Photographer Eric Gross captured these surreal alpine landscapes in Colorado’s Rocky Mountains. Although the lake’s surface appears to have frozen waves, the prevailing theory is that these mounds and divots occur when snowdrifts form atop the lake, melt and refreeze. Over multiple melting and freezing cycles, the lake builds up with what appear to be wind-driven waves frozen in time. (Image credit: E. Gross; via Colossal)
Tag: melting

Ice Patterns
Periods of freezing and thawing can leave complicated patterns in ice, as seen in this aerial photo of Binnewater Lake in New York. Ice rarely forms evenly on large bodies like this, so there are always underlying weaknesses. A hard freeze may have caused the ice to contract, forming the initial radial pattern. Then warmer periods of melting allowed water to rise into the cracks and expand them. As the process repeats, the visible pattern emerges.
Also note the star-like crack patterns near the shore. These may have formed in spots where something like a stick protruding from the water’s surface allowed warmer water up onto the ice to melt the snow sitting atop it. (Image credit: D. Spitzer; via EPOD; submitted by Kam-Yung Soh)

Ice Labyrinths
Pattern formation is extremely common in nature, from the dendritic growth of trees and snowflakes to the stripes of a tiger. A new paper describes how a thin layer of ice in a liquid can form labyrinthine patterns when illuminated with near-infrared light. Both the liquid and ice are maintained at a constant temperature below the melting point, but the ice absorbs the near-infrared light more effectively than the water. This means that parts of the ice that are far from the liquid warm and melt faster, creating holes that can then allow a pocket of liquid to seep in and reduce the absorption rate. The ice crystals themselves thin and expand across the surface at the expense of more holes, which eventually create larger channels that pock the ice. (Image and research credit: S. Preis et al.; via Nature; submitted by Kam-Yung Soh)

Melting
File this one under “Oddly Satisfying” – this timelapse video shows the process of melting a jawbreaker candy using a blowtorch. Over a minute and a half, each colorful layer of candy melts away to reveal the strata beneath. There’s a definite connection here to some of the previous research we’ve discussed on erosion, dissolution, and melting. The blowtorch’s flame generates a hot boundary layer around the candy surface; it’s thickest and hottest at the central stagnation point, but judging by the melting layer we see running all the way to the candy’s shoulder, its size and effect are substantial even there. It’s hard to tell from the video whether the surface of candy is getting roughened (a la scalloping) or whether that’s just an uneven layer of melted candy flow. Regardless, it’s a fun watch. (Video and image credit: Let’s Melt This; via Colossal)


Enormous Ice Disk
We’ve seen spinning ice disks before, but this month Westbrook, Maine has developed the largest one I’ve ever seen. A research paper from 2016 indicates that this seemingly alien formation spins due to an oddity of water. Water is at its densest around 4 degrees Celsius, so as the ice of the disk melts in the warmer waters of the river, it sinks. That downward plume sets up a vortex in the water beneath the disk. And as the water spins, it drags the ice with it, causing the disk’s rotation. The warmer the water is, the faster the disk spins. (Image credit: T. Radel/City of Westbrook; research credit: S. Dorbolo et al.; via Gizmodo; submitted by jpshoer)

Dissolving Candy
In nature, solid surfaces often evolve over time in conjunction with the flows around them. This is how stalactites, canyons, and hoodoos all form and change over time. Here researchers examine a surface formed from hard candy that is dissolving from below. Over time, the initially flat surface develops a pitted appearance (top image, scale bar is 1 cm) with roughness that is approximately 1 mm in scale. Flow visualization (bottom row) suggests that these pits result from local flow where narrow, millimeter-sized dense plumes fall away from the surface.
As material dissolves from the candy, it forms a dense layer of sugar-water mixture near the solid surface. Once that layer grows to a critical thickness, it will be too unstable for viscosity to counter. At that point, the Rayleigh-Taylor instability takes over, causing the dense sugar-water layer to break up into narrow, sinking plumes. Although each area is evolving independently, the rate at which material dissolves is uniform everywhere, so the dissolving body retains the same shape over time. (Image and research credit: M. Davies Wykes et al., source)

Snowmelt
Much of the rain that falls on Earth began as snow high in the atmosphere. As it falls through warmer layers of air, the snowflakes melt and form water droplets. The details of this melting process have been difficult to capture experimentally, but a new computational model may provide insight. The basic process has a couple stages. As snow begins to melt, surface tension draws the water into concave areas nearby. When those regions fill up, the water flows out and merges with neighboring liquid, forming water droplets around a melting ice core.
Although this same sequence was observed for many types of snow, scientists also observed some important differences between rimed and unrimed snowflakes. Rime forms when supercooled water droplets freeze onto the surface of a snowflake. Lightly rimed snow still looks light and fluffy, like the animation above, but heavily rimed snow forms denser and more spherical chunks. Because there are lots of porous gaps in heavily rimed snow, water tends to gather there during initial melting. Rimed snow was also more likely to form one large water droplet rather than breaking into multiple droplets like snow with less rime. For more, check out NASA’s video and the Bad Astronomy write-up. (Image credit: NASA, source; research credit: J. Leinonen and A. von Lerber; via Bad Astronomy; submitted by Kam Yung-Soh)

Scallops and Erosion
Although we often think of solids as immovable in the face of flow, the motion of air and water sculpts many parts of our world. One common pattern, seen both on surfaces that melt and those that dissolve into a flow, is called scalloping. Mathematical analysis shows that flat surfaces exposed to a flow that melts or dissolves them unavoidably develop these scallops. The surface becomes rougher as the scallops form, but the instability that drives them only works up to a specific level of roughness. Instead of the scallops becoming deeper and deeper, the flow shifts as the surface changes. Peaks in the surface erode faster than the valleys, which tends to keep the scallops relatively uniform in depth after they’ve formed. Scallops like these are often seen in soluble rocks like limestone or marble as well as in snow and ice. (Image credit: Seattle Times, G. Smith; research credit: P. Claudin et al., L. Ristroph)

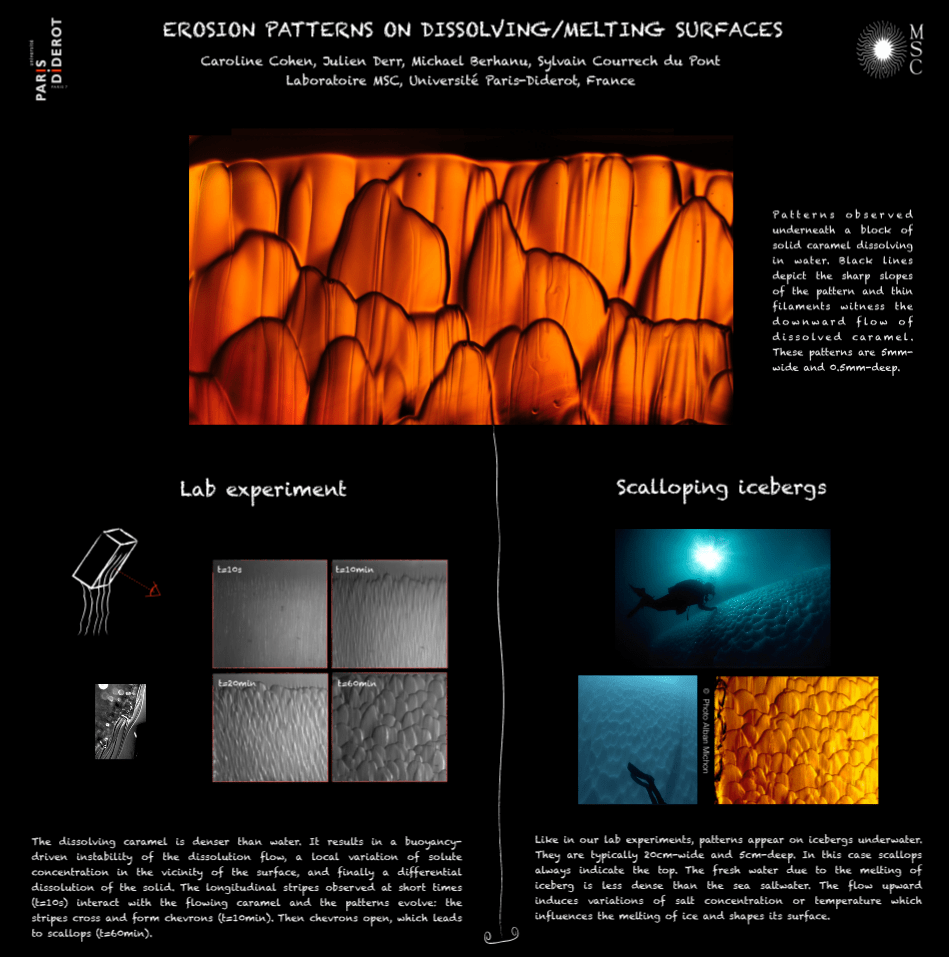
Icebergs and Caramel
What do icebergs and caramel have in common? Both have similar scalloped erosion patterns as they dissolve. When caramel dissolves in water, the denser caramel sinks in the buoyant water. An initially smooth surface will first form lines, then the flowing caramel and the uneven surface interact, forming chevrons, followed by larger scallops. A similar process happens with melting icebergs. The meltwater from an iceberg is less dense than the surrounding seawater, so it will rise as it melts. This causes variations in the salt concentration and temperature near the iceberg, which cause it to melt differently in different spots, ultimately leading to the same scallop shapes observed in the caramel. Check out the full-size PDF of the poster here. (Image credit: C. Cohen et al.)