During World War II, the Allies developed “dambuster” bombs that skipped repeatedly off the surface of the water before striking their target. The goal was to cleverly bypass their enemies’ defenses both above and below the surface. Although the original dambusters used spinning spheres, the ricochet physics works for many other configurations as well; essentially, the physics are identical to rock-skipping. Conventional bullets can also skip off the water, though the required angle for skipping depends strongly on the shape of the bullet. If the geometry of the bullet impact doesn’t generate enough hydrodynamic lift, there will be no skip. (Image credit: Barnes Wallis Foundation, source; research credit: V. Murali and S. Naik, pdf; submitted by Marc A.)
Tag: science

Testing Vesicles
In biology, vesicles contain a liquid surrounded by a lipid membrane. The characteristics of that membrane – like its stiffness – can change over time in ways that indicate other changes. For example, vesicles carrying HIV become stiffer as they grow more infectious. In the past, to observe these properties scientists used atomic force microscopes, which require removing the vesicles from the liquid in which they naturally reside. That’s problematic because it potentially changes how the vesicle responds.
Now researchers have developed a new method: a microfluidic system that subjects vesicles to electric fields in order to deform them and measures their properties without removing them from their carrier fluid. This provides a faster and more reliable method of testing a vesicle’s deformation, capable of testing hundreds of samples at a time. (Image credit: Wikimedia; research credit: A. Morshed et al.; submitted by Eric S.)

Pouring a Liquid Mirror
In this video, the Slow Mo Guys play with liquid gallium, giving us a chance to see how molten metals behave (outside of, say, the Terminator movies). Near its melting point, gallium is about six times denser than water, with a viscosity three times higher, and a surface tension about ten times greater. So how do those properties affect its behavior?
You may be surprised that when watching the gallium vibrate on a speaker or get poured into a pan, it doesn’t look all that different from water. Yes, it’s highly reflective, but, on the whole it doesn’t look radically different from a distance. We can use the Reynolds number to quantify what’s going on here. It’s a dimensionless number that compares the fluid’s inertial force to the viscous force. Imagine we have two versions of an experiment, one where we pour gallium at a given speed and one pouring at the same volume and speed but with water. If we compared the Reynolds numbers of the water and the gallium, they only differ by a factor of two. Overall, that’s not very much. That’s why the two pours look similar.
The story is different, though, if we look at individual drops of gallium and water, like when the first few drops of our pour hit the surface. Check out the gallium drops below. They’re conical on either end! This looks very different from what we expect with water droplets. You might think that’s because the metal is more viscous, but if we compare a water drop with a gallium drop of the same characteristic size and impact speed, we find a different story. For this, we’ll use the Ohnesorge number, which compares the viscous forces to a combination of inertia and surface tension. In this case, we find that the gallium drop’s Ohnesorge number is almost an order of magnitude smaller than the water droplet’s. That means that viscosity isn’t a major factor for our gallium drop. Both surface tension and inertia are more important.
But if the surface tension is so high, then why aren’t the droplets spherical? Mostly because they don’t have time to form spheres before they hit. Their shape suggests that they’ve only just broken into droplets, which makes sense if the pour is fast and the surface tension is strong. (Video and image credit: The Slow Mo Guys)

Liquid Magnets
Ferrofluids – those distinctively spiky liquids – are made up of magnetically sensitive nanoparticles in a carrier liquid, and although they respond to applied magnetic fields, they retain no magnetism outside of that field. But researchers have now succeeded in making actual liquid magnets. Shown above, these drops also contain ferromagnetic nanoparticles. But unlike traditional ferrofluids, in these drops the nanoparticles are not entirely free to move. They’re jammed together at the interface, so when a magnetic field is applied, the nanoparticles will align like tiny bar magnets. When that magnetic field is removed, though, the nanoparticles cannot easily reconfigure, so they remain aligned and the drops continue being magnetic.
Researchers hope these ultrasoft magnets, which can be manipulated remotely through magnetic fields, will be useful in the future for applications like targeted drug delivery. In theory one could introduce, say, chemotherapy drugs into one of these liquid magnets, then use magnetic fields to guide it directly to a cancerous tumor. (Image and research credit: X. Liu et al.; via Science News; submitted by Kam-Yung Soh)

Entraining Bubbles
If you stand on a bridge and watch the current flow past pylons below, you’ll see disturbances marking the wakes. Dragging a rod – or an oar – at a high enough speed through the water creates something similar: a wavy cavity in the fluid surface that surfs along behind the rod. The faster you pull the rod, the harder you’ll have to work, until that wake becomes so turbulent that it begins entraining air bubbles, like the tiny ones seen above. Once entrainment starts, the drag coefficient drops somewhat, presumably due to changes in the pressure distribution around the rod. The characteristics of air entrainment change with object size as well. Larger rods can entrain air through the cavity and not just in the wake. (Image and research credit: V. Ageorges et al.)

The Snowy Salt of the Dead Sea
At nearly 10 times saltier than the ocean, the Dead Sea is one of the saltiest places on Earth, and since 1979, scientists have observed it growing even saltier as snow-like salt precipitates to the bottom of the lake. Numerical simulations have now confirmed that this salt-fall is the result of double-diffusive salt fingers.
Here’s how the mechanism works: the upper layer of the lake is made up of warmer, saltier water covering deeper, colder waters. As the sun evaporates water near the surface, what’s left behind becomes saltier and heavier. Tiny pockets of this warm, salty water sink into colder regions and rapidly cool. The heat can move a lot more quickly than the salt, though, and since cold water cannot hold as much salt as warmer water, some of the salt precipitates out. That forms the falling crystals scientists observe sinking to the bottom of the lake. (Image and research credit: R. Ouillon et al., source; via Physics World; submitted by Kam-Yung Soh)

Superwalkers
Walking droplets – drops that bounce their way across a pool of the same liquid without coalescing – have fascinated researchers in recent years with their unusual behaviors, some of which mimic quantum phenomena. In a new experiment, researchers vibrate the pool at two frequencies simultaneously, which helps support much larger droplets, known as superwalkers. When the two driving frequencies are close to a harmonic match – like at 80 Hz and just under half that at 39.5 Hz – the droplets will walk, then come to a stop, and then begin walking again. (Image and research credit: R. Valani et al.; via APS Physics; submitted by Justin B and Kam-Yung Soh)

Dissolving Pills
This short film from Macro Room shows how pills dissolve in timelapse. Dissolution is a complex process driven both by flow and chemical concentration. Any small motion in the water helps erode the surface, and as the chemicals dissolve, the subsequent variations in the concentration drive additional flow. This is why we often see a turning point in how quickly the pills come apart. The initial breakdown is slow, but once enough of the pill dissolves, it enhances the surrounding flow, which increases erosion. Notice how many of the pills – liquid-filled capsules, especially – have a point where fluid begins streaming away from them. Unlike the capsules, the solid pills seem to get an extra boost from bubbles that form and then pull away material. (Image and video credit: Macro Room; submitted by clogwog)

Jets from Lasers
Laser-induced forward transfer (LIFT) is an industrial printing technique where a laser pulse aimed at a thin layer of ink creates a tiny jet that deposits the ink on a surface. In practice, the technique is plagued with reproducibility issues, in part because it’s difficult to produce only a single cavitation bubble when aiming a laser at the liquid layer. This is what we see above.
The laser pulse creates its initial bubble just above the middle of the liquid layer. Shock waves expand from that first bubble and quickly reflect off the liquid surface (top) and wall (bottom). When reflected, the shock waves become rarefaction waves, which reduce the pressure rather than increasing it. This helps trigger the clouds of tiny bubbles we see above and below the main bubble.
The effect is worst along the path of the laser pulse because that part of the liquid has been weakened by pre-heating, but impurities and dissolved gases in the liquid layer are also prone to bubble formation, as seen far from the bubble. The trouble with all these unintended bubbles is that they can easily rise to the surface, burst, and cause additional jets of ink that splatter where users don’t intend. (Image and research credit: M. Jalaal et al.; submitted by Maziyar J.)

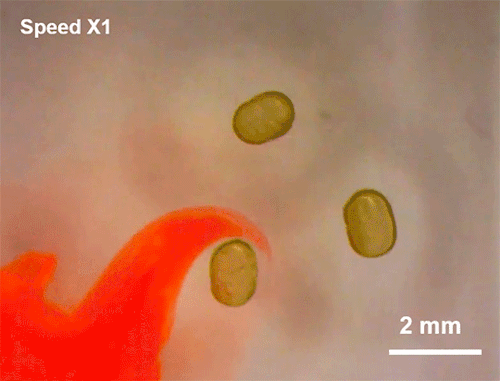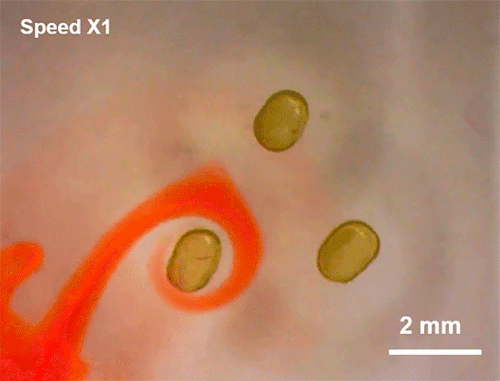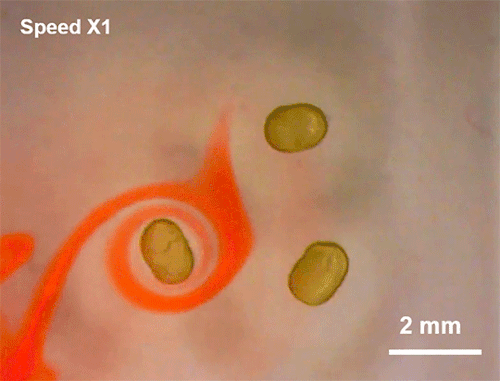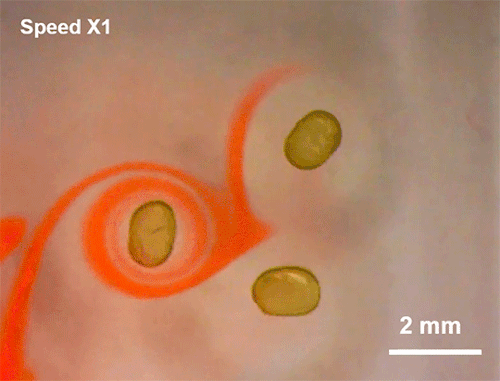
Avoiding Droplet Contact
Cold rain splashing on airplane wings can freeze in instants. To prevent that, researchers look for ways to minimize the time and area of contact a drop has. Hydrophobic coatings and textures can do some of the work, but they are easily damaged and don’t always work well when it comes to freezing.
The new technique shown here uses ring-shaped “waterbowls” to help deflect drops. As the drop impacts and spreads, the walls of the ring texture force the lamella up and off the surface. This reduces both the time and area of contact and, under the right circumstances, cuts the heat transfer between the fluid and surface in half. The technique is useful for more than just preventing freezing, though; it would also be helpful for waterproofing breathable fabrics, where shedding moisture quickly without clogging pores is key to keeping the wearer dry. (Image and research credit: H. Girard et al.; via MIT News and Gizmodo)