Here’s a different take on ferrofluids. Instead of spikes, we get 2D patterns reminiscent of these ones. Most likely the ferrofluid is trapped under glass as part of a Hele-Shaw cell. The results remind me some of chaotic Rayleigh-Benard convection cells, actually.
Month: January 2011

Butterfly Soap Spiral
A stationary soap film disturbed by a flapping foil (seen in the top center) creates a butterfly-like double spiral roll. Two vortices form at the tip of the foil each time it changes direction; look carefully and you can see those tiny vortices all the way through the spirals. (From the 2010 Gallery of Fluid Motion; pdf)
Reader Question: Hot Air Balloon Physics
lazenby asks:
and boyancy in air? is the lifting capacity of a hot air balloon equal to the modulo of the weight of the air in the balloon with the weight of the same volume of air outside the balloon?
for that matter, does the lift of a big helium weather balloon decrease as air pressure, and so weight of the air outside the balloon, drops? and is this exactly counterbalanced by the lessening density of the helium in the balloon?
all of these things keep me awake.
Hopefully you won’t be sleepless much longer. Buoyancy in air follows the same principles as buoyancy in water. Determining the lifting capacity of a balloon is a matter of determining how heavy the balloon can be before the buoyant force is equal to the weight. See the free body diagram and little derivation below to see what the maximum payload mass is for a helium balloon. You can click on the picture to enlarge it.

The second part of your question raises some interesting points. As a balloon’s altitude increases, the atmosphere around it gets colder and less dense, all of which should reduce the buoyant force. At the same time, the balloon itself expands to equalize the pressure inside and outside of the balloon, which should increase the buoyant force. (At some point the pressure drops sufficiently that the tensile strength of the balloon material is unable to cope with that expansion and the balloon bursts, but we’ll ignore that here.) For this problem, we’d want to know what payload the balloon can carry without losing lift, and, with a couple assumptions, that’s pretty easy to figure out. I’ve done that derivation below.

The real key to the calculation is assuming that the helium in the balloon maintains the same temperature as the air outside. Since balloons rise slowly, this seemed a more reasonable assumption than imagining that the balloon remains warm compared to its surroundings. That calculation is doable as well but requires more than a couple lines, unfortunately! Thanks for your questions!
The GE Show
[original media no longer available]
While this video is not strictly about fluid dynamics, there are some pretty cool high-speed fluids moments in it. Watch the reaction of the gelatins as objects hit them and observe the deformation of the water balloons as they strike. (via JetForMe)
Frost on Superhydrophobic Surfaces
Frost formation and ice adhesion on superhydrophobic surfaces
For anyone with further interest in the ice formation on superhydrophobic surfaces story we posted recently, the published paper is currently offered by AIP for free. #

Reader Question: Swimming and Buoyancy
aniiika asks:
How does buoyancy relate to swimming?
Buoyancy is the force that enables a swimmer to float in the water, even when still. Buoyant force is equal to the weight of the fluid displaced by the swimmer; in other words, the density of the fluid multiplied by the volume of the swimmer that is submerged.
Different people float at different heights in the water depending on many factors, such as body shape, amount of fat, and how much air is in their lungs. All of these things affect a person’s volume and/or density, thereby affecting the buoyant force they experience.
Because a person’s body is not fully submerged their center of buoyancy–the point where all buoyant forces on the body can be represented by a single force–does not coincide with the center of mass (sometimes referred to as center of gravity). Where those forces are relative to one another determines the stability of a person floating in the water. Everyone’s center of buoyancy is higher than their center of mass, so people always float stably in an upright orientation. Our legs, for example, don’t float as well as our torsos, so, when floating horizontally, one’s legs will tend to sink.
Swimmers can control their buoyancy to their advantage by actually pressing their upper chests further into the water. This tends to bring one’s hips closer to the surface and can reduce drag (#).

Chaos in Suspension
In science, the term chaotic is used to describe a system whose behavior is highly sensitive to initial conditions. This means that the end state can vary widely based on small changes at the start–also commonly known as the butterfly effect. Many fluid dynamical systems are chaotic, especially turbulent ones. Above are a series of photos showing the suspension of particles in a horizontally rotating cylinder. In parts A-D, the speed of rotation of the cylinder is increased, resulting in dispersion of the particles. As rotation rate is increased further, interesting concentration patterns form. #

Levitating Liquid Oxygen
The Leidenfrost effect occurs between a fluid and a solid of vastly different temperatures. In the case of liquid oxygen, a thin layer of the oxygen vaporizes on contact with the room temperature solid, leaving a droplet of liquid oxygen to float along on its own vapor. Oxygen droplets are paramagnetic, meaning that they are susceptible to magnetic fields; in this video, scientists demonstrate how magnets can affect the motion of these droplets.

Vibrating Fluid Interfaces
The Faraday instability forms when a fluid interface is vibrated. This high-speed video shows the differences in the shapes formed by a vibrated fluid interface when the two fluids are miscible–capable of mixing–and when they are immiscible–like oil and water. Note how the miscible interface breaks down quickly into turbulence, but the immiscible interface maintains a complex shape.

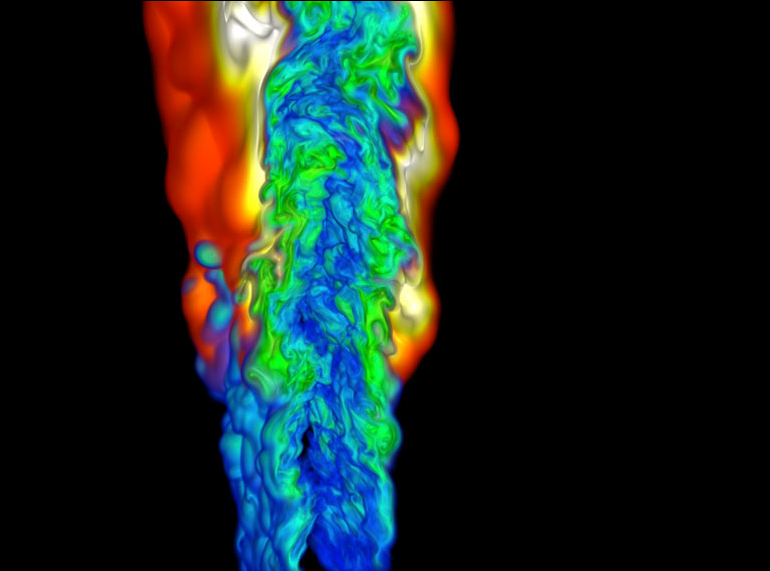
Colorful Computational Combustion
Many fluid dynamics problems are so complicated that they require supercomputers to calculate the mathematical and physical details. This image shows a computer simulation of a cold ethylene jet combusting in hot air. Different colors indicate different combustion by-products. Researchers use simulations like this one to investigate ideal flames that improve efficiency in applications like cars or jet engines. #