Expedition guide and photographer Ryan Newburn captures the ephemeral beauty of the glacial caves he explores in Iceland. These caves are in constant flux, thanks to the run and melt of water. The scalloped walls are a sign of this process of melting and dissolution. The icicles, too, hint at ongoing melting and refreezing. Caves can appear and disappear rapidly; they’re a dangerous environ, but Newburn freezes them in time, letting the rest of us experience a piece of their majesty. See more of his images on his Instagram. (Image credit: R. Newburn; via Colossal)
Tag: melting

“Eternal Spring”
With every spring comes the thaw. Warming temperatures melt winter’s ice, carving it away to reveal the surfaces beneath. Christopher Dormoy’s macroscale timelapse “Eternal Spring” captures this dynamic, showing the process drop-by-drop and rivulet-by-rivulet. It’s also a commentary on melting in general as human-driven climate change chips away at ice that formed over millennia. (Video and image credit: C. Dormoy)

Summer Melt
A warm summer in 2022 has resulted in record melting on Svalbard. Located halfway between the Norwegian mainland and the North Pole, more than half of Svalbard is normally covered in ice. But with glaciers in retreat and firn — a surface layer of compressed porous snow — melting, pale blue ice is getting direct exposure to the sun and warm air temperatures. The result has been melting 3.5 times larger than the average melt between 1981 and 2010. Look closely and you’ll find deep blue meltwater ponds dotting the ice, too. The run-off of meltwater has likely carried extra sediment into the surrounding waters, accounting for some of the paler water colors along the coast. (Image credit: J. Stevens/USGS; via NASA Earth Observatory)

Leidenfrost On Ice
We’ve seen many forms of Leidenfrost effect — that wild, near-frictionless glide that liquid droplets make on a very hot surface — over the years, but here’s a new one: the three-phase Leidenfrost effect. Researchers found that they could generate a Leidenfrost effect using an ice disk placed on an extremely hot surface. During the effect, the ice and its melting layer of water glide on vapor, hence the name.
The team found that getting a three-phase Leidenfrost effect requires a much, much higher temperature than the regular Leidenfrost effect. Water will get its glide on at 150 degrees Celsius. Getting ice to glide on the same surface required a stunning 550 degrees Celsius! Why the big difference? The challenge is that water layer, which, by definition, has a 100-degree difference between its boiling side and its frozen boundary. It takes so much heat to maintain that layer that there’s little energy left over for evaporation; that’s why it takes so much more energy to get the three-phase Leidenfrost effect. (Image and research credit: M. Edalatpour et al.; via Ars Technica; submitted by Kam-Yung Soh)

Mushy Layers
In many geophysical and metallurgical processes, there is a stage with a porous layer of liquid-infused solid known as a mushy layer. Such layers form in sea ice, in cooling metals, and even in the depths of our mantle. Within the mushy layer, temperature, density, and concentration can vary dramatically from one location to another.
The image above shows a mushy layer made from a mixture of water and ammonium chloride. Above the mushy layer, green plumes drift upward, carrying lighter fluid. Look closely within the mushy layer and you’ll see narrow channels feeding up to the surface. These are known as chimneys. In sea ice, chimneys like these carry salty brine out of the ice and into the seawater, increasing its salinity. See this Physics Today article for more details on the dynamics of mushy layers. (Image credit: J. Kyselica; via Physics Today)

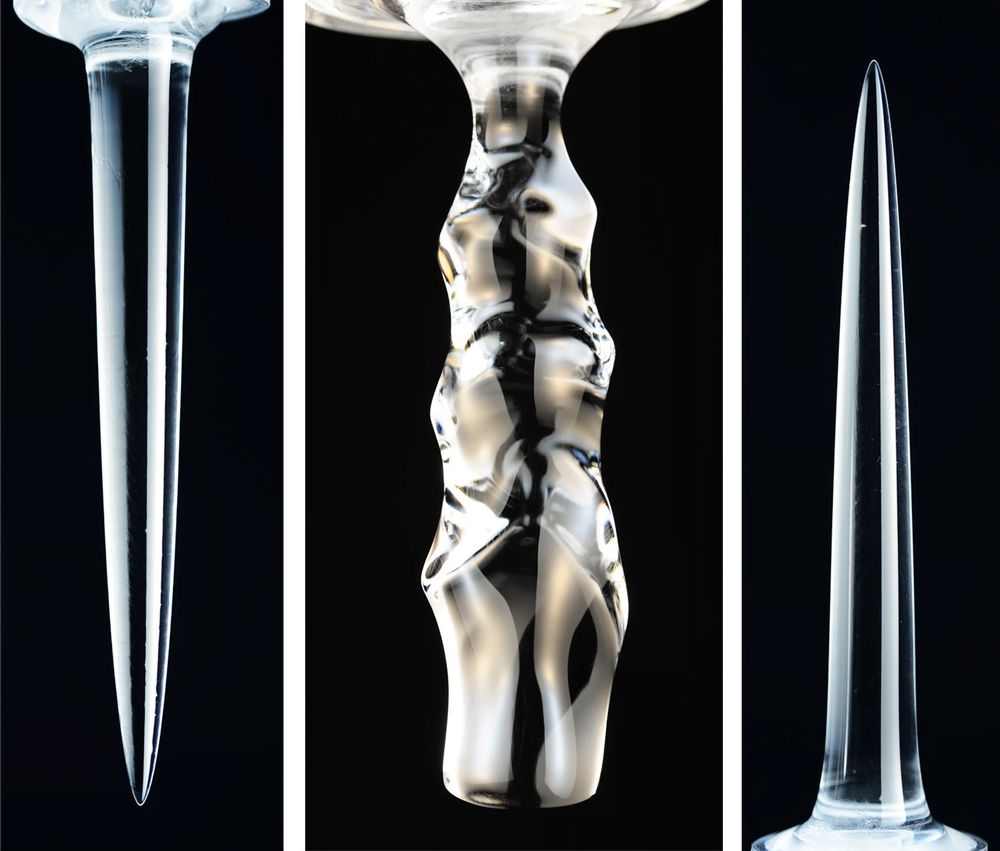
The Shapes of Melting Ice
Water is an odd substance because it is densest at 4 degrees Celsius, well above its melting point at 0 degrees Celsius. This density anomaly means that melting ice takes on very different shapes, depending on the temperature of the water surrounding it. At low temperatures (under 4 degrees Celsius), the cold water melting off the ice is denser than the surroundings, so it sinks. The sinking fluid melts lower portions of the ice faster, leading to an inverted pinnacle (Image 1).
In contrast, at higher temperatures (above 7 degrees Celsius), the meltwater is lighter than the surroundings and therefore rises, creating an upward-pointing pinnacle (Image 3). At intermediate temperatures, some areas of the ice see rising meltwater and some see sinking. This complicated flow pattern sets up vortices that result in a scalloped edge along the ice (Image 2). (Image and research credit: S. Weady et al.; via APS Physics)

Antarctic Meltwaters
Cerulean blue meltwater glints in this satellite image of the George VI Ice Shelf. Wedged between the Antarctic Peninsula on the right and Alexander Island on the left, the ice shelf itself floats on the ocean. When ice shelves collapse, they do not directly raise sea levels since their weight has already displaced water; but a collapsed ice shelf lets glaciers flow and break up faster, thereby raising water levels.
In past ice shelf collapses, scientists have noted major buildup and sudden drainage of surface lakes like the ones seen here. Meltwater penetrating through snow and ice can destabilize the shelf and hasten collapse, but the exact mechanisms are hard to track. This Physics Today article summarizes our understanding of the process and some of the methods scientists use to study it. (Image credit: L. Dauphin/NASA Earth Observatory; see also Physics Today)

Iceberg Melting Depends on Shape
Not all icebergs melt equally. Through a combination of experiment and numerical simulation, researchers have shown that an iceberg’s shape underwater strongly affects how it melts. Specifically, icebergs in a flow melt more quickly on the front and side surfaces and slower on the underside. This means that narrow icebergs that project deep into the water will melt faster than wider, shallow ones. Currently, climate models don’t account for this variation, but the researchers hope their work will help build more accurate models for future studies. (Image credit: iceberg – C. Matias, experiment – E. Hester et al.; research credit: E. Hester et al.; see also APS Physics)


Albedo Effect
Temperature isn’t the only factor that determines how ice will melt. In this photo, a dark oak leaf absorbed more solar radiation than the reflective ice around it, causing the ice beneath it to melt. Scientifically, this effect is described by albedo; darker, more absorptive surfaces like the leaf have a lower albedo, whereas light, reflective ice and snow have a high albedo and can better resist melting on sunny days. (Image credit: K. James; submitted by Kam-Yung Soh)

Eroding Ice
When glaciers form, they do so in layers, with clear blue ice sandwiched between sediment and air-bubble-filled white ice. Because each of these layers absorbs sunlight differently, they don’t melt evenly. The spikes and ridges seen in this ice formed because of this differential melting between layers. The blue ice is particularly good at absorbing visible wavelengths of light, and so erodes more easily than the other layers.
Although the results look somewhat similar to the penitente ice seen at high altitudes, the formation mechanisms are a little different. Penitentes rely heavily on sublimation — where their ice and snow change directly into a gas — rather than the melting seen here. That said, both eroded forms depend strongly on how different layers within them absorb and scatter sunlight. (Image credit: J. Van Gundy; via EPOD; submitted by Kam-Yung Soh)