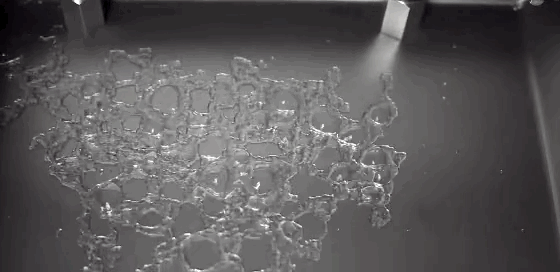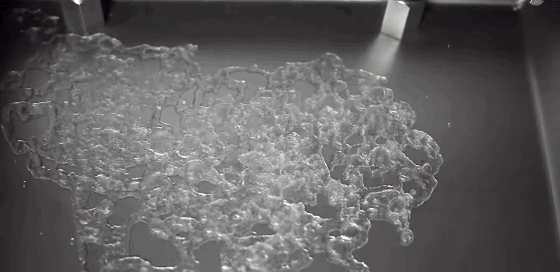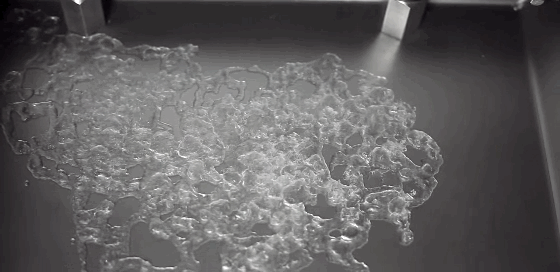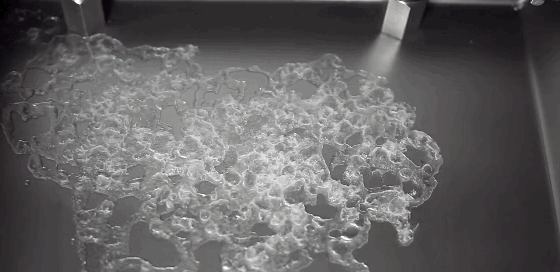A Leidenfrost droplet impregnated with hydrophilic beads hovers on a thin film of its own vapor. The Leidenfrost effect occurs when a liquid touches a solid surface much, much hotter than its boiling point. Instead of boiling entirely away, part of the liquid vaporizes and the remaining liquid survives for extended periods while the vapor layer insulates it from the hot surface. Hydrophilic beads inserted into Leidenfrost water droplets initially sink and are completely enveloped by the liquid. But, as the drop evaporates, the beads self-organize, forming a monolayer that coats the surface of the drop. The outer surface of the beads drys out, trapping the beads and causing the evaporation rate to slow because less liquid is exposed. (Photo credit: L. Maquet et al.; research paper – pdf)
Tag: evaporation

Does Liquid in a Vacuum Boil or Freeze?
What happens to a liquid in a cold vacuum? Does it boil or freeze? These animations of liquid nitrogen (LN2) in a vacuum chamber demonstrate the answer: first one, then the other! The top image shows an overview of the process. At standard conditions, liquid nitrogen has a boiling point of 77 Kelvin, about 200 degrees C below room temperature; as a result, LN2 boils at room temperature. As pressure is lowered in the vacuum chamber, LN2’s boiling point also decreases. In response, the boiling becomes more vigorous, as seen in the second row of images. This increased boiling hastens the evaporation of the nitrogen, causing the temperature of the remaining LN2 to drop, the same way sweat evaporating cools our bodies. When the temperature drops low enough, the nitrogen freezes, as seen in the third row of images. This freezing happens so quickly that the nitrogen molecules do not form a crystalline lattice. Instead they are an amorphous solid, like glass. As the residual heat of the metal surface warms the solid nitrogen, the molecules realign into a crystalline lattice, causing the snow-like flakes and transition seen in the last image. Water can also form an amorphous ice if frozen quickly enough. In fact, scientists suspect this to be the most common form of water ice in the interstellar medium. (GIF credit: scientificvisuals; original source: Chef Steps, video; h/t to freshphotons)
The Inside of an Evaporating Drop
[original media no longer available]
Evaporating droplets may not look like much to the naked eye, but they contain complicated flow patterns. The type of pattern observed depends strongly on the contact line, the place where the liquid, solid, and air meet. When the contact line is pinned–kept unchanged–during evaporation, any particulates in the drop get pulled toward the edges as the drop evaporates. This is what leaves the classic coffee ring stain. It is also what is shown in the first clip in the video above. Contrast this with the second clip, in which the contact line is unpinned and varies irregularly as the drop evaporates. In the unpinned drop, particles are drawn inward during evaporation. The flow patterns are very different as well, complicated by swirling that is the result of force imbalances caused by the irregularly receding contact line. (Video credit: H. Kim)

Mushrooms Make Their Own Breeze
Mushrooms don’t rely on a stray breeze to spread their spores; they generate their own air currents instead. The key is evaporation. The mushroom cap contains large amounts of water, and, as this water evaporates, it cools the mushroom and the air next to it. This cool air is denser than the surrounding air, and so tends to spread out and convect. At the same time, though, the water vapor that evaporated from the mushroom is less dense than nearby air, which helps it rise. This combination of spreading and rising air carries spores away from the mushroom cap and, as seen in the video above, can combine to form beautiful and complex currents that spread the spores. (Video credit: E. Dressaire et al.)

Evaporating Drops
When still drops evaporate from a surface, they do so in several phases, as illustrated in the video above. Initially, the drop forms a spherical cap. At this point the velocity within the droplet is so small that it is difficult to resolve, but particles within the drop move outward toward the contact line. As the drop evaporates, they form a circle of sediment – the familiar coffee ring. As the drop flattens, radial velocity increases, drawing more and more particles to the coffee ring. Eventually the drop pulls away from the ring, leaving surface tension and evaporation to compete in driving the internal flow. During this phase, some parts of the contact line try to re-establish the flow pattern that made the first ring; this leaves behind circular segments broken up by the increasing instabilities in the contact line. In the final stage, surface tension smooths some of the irregularities and drives an inward velocity that leaves behind radial sediment segments. (Video credit: G. Hernandez-Cruz et al.)

Self-Assembly via Evaporation
When working at the microscale, engineering structures like those used for drug delivery systems requires ingenuity. Since it isn’t possible to manipulate particles manually, researchers harness physical effects to do the work for them. Here a droplet filled with millions of polystyrene microparticles sits on a hydrophobic surface, which helps keep the drop’s spherical shape. As the drop evaporates, surface tension and internal flow in the drop help the microparticles self-assemble into a microscopic soccer-ball-like shape. (Video credit: A. Marin et al.; submission by A. Marin)

Evaporation and Surface Effects
Surface properties can have surprising effects on fluid behavior. This image shows the evaporation of several droplets over time. All of the initial droplets are of the same volume, but they are placed on a surface which is a) superhydrophobic, b) hydrophobic, or c) hydrophilic. The more hydrophobic the surface, the larger the initial contact angle between the droplet and surface and the smaller the wetted area of the surface. Yet despite this seemingly large surface area exposure to air, the droplet on the superhydrophobic surface is the slowest to evaporate. (Photo credit: C. Choi)

Tears of Wine
Wine drinkers may be familiar with the “tears of wine” often seen on the wall’s of a glass. The effect is a combination of evaporation and surface tension. As the low-surface-tension alcohol evaporates from the wine film left by swirling the glass, the higher local surface tension draws wine up the walls of the glass. Eventually enough wine gathers that droplets form and slide back down. This timelapse video shows how the beads form and move, almost dancing around the glass. The video’s author, Dan Quinn, has a second video with an awesome visual explanation of the behavior that’s well worth watching, too! (Video credit and submission: D. Quinn)

Tears of Wine
Physicist Richard Feynman once famously ended a lecture by describing how the whole universe can be found in a glass of wine. And there is certainly plenty of fluid dynamics in one. In the photo above, we see in the shadows how a film of wine drips down into the main pool below. This effect is known by many names, including tears of wine and wine legs; it can also be found in other high alcohol content beverages. Several effects are at play. Capillary action, the same effect that allows plants to draw water up from their roots, helps the wine flow up the wall of the glass. At the same time, the alcohol in this wine film evaporates faster than the water, raising the surface tension of the wine film relative to the main pool of wine below. Because of this gradient in surface tension, the wine will tend to flow up the walls of the glass away from the area of lower surface tension. This Marangoni effect also helps draw the wine upward. When the weight of the wine film is too great for capillary action and surface tension to hold it in place, droplets of wine–the legs themselves–flow back downward. (Photo credit: Greg Emel)

“Surface Tension”
From a series called “Surface Tension,” these ink and water drawings by Marguerite French explore the effects of diffusion, surface tension, and evaporation. The forms left by the thin layer of liquids suggest other natural processes like erosion, weathering, and the rings inside trees. (Photo credits: Marguerite French)