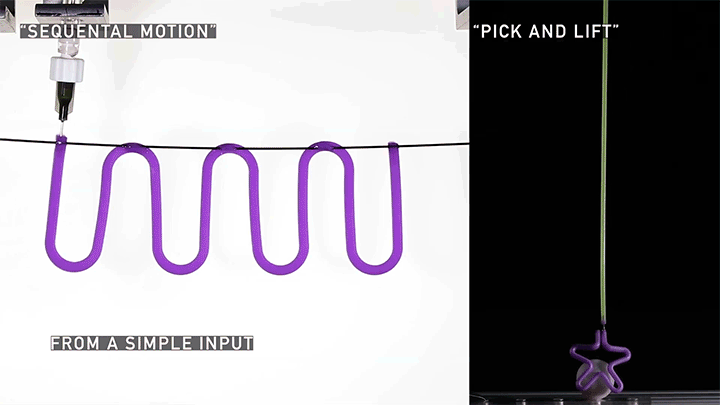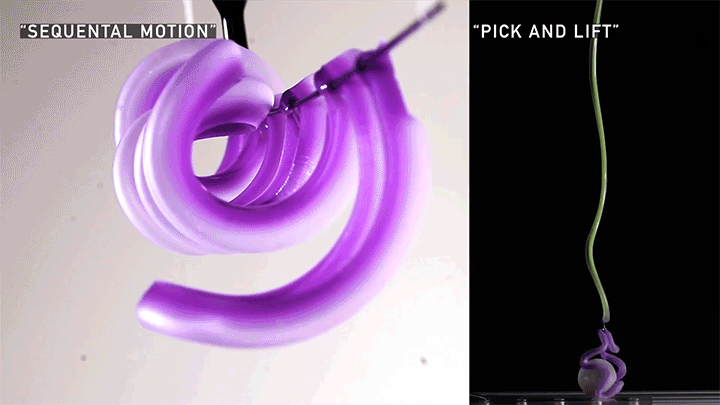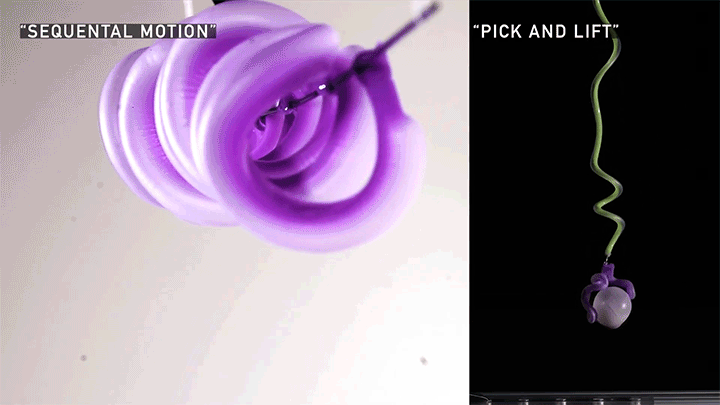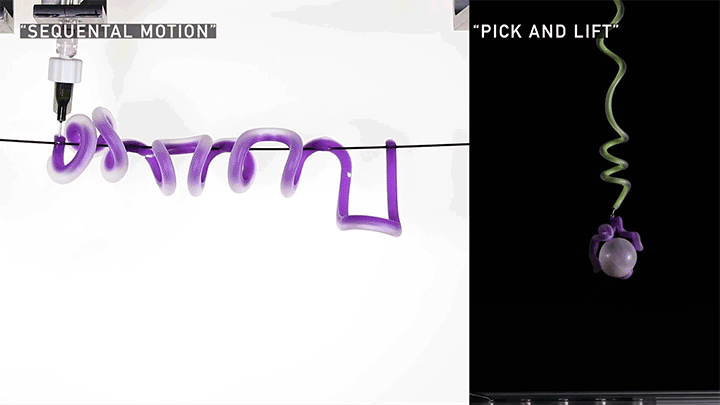
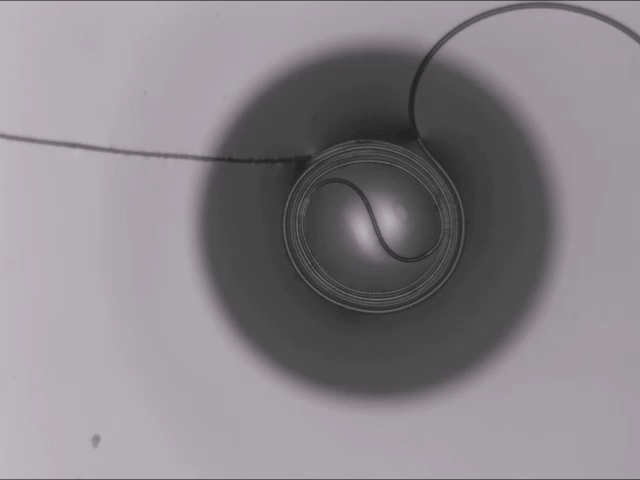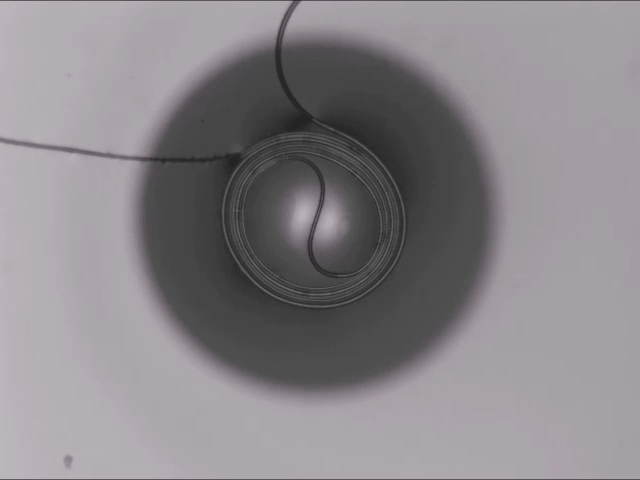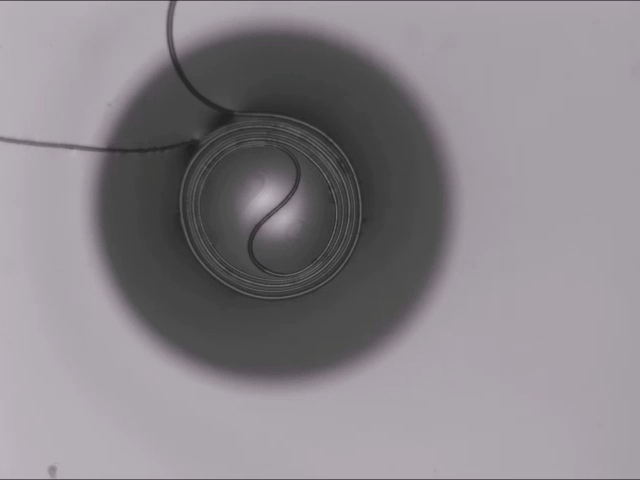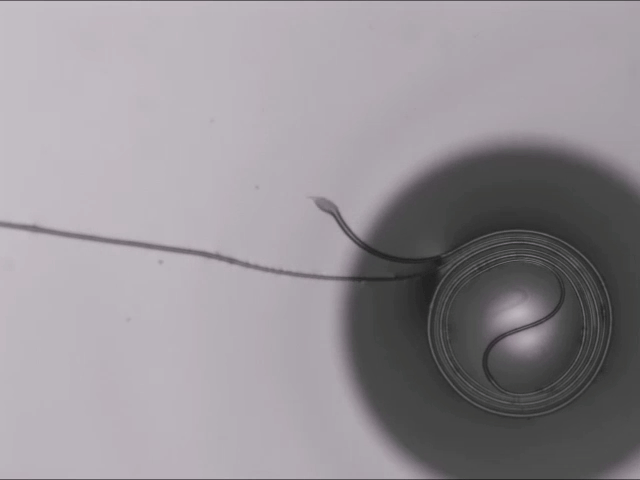
Hard materials don’t always work well in robotics. Here, researchers build soft actuators that can bend, curl, and tighten in order to manipulate objects. They begin by injecting liquid elastomer into a tube (Image 1), followed by a bubble of air. Buoyancy makes the air bubble rise within the tube, creating an asymmetric cross-section where the solidified elastomer has a thin shell along one side and a thicker wall along the other (Image 2). When high-pressure air is pumped into the soft tubes, their asymmetric cross-section makes them bend and twist (Image 3). The team found that they can tune the elastomer tubes to form complex shapes good for gripping and flexing — perfect for a soft robot! (Video and image credit: T. Jones et al.; research credit: T. Jones et al.)
Tag: solid mechanics

Bendable Ice
Ice — as we typically encounter it — is extremely brittle and easily broken. That’s due to defects in the ice, places where atoms have settled into a spot that does not match the perfect crystalline alignment. Because tiny defect-free threads of ice made by researchers turn out to be wildly flexible!
To make these perfect ice strands, each of which is a tiny fraction of the thickness of a human hair, researchers applied an electric voltage to a needle in a water-vapor-filled chamber. The technique condensed ice microfibers with perfect crystal structures in a matter of seconds. When bent, the microfibers actually shift from one crystalline arrangement to another in order to carry stress, and once the force is removed, the thread reverts back to its initial straight form. (Image and research credit: P. Xu et al.; via Science News; submitted by Kam-Yung Soh)

All Wound Up
A thin fiber sitting atop a bubble can spontaneously coil around the bubble thanks to elastocapillarity. (This seemingly bizarre behavior is also why wet strands of hair clump together.) Here’s the situation: The dark circle you see is all bubble; only a portion of the bubble — known as a spherical cap — sticks above the surface of the liquid. When a fiber sits across the top of the bubble, two things can happen: 1) the fiber simply sits there until the bubble bursts, or 2) the fiber starts to bend and wind around the bubble’s cap.
Bending the fiber takes energy. In this case, that bending energy comes from the system as a whole reducing its free energy. The fiber actually sinks into the bubble film in what the researchers call a “bridged” configuration, where the fiber sits inside the liquid film while also touching the air inside and outside the bubble. In this position, the interfacial energy of the fiber-bubble system is lower, leaving enough excess energy savings for the fiber to coil. (Image and research credit: A. Fortais et al.)

Tougher Hydrogels
Hydrogels are soft, stretchy solids made from polymer chains immersed in water. Engineers hope these materials will be good candidates for medical implants, but to reach that goal, hydrogels need to be durable enough to withstand repeated stretching and contortion without tearing. One team has built a better hydrogel by encouraging entanglement within the gel’s polymer network.
The polymers inside a hydrogel form their network with two main components: physical entanglements between polymer chains and chemical cross-links. If you imagine the polymers as a tangle of yarn, the cross-links would be spots where pieces of yarn are knotted together and the entanglements are spots where strands wrap and cross without knotting. If you pull on the network, cross-links (knots) will allow very little stretching, whereas the looser entanglements can stretch and deform without tearing. In a hydrogel with lots of entangled polymers but very few cross-links, the material is strong and stretchy without becoming brittle or easily torn. (Video credit: Science; research credit: J. Kim et al.)

Adjusting for Gusts
In flight, birds must adjust quickly to wind gusts or risk crashing. Research shows that the structure of birds’ wings enables them to respond faster than their brains can. The wings essentially act like a suspension system, with the shoulder joint allowing them to lift rapidly in response to vertical gusts. This motion keeps the bird’s head and torso steady, so they can focus on more complex tasks like landing, obstacle avoidance, and prey capture. (Image and research credit: J. Cheney et al.; submitted by Kam-Yung Soh)

Why Slicing Tomatoes Works
Picture it: a nice, ripe tomato. Your not-so-recently sharpened kitchen knife. You press the blade down into the soft flesh and… it explodes. Soft solids – like a tomato – don’t react well to cutting, but they slice just fine. Examining why that’s the case is at the heart of this model.
Tomatoes are essentially a gel encased in a thin skin. Gels are a kind of hybrid material — not quite liquid and not quite solid. They consist of a network of particles or polymers bonded together and immersed in a liquid. To cut that network apart, the downward force of the blade has to strain the gel past its limits, which squeezes out the surrounding liquid.
The researchers found that this liquid layer is key to how force from the knife’s motion gets transmitted. In particular, they found that the horizontal motion of a slice is necessary to initiate a cut, and that the gel parts most easily when the downward knife velocity is no more than 24% of the horizontal cutting speed. Press down any faster and the strain propagation fluctuates, creating that unfortunate tomato explosion. (Image credit: G. Fring; research credit: S. Mora and Y. Pomeau; via Ars Technica; submitted by Kam-Yung Soh)

Event: Machine Learning in Mechanics
This Thursday, August 27th, the U.S. National Committee on Theoretical and Applied Mechanics is holding a special free webinar series on Machine Learning in Mechanics. Details for each talk and a link to register are available here. Note that the event is free but registration is necessary if you want to receive the Zoom link.
Full disclosure: I am a member-at-large of the U.S. National Committee on Theoretical and Applied Mechanics.

Paint Versus Hydrogel
In this bizarre short film, we get to see a battle between dissolution and absorption. I think the Chemical Bouillon team has coated hydrogel beads in a layer of paint and then immersed them in water. As the beads absorb water, they expand and grow, tearing their fragile outer layer of paint to smithereens.
One thing that struck me when watching several of the sequences is just how regular the hole spacing in the paint is for the round hydrogels. That hints at an orderly breakdown in the solid paint layer while the interior hydrogel polymer symmetrically expands. It’s a little like watching holes grow in a splash curtain. (Video and image credit: Chemical Bouillon)

The Physics of Al Dente
It’s a simple weeknight routine: toss a handful of spaghetti noodles in boiling water, wait a few minutes, and enjoy with the sauce of your choice. But there’s a surprising amount of physics in the humble strand of spaghetti, and a new model focuses on the way spaghetti sags and curls as it cooks.
Spaghetti, like most pastas, is made of semolina flour mixed with water, extruded (in commercially produced spaghetti), and then dried. Once immersed in water, the rod of pasta begins to swell and soften as water works its way slowly inward. At the same time, it will lose some of its starches to the surrounding water. If the water is hot enough, the pasta undergoes an additional process, starch gelatinization, which is responsible for cooked pasta’s characteristic texture. That perfect al dente condition occurs right as the hydration front reaches the pasta’s core.
As all of this happens, the initially straight spaghetti strand sags, settles, and curls. Researchers found that, even with a relatively simple model that assumes spaghetti doesn’t stick to the pot, they could capture shape change of individual spaghetti strands, suggesting it’s possible to identify perfectly cooked pasta by shape alone. (Image credit: Pixabay; research credit: N. Goldberg and O. O’Reilly; via Ars Technica)

Blooming Deposits
Evaporate a droplet full of silica nanoparticles, and you’ll get beautiful, flower-like films. As the water evaporates, dry nanoparticles build up in a solid deposit. The evaporation creates a pressure gradient that pulls toward the center of the drop, forcing the deposit to bend. As stress builds in the deposit, cracks form petal-like segments. The number of cracks is indicative of how much of the drop was solid material; the higher the volume fraction of particles is, the fewer cracks form and the less the deposit bends. (Image, video, and research credit: P. Lilin et al.)