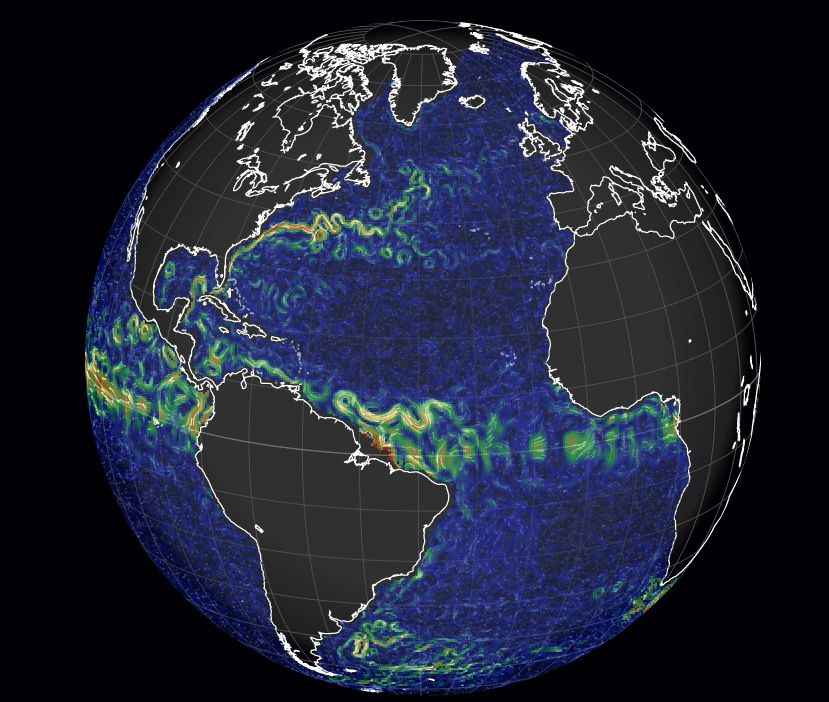
Much like the wind map we featured previously, designer Cameron Beccario’s visualizations of wind and ocean surface current data draw from near-real-time sources to create a stunning picture of fluid dynamics on a planetary scale. The number of options in terms of projections and data are really quite incredible, and you’ll want to play around to get a real sense for it. Want to see the wind and total precipitable water at 1000 hPa? Here you go. Maybe you prefer studying Pacific ocean currents. All the data are there to play with. People often wonder why weather forecasts aren’t always right, but, when you look at the scale and complexity of these flows, it’s almost a wonder that we can predict them at all. (Image credits:C. Beccario/earth; via skunkbear and io9)

Protostellar Jets
As young stars form, they often produce narrow high-speed jets from their poles. By astronomical standards, these fountains are dense, narrowly collimated, and quickly changing. The jets have been measured at velocities greater than 200 km/s and Mach numbers as high as 20. The animation above (which you should watch in its full and glorious resolution here) is a numerical simulation of a protostellar jet. Every few decades the source star releases a new pulse, which expands, cools, and becomes unstable as it travels away from the star. Models like these, combined with observations from telescopes like Hubble, help astronomers unravel how and why these jets form. (Image credit: J. Stone and M. Norman)
ETA: As it happens, the APOD today is also about protostellar jets, so check that out for an image of the real thing. Thanks, jshoer!

The Structure of Turbulence
Though they may appear random at first glance, turbulent flows do possess structure. The video above shows a numerical simulation of a mixing layer, a flow in which two adjacent regions of fluid move with different velocities. The upper third of the frame shows a top view, and the bottom frame shows a side view, in which the upper fluid layer moves faster than the lower one. The difference in velocities creates shear which quickly drives the mixing layer into turbulence. But watch the chaos carefully, and your eye will pick out vortices rolling clockwise in the largest scales of the mixing layer. These features are known as coherent structures, and they are key to current efforts to understand and model turbulent flows. (Video credit: A. McMullan)

Bubble Vortices
Vortices appear in scales both large and small, from your shower and the flap of an insect’s wing to cyclones and massive storms on other planets. Especially with these large-scale vortices, it can be difficult to understand the factors that affect their trajectories and intensities over time. Here researchers have studied the vortices produced on a heated half bubble for clues as to their long-term behavior. Heating the base of the bubble creates large thermal plumes which rise and generate large vortices, like the one seen above, on the bubble’s surface. Researchers observed the behavior of the vortices with and without rotation of the bubble. They found that rotating bubbles favored vortices near the polar latitudes of the bubble, just as planets like the Earth and Saturn have long-lived polar vortices. They also found that the intensification of both bubble vortices and hurricanes was reasonably captured by a single time constant, which may lead to better predictions of storm behaviors. Their latest paper is freely available here. (Image credit: H. Kellay et al.; research credit: T. Meuel et al.; via io9)

Hydraulic Jump in the Lab
When fast-moving liquids encounter regions of slow-moving liquids, they decelerate rapidly, trading their kinetic energy for potential energy and creating a hydraulic jump. Flow in the video above is from left to right. The depth difference between the incoming and outgoing water can be directly related to the velocity of the incoming fluid. Hydraulic jumps in rivers and spillways are often extremely turbulent, like the one in this video, but laminar examples exist as well. In fact, with the right height and flow rate, you can create stable hydraulic jumps right in your kitchen sink. The hydraulic jumps formed from a falling jet are typically circular, but with the right conditions, all sorts of wild shapes can be observed. (Video credit: H. Chanson)

Vibrations from Vortices
Vortex shedding frequently happens in the wakes of non-streamlined bodies as a result of flow around the obstacle. Newton’s third law states that forces come in equal and opposite pairs, meaning that the vortex shedding behind an obstacle is accompanied by a force on the obstacle. For a fixed cylinder, this is not always apparent, but for a pendulum, like the ones demonstrated in this video, this vortex-induced vibration causes significant motion. This same effect can make traffic lights and industrial chimneys sway. You’ve likely experienced it yourself as well, if while swimming you’ve ever spread your fingers underwater and spun in place. Try it sometime with your arm out and you’ll feel the vortices make your arm vibrate up and down as you spin. (Video credit: Harvard Natural Sciences Lecture Demonstrations)

Snow Rollers
Snow rollers are nature’s snowballs, formed when high winds roll a chunk of snow along the surface, allowing it to accumulate more and more material. They occur relatively rarely because their appearance is the culmination of several specific meteorological factors. To form rollers, the ground needs to be icy, with a layer of loose, wet snow above the ice. And, of course, it needs to be windy enough to move the snow without being so windy that snow breaks up. In the photos above, the snow roller got too large for the wind to continue moving it, but the wind didn’t stop blowing. Instead, the snow roller became an obstacle to the flow and a horseshoe vortex formed at its base. The spinning of the vortex dug out the trench in front of and along the sides of the snow roller. This same effect is often seen on the windward side of trees in winter. (Photo credit and submission: S. Benton)