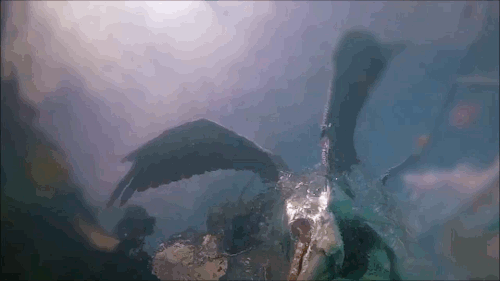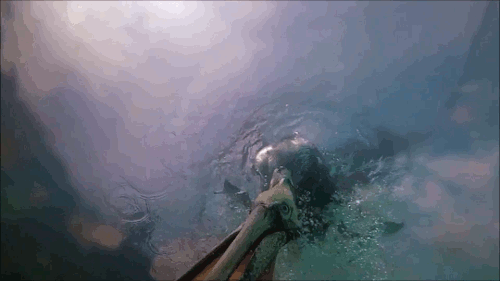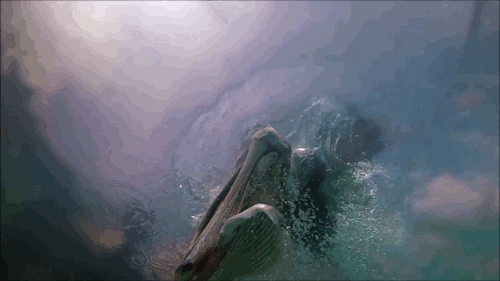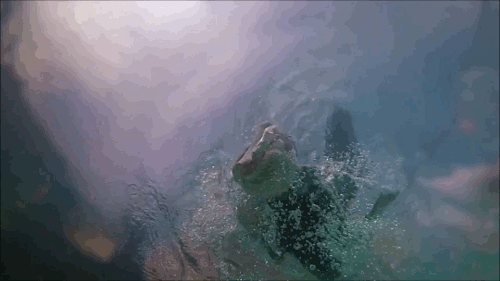Surface tension is the result of an imbalance between intermolecular forces near an interface. Imagine a water molecule far from the surface; it is surrounded on all sides by other water molecules and feels each of those pulling on it. Since all the nearby molecules are water, the tugs from every direction balance and there is no net force. Now imagine that water molecule near the air interface. Instead of being influenced on all sides by water, our molecule now feels water in some directions and air molecules in another. The water molecules tug harder on it than air, leaving a net force that pulls along the interface. This is surface tension, and, for a liquid-gas interface, it behaves somewhat like an elastic sheet. Surface tension is even strong enough to let a jet of soap solution bounce repeatedly off a soap film. Each bounce deforms the interface, like a trampoline dimpling when someone jumps on it, but surface tension keeps the interface taut enough for the jet to skip off without breaking it. (Image credit: C. Kalelkar and S. Phansalkar, source)
Month: October 2017

Flag Flapping
Everyone has watched a flag flutter in the breeze, but you may not have given much thought to it. One of the earliest scientists to consider the problem was Lord Rayleigh, who wrote an aside on the mathematics of an infinite flag flapping in a paper on jets (pdf). Today researchers consider the problem in terms of fluid-solid interaction; in other words, to study a fluttering flag, you must consider both the properties of the flag – its flexibility, length, elasticity, and so on – and the properties of the fluid – air speed, viscosity, etc. The combination of these factors governs the complicated shapes taken on by a flag. The image above is a composite of several photos of a string (a 1-d flag) flapping in a flow that moves from left to right. By combining photos, the image highlights the envelope of shapes the flag takes and demonstrates at a glance just how far the flag flutters in either direction along its length. (Image credit: C. Eloy)

Porous Fingers
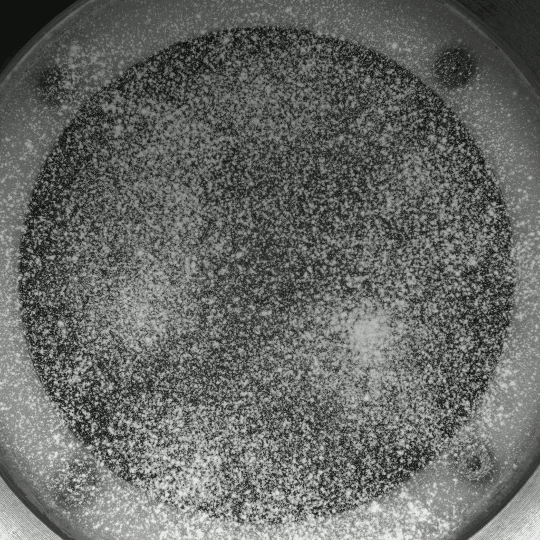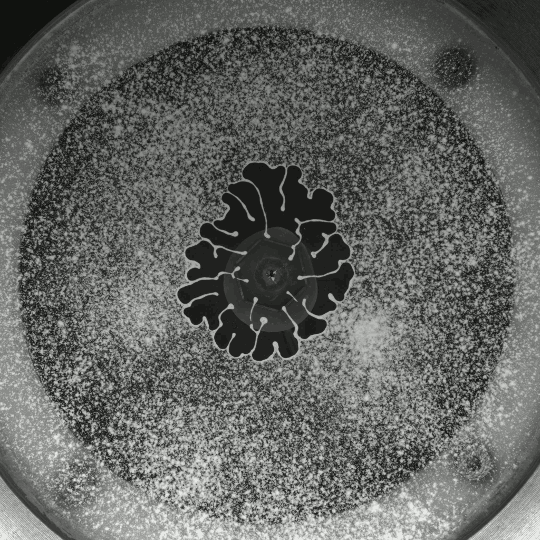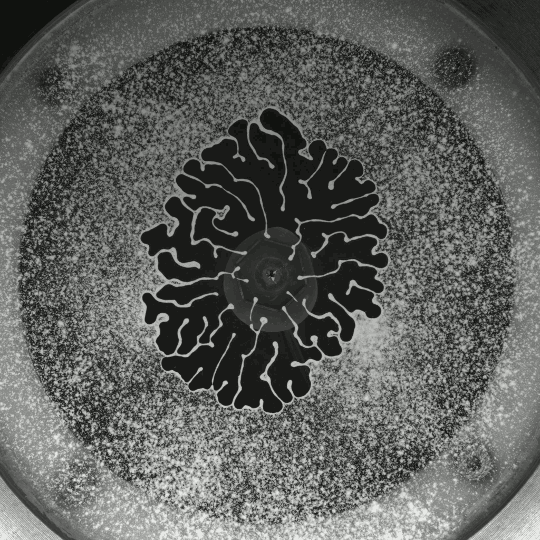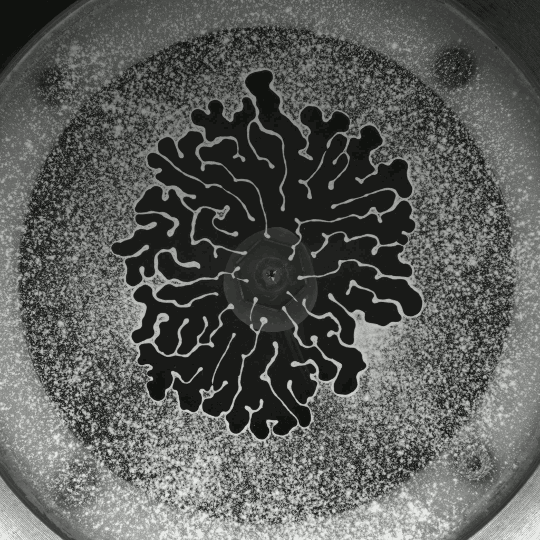
If you inject a less viscous fluid, like air, into a narrow gap between two glass plates filled with a more viscous fluid, you’ll get a finger-like instability known as the Saffman-Taylor instability. If you invert the situation – injecting something viscous like water into air – the water will simply expand radially; you’ll get no fingers. But that situation doesn’t hold if there are wettable particles in the air-filled gap. Inject water into a particle-strewn air gap and you get a pattern like the one above. In this case, as the water expands, it collects particles on the meniscus between it and the air. Once the concentration of particles on the meniscus is too high for more particles to fit there, the flow starts to branch into fingers. This creates a greater surface area for interface so that more particles can get swept up as the water expands. (Image and research credit: I. Bihi et al., source)

The Rose-Window Instability
This polygonal pattern is known as the rose-window instability. It’s formed between two electrodes – one a needle-like point, the other flat – separated by a layer of oil. The pointed electrode’s voltage ionizes the air nearby, creating a stream of ions that travel toward the flat electrode below. Oil is a poor conductor, however, so the ions build up on its surface until they’re concentrated enough to form a dimple that lets them reach the lower electrode. At higher voltages, the electrical forces driving the ions and the gravitational force trying to flatten the oil reach a balance in the form of the polygonal cell pattern seen above. Smaller cells form near the needle electrode, where the electrical field is strongest and the temperature is highest, as revealed in thermal and schlieren imaging (lower images) that shows a warm stream of gas impacting there.
As a final note, I’ll add that the latest in this research comes from a paper by a Pakastani teenager. It’s never too early to start contributing to research! (Image and research credit: M. Niazi; via NYTimes; submitted by Kam-Yung Soh)

Detergency
Have you ever wondered just how detergents are able to get grease and oil off a surface? This simple example demonstrates one method. In the top image, a drop of oil sits attached to a solid surface; both are immersed in water. An eyedropper injects a surfactant chemical near the oil drop. This lowers the surface tension of the surrounding water and allows the mixture to better wet the solid. That eats away at the oil drop’s contact with the surface. It takes awhile – the middle animation is drastically sped up – but the oil droplet maintains less and less contact with the surface as the surfactant works. Eventually, in the bottom image, most of the oil drop detaches from the surface and floats away. (Image credits: C. Kalelkar and A. Sahni, source)

Sunglinting Seas
Sunlight reflecting off the Earth can reveal a remarkably rich picture of our planet’s activity. The silver-gray areas seen in this satellite image are sunglint, where lots of light is reflected back to space. Sunglint occurs in regions with very few waves; more waves – like in the bluer areas – mean more directions in which light can be scattered. The reason for these rough and smooth waters is atmospheric: the prevailing summer winds blow across the Aegean from the north. In open water, that wind drives up the waves, but rocky islands disrupt the flow, leaving “wind shadows” on their southern, leeward sides where the waves are smaller. (Image credit: J. Schmaltz; via NASA Earth Observatory)

Surfing Mists
Watch your hot cup of coffee or tea carefully, and you may notice a white mist of tiny micron-sized droplets hovering near the surface. These microdroplets are a little understood part of evaporation. They form over a heated liquid, levitating on vapor that diffuses out from them and reflects off the liquid surface. (This is similar to the Leidenfrost effect, but the authors note it occurs at much lower temperatures. Unrelated research has suggested the Leidenfrost effect can occur at lower temperatures when there is very little surface roughness.)
One of the particularly peculiar behaviors of these tiny levitating microdroplets is that they can exist over dry surfaces as well. The image above shows microdroplets migrating from a liquid surface (right) to a dry surface (center and left). When the droplets near the contact line, they encounter a strong upward flow due to increased evaporation there. This launches the droplets upward and they sail to the dry area. There, their vapor layers continue creating levitation and provide a cushion between them and their neighbors, causing the drops to self-organize into arrays. (Image credit: D. Zaitsev et al.; via Physics World; submitted by Kam-Yung Soh)

Pelican Diving
Pelicans, like many sea birds, are aerial divers. They spot their prey from high above, bank, and dive into the water to catch the fish. Although they hit the water at high speeds, pelican diving techniques differ somewhat from plunge divers like gannets or boobies. Pelicans are only aiming for a shallow dive, so they have features – like their expandable neck pouch – that help them decelerate quickly instead of taking a full-body plunge. The goal is to increase drag after the head enters, slowing everything down. That can add more stress to the bird’s neck – the rest of the body is still moving quickly even after the head begins to slow. To counter this compression, the birds must have strong neck muscles to stabilize their spines during the impact process. (Video and image credit: Deep Look)


Controlling Leidenfrost Drops
On a surface much hotter than their boiling point, droplets can surf on a layer of their own vapor due to the Leidenfrost effect. Recent research has shown that textured surfaces like ratchets can create corrals, traps, and mazes for such droplets. Here, researchers manipulate the propulsion of Leidenfrost drops using non-parallel grooves instead. When placed between two non-parallel plates, the droplet is squeezed by side forces perpendicular to the walls, with the resultant force in the direction where the gap widens. In most states, friction forms an opposition to this squeeze, but for Leidenfrost droplets that frictional force is negligible. Instead, the squeezing from the plates launches droplets toward the wider end of the groove, allowing researchers to design repellers (top) and traps (bottom) for the fast-moving drops. (Image credits: C. Luo et al., source)

Convection
Blue paint in alcohol forms an array of polygonal convection cells. We’re accustomed to associating convection with temperature differences; patterns like the one above are seen in hot cooking oil, cocoa, and even on Pluto. In all of those cases, temperature differences are a defining feature, but they are not the fundamental driver of the fluid behavior. The most important factors – both in those cases and the present one – are density and surface tension variations. Changing temperature affects both of these factors, which is why its so often seen in Benard-Marangoni convection.
For the paint-in-alcohol, density and surface tension differences are inherent to the two fluids. Because alcohol is volatile and evaporates quickly, its concentration is constantly changing, which in turn changes the local surface tension. Areas of higher surface tension pull on those of lower surface tension; this draws fluid from the center of each cell toward the perimeter. At the same time, alcohol evaporating at the surface changes the density of the fluid. As it loses alcohol and becomes denser, it sinks at the edges of the cell. Below the surface, it will absorb more alcohol, become lighter, and eventually rise at the cell center, continuing the convective process. (Image credit: Beauty of Science, source)