Among objects in our solar system, the Moon is rather unusual. It’s the only large moon paired with a rocky planet, and only Pluto’s Charon boasts a larger size relative to its planet. Chemically speaking, the Moon is also extremely similar to the Earth, which is part of why scientists theorized that the moon coalesced after the proto-Earth collided with a Mars-sized object. But lingering questions remained, like why the Moon is rich in iron oxide compared to the Earth.
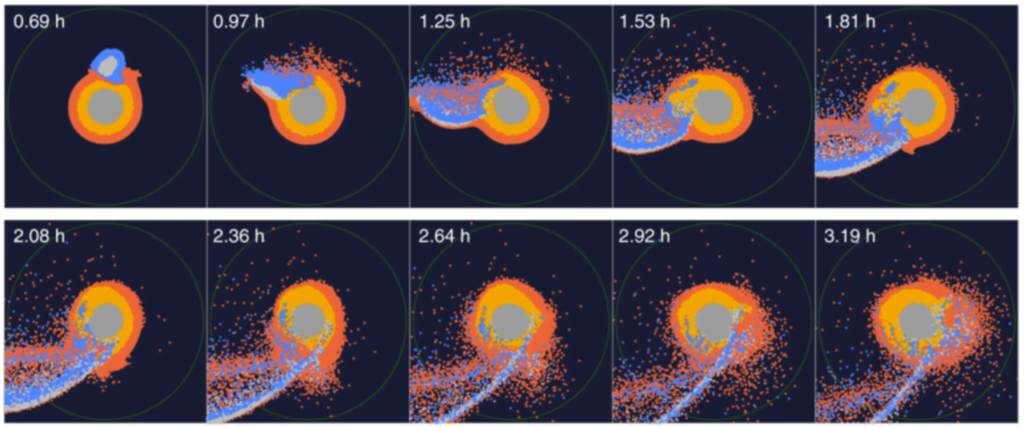
A new study tweaks the idea of the giant impactor by adding a magma ocean to the proto-Earth. In the early days of the solar system, collisions were so common that larger bodies (> 2*Mars) probably maintained a molten ocean. By simulating collisions with and without a magma ocean and studying the final composition of these simulated Earth-Moon-systems, the researchers found that a molten ocean not only matches the expected size and orbital characteristics of the two bodies, but the results reflect the actual chemical make-up of the real Earth and Moon, too! (Image credits: moon – N. Thomas, impact simulation – N. Hosono et al.; research credit: N. Hosono et al.; via Ars Technica; submitted by Kam-Yung Soh)