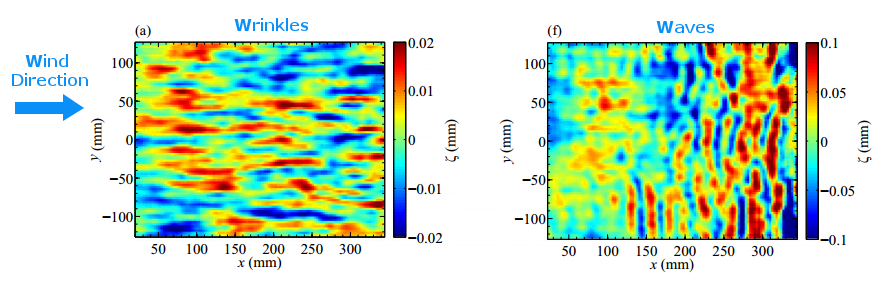
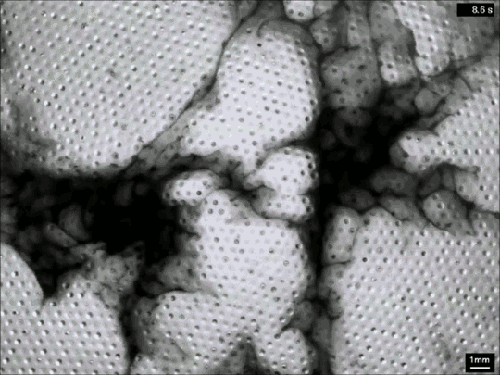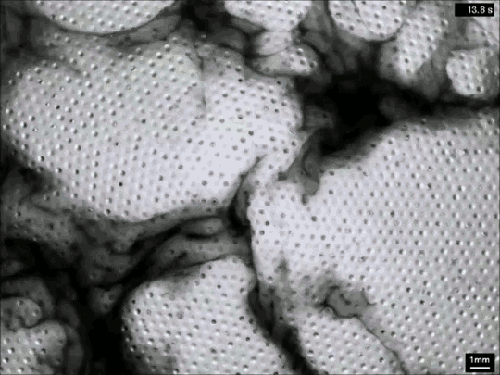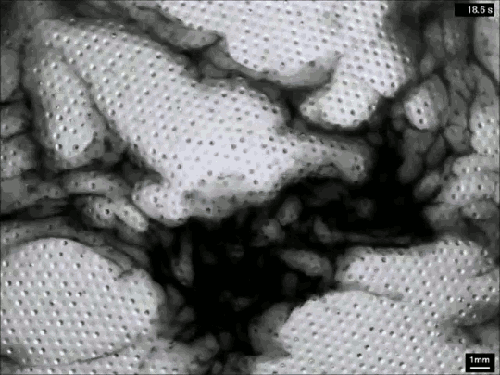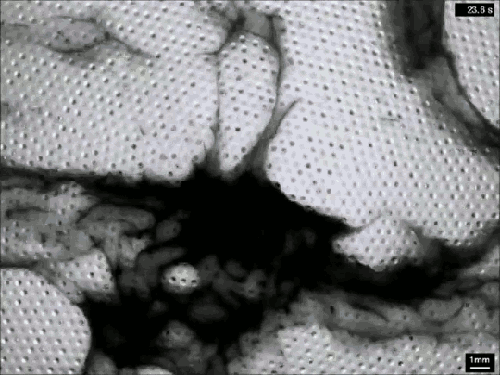
Extensive rains in California have brought an unusual sight to Lake Berryessa – an overflowing spillway. The upper photo, taken in 2010, shows the concrete structure of the spillway’s entrance, known as a bellmouth – or, in the words of locals, a glory hole. When the water level rises above the concrete, water begins to cascade down the spillway to relieve flooding.
The flow is rather mesmerizing and beautifully laminar until it’s fallen many feet down the hole. This is intentional on the part of the designers – at least the laminar part. It means that the flow velocity at the entrance is slow, so that animals (or trespassing people) nearby are not going to get sucked down the spillway a la Charybdis. Nevertheless, the spillway does make quick work of excess water. The New York Times reported that on February 21st about two million gallons (7.5 million liters) of water a minute flowed down the spillway. (Image credits: J. Brooks; T. Van Hoosear; video credit: Lake Berryessa News; submitted by Zach B.)