Wispy white cirrus clouds cover dark skies glittering with stars in Roman De Giuli’s “Heaven”. Or so it appears. In reality, these skyscapes are made with watercolors, ink, and acrylic paint. The vistas are gorgeous regardless of whether they’re driven by turbulent convection (as in the atmosphere) or the Marangoni effect (as in this video)! (Video and image credit: R. De Giuli)
Tag: surface tension

When Bubbles Don’t Die
In a pure liquid, most bubbles pop almost immediately. But with a simple ingredient — a little heat — bubbles can live almost indefinitely. The mechanism is revealed in this video when the researchers use an infrared camera to watch a bubble on a heated pool. The top of the bubble is cooler than the rest of the liquid, forming colder, denser droplets that slide down. But the cooler liquid also has a higher surface tension, which draws warm liquid up the bubble, replenishing it. The result is a stable bubble that simply carries on. (Image and video credit: S. Nath et al.)

Morphing Particle Rafts
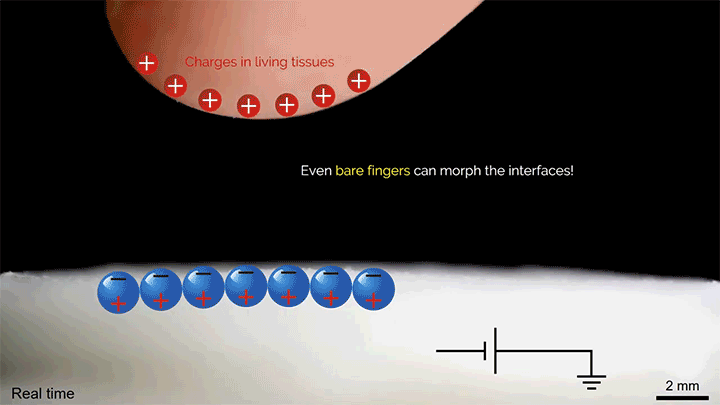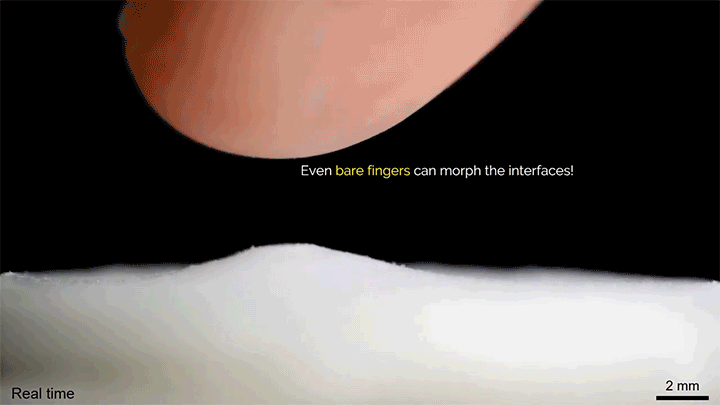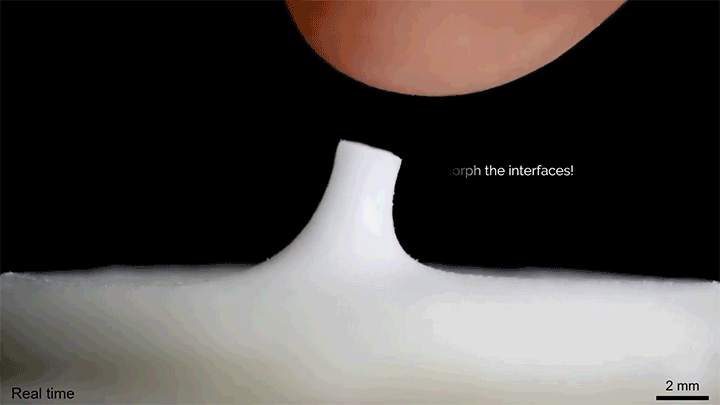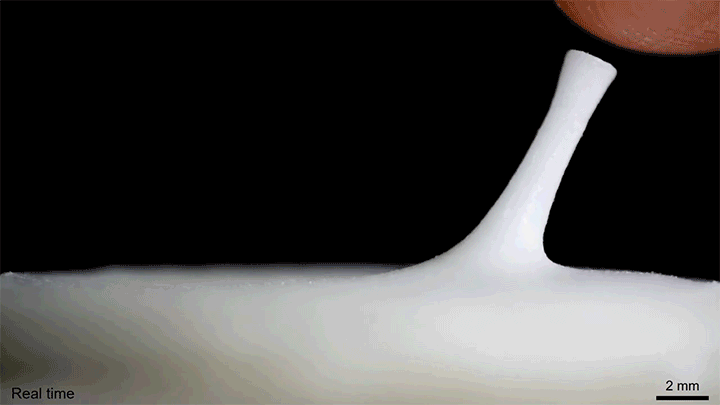
A layer of tiny glass beads sitting atop a pool of castor oil becomes a morphing surface in this video. Applying an electric field creates enough electrostatic force to draw the interface upward against the power of both gravity and surface tension. Moving the electric field — either by shifting the electrode or simply moving a finger over the surface — is enough to pull columns of fluid along! I could imagine this making some very cool human-machine interfaces one day. (Image and video credit: K. Sun et al.)

“Delusion”
Soap films are ephemeral and ever-changing. The shifting concentration of surfactants along the surface of the film, combined with thermally-driven convection, keeps the fluid in motion. The shifting colors reflect subtle changes in the soap film’s thickness. Over time, gravity drains fluid from the top of the film, thinning it to the point that it appears black. This photo from Bruno Militelli captures all of that detail in a striking and fascinating image that earned him 2nd place in the Manmade category of the Close-Up Photographer of the Year awards. You can find more winners of the competition here, and more of Militelli’s work on his website and Instagram. (Image credit: B. Militelli)

Acrylic Paint Fractals
Here’s a simple fluids experiment you can try at home using acrylic paints, ink, isopropyl alcohol and a few other ingredients. When dropped onto diluted acrylic paint, a mixture of black ink and alcohol spreads in a fractal fingering pattern. The radial (outward) flow is driven by the alcohol’s evaporation, which increases the local surface tension and draws fluid outward. The shape and density of the fingers depends, at least in part, on the viscosity of the underlying paint layer; more viscous paint layers grow smaller and denser fractal patterns. (Image and video credit: S. Chan et al.)

The Assassin’s Teapot
The assassin’s teapot is a cleverly designed container that can pour from different reservoirs depending on how it’s held. Steve Mould digs into the physics in this video, and he builds a transparent cutaway version of the pot to show exactly how it works. This design uses two separate reservoirs, each with two holes — one in the spout and one concealed near the pot’s handle. By covering this breather hole, the server blocks air from flowing into the teapot, which also keeps the liquid inside from flowing out.
What holds the liquid in? Air pressure, with an assist from surface tension. Atmospheric pressure is enough to hold the fluid inside the pot, provided air has no separate way in. To get in through the spout, air would have to push into the pot at the same time as water coming out. Surface tension prevents that, though, because the spout is too narrow. The same physics keeps water inside a larger bottle with a wire mesh over its mouth. The mesh’s tiny holes are smaller than the capillary length of water, which is the length scale at which surface tension and gravity balance one another. As long as the spout and holes are smaller than that length, surface tension will keep the liquid from deforming enough to get out. (Video and image credit: S. Mould)

The Yarning Droplet
Marangoni bursting takes place in alcohol-water droplets; as the alcohol evaporates, surface tension changes across the liquid surface, generating a flow that tears the original drop into smaller droplets. Here researchers add a twist to the experiment using PMMA, an additive that dissolves well in alcohol but poorly in water. As the alcohol evaporates, the PMMA precipitates back out of the water-rich droplet, forming yarn-like strands. (Image and video credit: C. Seyfert and A. Marin)

Laser-Induced Jet Break-Up
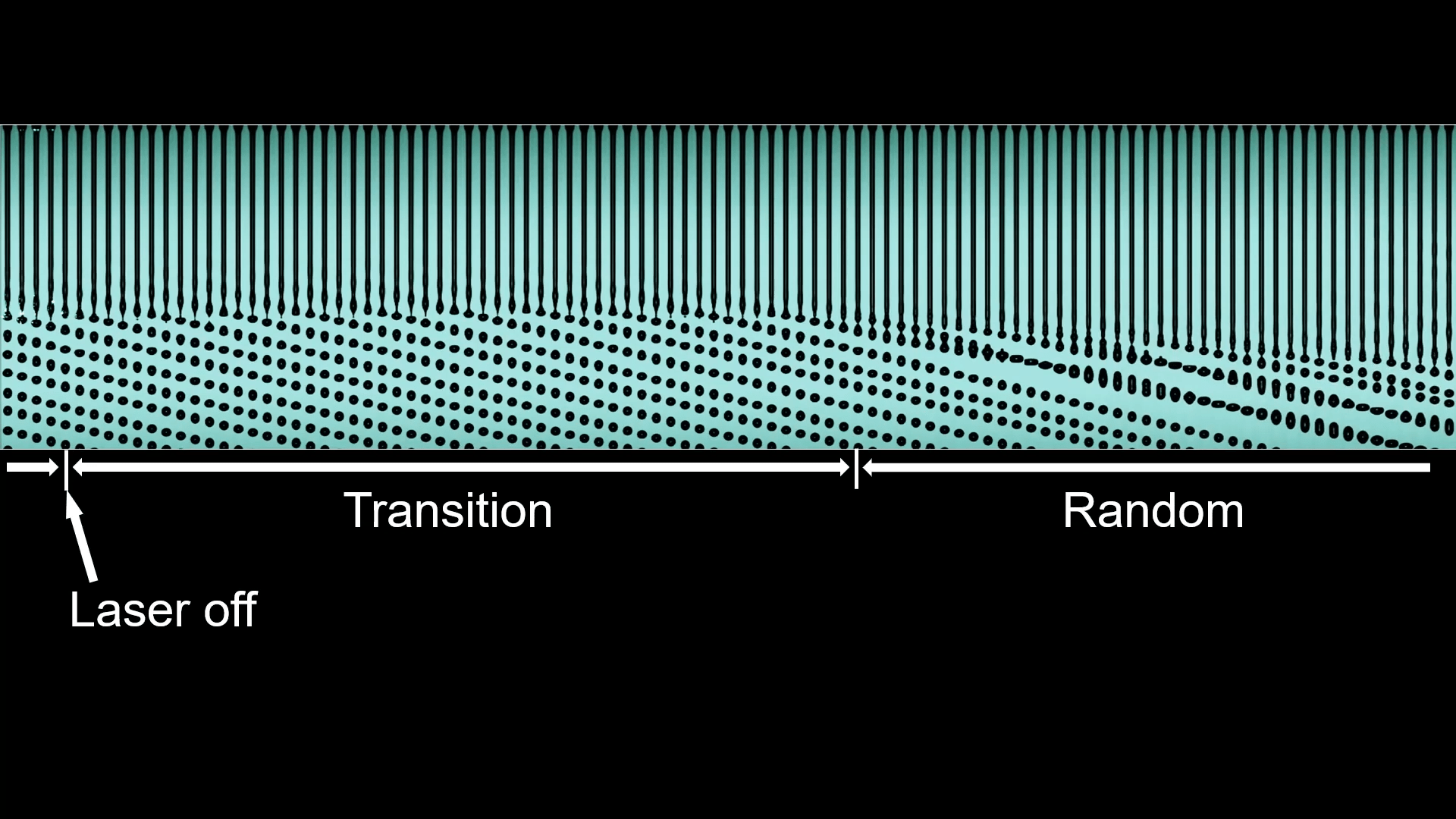
A falling stream of water will naturally break up into droplets via the Plateau-Rayleigh instability. Those droplets are random, unless something like vibration of the nozzle sets their size. In this study, though, researchers found that shining a laser beam on the stream can trigger an orderly break-up with droplets that are consistent in size and spacing.
The optofluidic phenomenon depends on a few different effects. The changing curvature of the liquid stream reflects the laser light, some of which undergoes total internal reflection and travels up the jet as if it were a fiber optic cable. Look closely in the right side of the second image, and you’ll see a periodic flicker of green light at the mouth of the nozzle. Those flashes of green reveal that the liquid jet is guiding the light upstream in bursts, each of which exerts an optical pressure that triggers the Plateau-Rayleigh instability.
When the laser first turns on, there’s a transition period before the orderly break-up begins, and, likewise, turning the laser off triggers a transition from orderly to random (top image). (Image and research credit: H. Liu et al.; via APS Physics; submitted by Kam-Yung Soh)

Solving the Teapot Effect
The teapot effect — that tendency for liquid to dribble down the outside of the spout when pouring — is a frustration to many tea drinkers. Unraveling the fluid dynamics of this phenomenon has taken various researchers decades, but a team now believe they’ve captured the problem fully. Their full mathematical description is quite dense, but it boils down to a subtle interplay of capillary, viscous, and inertial forces.
Essentially, they found that droplets will always form just under the lip of the spout, thereby keeping that area wetted. The flow rate of the pour (along with the geometry and surface characteristics of the spout) determines how large those droplets can grow. At low flow rates, the droplets can grow large enough to redirect the entire stream around the spout’s edge, creating a hugely frustrating mess. You can see this flow rate effect in the high-speed video below. (Image credit: S. Ferrari; video and research credit: B. Scheichl et al.; via Ars Technica; submitted by Kam-Yung Soh)

Breaking Compound Ligaments
When pulled, viscous liquids stretch into ligaments that thin and then break into droplets. In this video, researchers investigate how these ligaments break up, depending on their composition. The initial views show the break-up of a water-glycerol ligament (Image 1) and an oil ligament (Image 2). By placing a water droplet inside oil, the researchers got quite different results, including oil-encapsulated droplets (Image 3). The technique could be useful for making compound droplets, even with more than two components. (Image and video credit: V. Thiévenaz and A. Sauret)