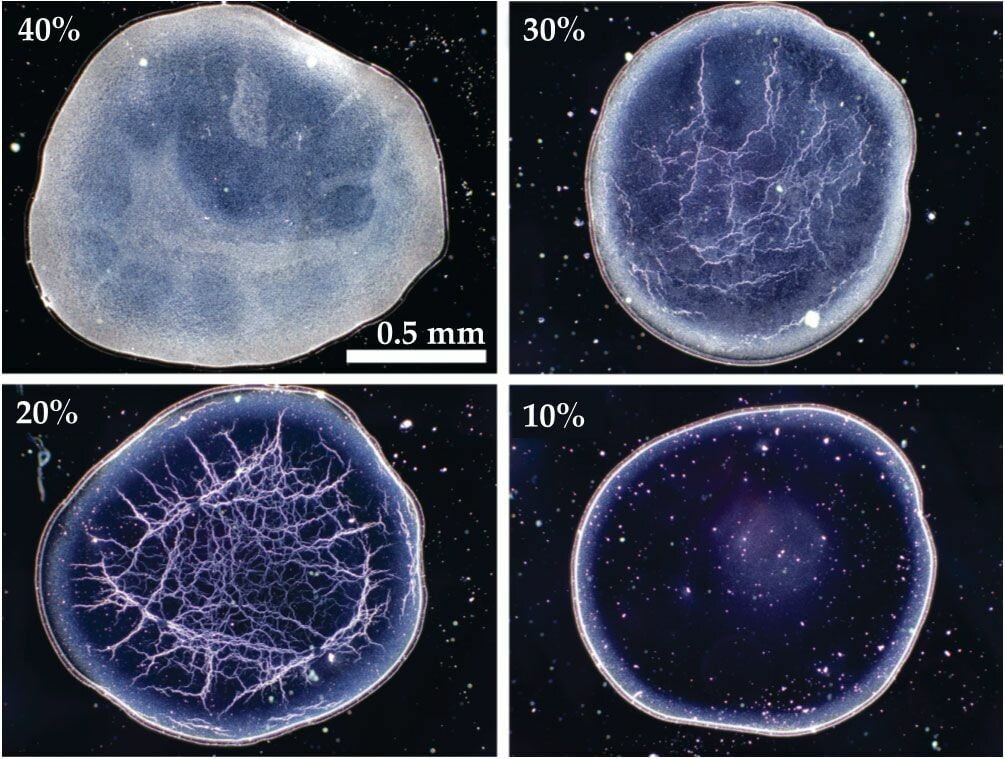
Soap film ruptures are well understood for your typical bubble solution, but what happens when tiny particles get added to the soap film? That’s the question in this recent study. Researchers added 660-nanometer particles, in varying amounts, to their soap films to see how it affected rupture. When they broke the films just after formation (top image), they found results that were quite similar to the usual, particle-free case. But when the films sat for awhile before breaking spontaneously (bottom image), the rupture caused wrinkling and folding similar to a piece of fabric. The researchers hypothesize that aging allowed the soap film to thin until the film and the particles were similar in size. Then, when the film ruptured, the particles affected how it broke up. (Image and research credit: P. Shah et al.)