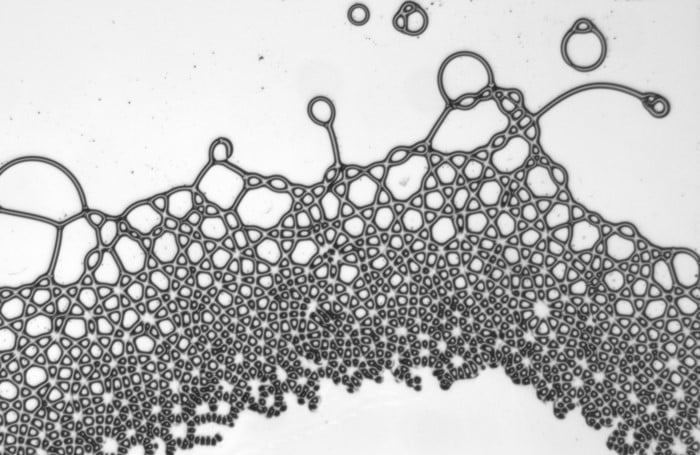
With the right application of force, liquids can take on shapes that defy our intuition. Here researchers sandwiched two immiscible oils between glass slides and applied an electric field. Because the two oils have different electrical responses, charges build along the interface between them. These charges lead to non-trivial electrohydrodynamic flows and a multitude of bizarre shapes. They observed polygonal droplets, streaming droplet lattices, and spinning filaments among others. As long as the electric field remains on, the wild behaviors continue; once the field is turned off, the oils relax back to typical, rounded drops. (Image, video, and research credit: G. Raju et al.; via Physics World)
Tag: droplets

Spreading By Island
How does a droplet sinking through an immiscible liquid settle onto a surface? Conventional wisdom suggests that the settling drop will slowly squeeze the ambient fluid film out of the way, form a liquid bridge to the solid beneath, and spread onto the surface. But for some droplets, that’s not how it goes.
While watching a glycerol droplet settle through silicone oil, researchers discovered a new mechanism for wetting. Initially, the silicone oil drained from beneath the drop, as expected. But then the thinning of the film stalled. Tiny bright spots (above) appeared beneath the light and dark interference fringes of the parent drop. These are spots of glycerol, formed when material from the main drop dissolved into the oil and then nucleated onto the solid surface below. Over time, the island-like spots of glycerol grew. Eventually one grew large enough to coalesce with its parent drop (below), causing the glycerol to quickly spread over the solid surface!

Islands of liquid (darker rings) grow beneath a parent drop (brighter rings) until reaching a size where they coalesce, causing the interference fringes to disappear. The key to this phenomenon seems to be that immiscibility isn’t perfect. Even trace amounts of solubility between the drop and surrounding fluid are enough to allow these islands to form. And once formed, the islands will grow as long as the drop fluid and the solid surface are chemically attractive. (Image, research, and submission credit: S. Borkar and A. Ramachandran; see also Nature Behind the Paper)

Liquid Umbrellas
Two well-timed and properly aligned droplets combine to create these umbrella-like fluid sculptures. The initial drop creates a jet that shoots upward. When the second drop hits that jet, it forms an expanding sheet of liquid like a miniature parasol. The higher the viscosity of the drops, the less lacy and unstable the sheet’s rim will be.
Although set-ups for these sorts of pictures can be finicky, they’re very doable, even for amateur photographers. In fact, the techniques used here have been around for about a century! (Image and research credit: A. Kiyama et al.)

Sliding Along
Robust, self-cleaning surfaces are a holy grail for many engineers, but they’re tough to achieve. One necessary ingredient for a self-cleaning surface is the ability to shed water, which is why superhydrophobic coatings and surface treatments are popular. Here, researchers prompt their droplets to move at speeds up to 16 cm/s by dropping them onto a thin layer of heated oil.
Longtime readers will no doubt be reminded of self-propelling Leidenfrost drops, but this situation is not quite the same. In general, the oil layer suppresses the Leidenfrost effect. Instead, the oil heats the drop, evaporating its vapor. A bubble of vapor will nucleate at a random location in the droplet and eject itself, pushing the drop in the opposite direction. Because of the disruption caused by that ejection, new bubbles will preferentially form at the same spot, providing an ongoing supply of vapor that keeps the drop sliding in the same direction. It’s like a miniature rocket zooming along the oil film! (Image and research credit: V. Leon and K. Varanasi; via APS Physics)

Taking A Turn
Water droplets immersed in a mixture of oil and surfactants will move about, propelled by the Marangoni effect. Surfactant molecules congregate along the interface between the water and oil, but they do not do so uniformly. This uneven grouping causes variations in the surface tension, which in turn creates flows inside the droplet from areas of low surface tension to ones with higher surface tension. Those internal flows then dictate how the droplet as a whole moves.
Researchers found that droplet trajectories in these systems depend on the droplet’s size. Small droplets move in relatively straight lines, whereas larger droplets take highly curved paths. The difference comes from the way surfactants get distributed around the drop’s interface. Larger drops are more sensitive to shifts in surfactant location, making them more prone to take changeable, curving paths. (Image credits: top – P. Godfrey, others – S. Suda et al.; research credit: S. Suda et al.; via APS Physics)

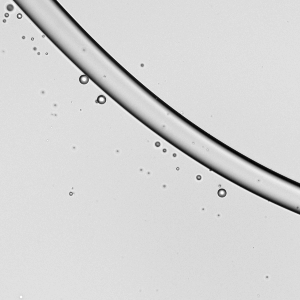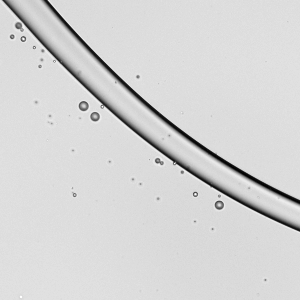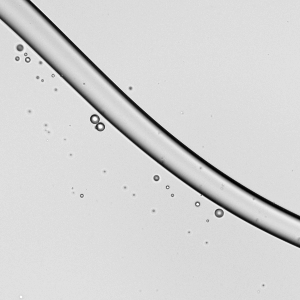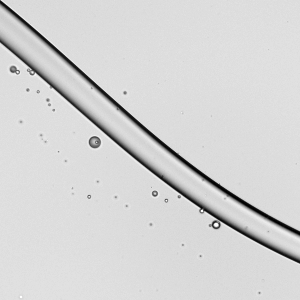
Falling Beads
Liquids flowing down a fiber can form bead-like droplets that may sit symmetrically (a) or asymmetrically (b) on the fiber. In general, the asymmetric droplets appear as surface tension increases or as the fiber diameter increases. The pattern of the droplets changes with flow rate. Within each subfigure, the flow rate increases from left to right. At low flow rates, we see only one or two large droplets migrating down the fiber. At moderate flow rates, a regular pattern of drops emerges. And at high flow rates, droplets coalesce on the fiber to form drops large enough that they fall and sweep up the downstream droplets. (Image and research credit: C. Gabbard and J. Bostwick)

Acidic Sea Spray
As waves crash and break, they generate a spray of droplets — known as aerosols — that make their way into the atmosphere. Researchers investigated the chemistry of these aerosol droplets by generating spray in a wave tank filled with ocean water. They found that aerosol droplets are far more acidic than the ocean they come from, and the smaller the droplet, the more acidic it is. This acidification happens in a matter of minutes, as acidic gases interact with the spray. Their findings will be critical for accurately modeling the climate connections between our oceans and atmosphere. (Image credit: Elle; research credit: K. Angle et al.; via OceanBites; submitted by Kam-Yung Soh)

Gathering Droplets
In deserts around the world, plants have adapted to collect as much moisture as they can. Geometry aids them in this endeavor because droplets on the tip of a cone will move toward its thicker base. The motion takes place due to a imbalance in surface tension forces on either end of the droplet.
As the droplet moves up a cone, it changes shape from a barrel-like drop that fully covers the conical surface to a clamshell-shaped droplet that hangs only from the bottom of the cone. (Image and research credit: J. Van Hulle et al.)

Oil in Water
In the decade since the Deepwater Horizons oil spill, scientists have been working hard to understand the intricacies of how liquid and gaseous hydrocarbons behave underwater. The high pressures, low temperatures, and varying density of the surrounding ocean water all complicate the situation.
Released hydrocarbons form a plume made up of oil drops and gas bubbles of many sizes. Large drops and bubbles rise relatively quickly due to their buoyancy, so they remain confined to a relatively small area around the leak. Smaller drops are slower to rise and can instead get picked up by ocean currents, allowing them to spread. The smallest micro-droplets of oil hardly rise at all; instead they remained trapped in the water column, where currents can move them tens to hundreds of kilometers from their point of release. (Image and research credit: M. Boufadel et al.; via AGU Eos; submitted by Kam-Yung Soh)