Marangoni bursting takes place in alcohol-water droplets; as the alcohol evaporates, surface tension changes across the liquid surface, generating a flow that tears the original drop into smaller droplets. Here researchers add a twist to the experiment using PMMA, an additive that dissolves well in alcohol but poorly in water. As the alcohol evaporates, the PMMA precipitates back out of the water-rich droplet, forming yarn-like strands. (Image and video credit: C. Seyfert and A. Marin)
Tag: physics

Viscosity and Quantum Mechanics
Viscosity describes a fluid’s resistance to changing its shape. Like surface tension, it’s a fundamental property of a fluid that comes from the interactions between molecules. But viscosity is a slippery beast, and especially so for liquids. There is no generic way to calculate a liquid’s thermodynamic properties from quantum dynamical first principles. But that hasn’t stopped theoretical physicists from making progress on deducing the connections between quantum mechanics and liquids.
Although viscosity changes with temperature, all liquids have a minimum viscosity, and those minima are all fairly close to the same value as water’s (excluding any superfluids, which are their own brand of quantum weirdness). Why would liquids share a similar minimum viscosity? Because it turns out the minimum viscosity is quantum! Physicists found that the minimum viscosity is set by an equation depending on Planck’s constant and the mass of an electron — both fundamental constants.
Physicists sometimes like to conjecture about the habitability of the universe if fundamental quantities like Planck’s constant had a different value. This work shows that changing that value would alter water’s viscosity, completely changing the viability of microscopic life! (Image credit: A. Rozetsky; research credit: K. Trachenko and V. Brazhkin; via Physics Today)

Moody Waves
Lines of waves emerge from thick morning fog in this series by photographer Raf Maes. The eerie, slightly surreal images were captured in Venice, near Los Angeles. So often ocean photography features huge, turbulent breaking waves. I find it really neat to see these long, unbroken wave crests appearing from the mist. (Image credits: R. Maes; via Colossal)

The Return of the Ice Disk
Maine’s giant, spinning ice disk is taking shape again. In 2019, it reached about 91 meters across, rotating slowly in the Presumpscot River. How exactly these features form is still a matter of debate, but scientists have worked out a few relevant mechanisms. The spinning of the disk seems to depend on a vortex that forms beneath the ice as melting water sinks. (One of water’s peculiarities is that it’s densest around 4 degrees Celsius, so newly melted water is actually denser than ice. Otherwise ice wouldn’t float!) The plume of sinking water sets up a vortex that drags the ice disk with it as it spins in the river beneath. (Image credit: R. Bukaty/AP; via Gizmodo)

Laser-Induced Jet Break-Up
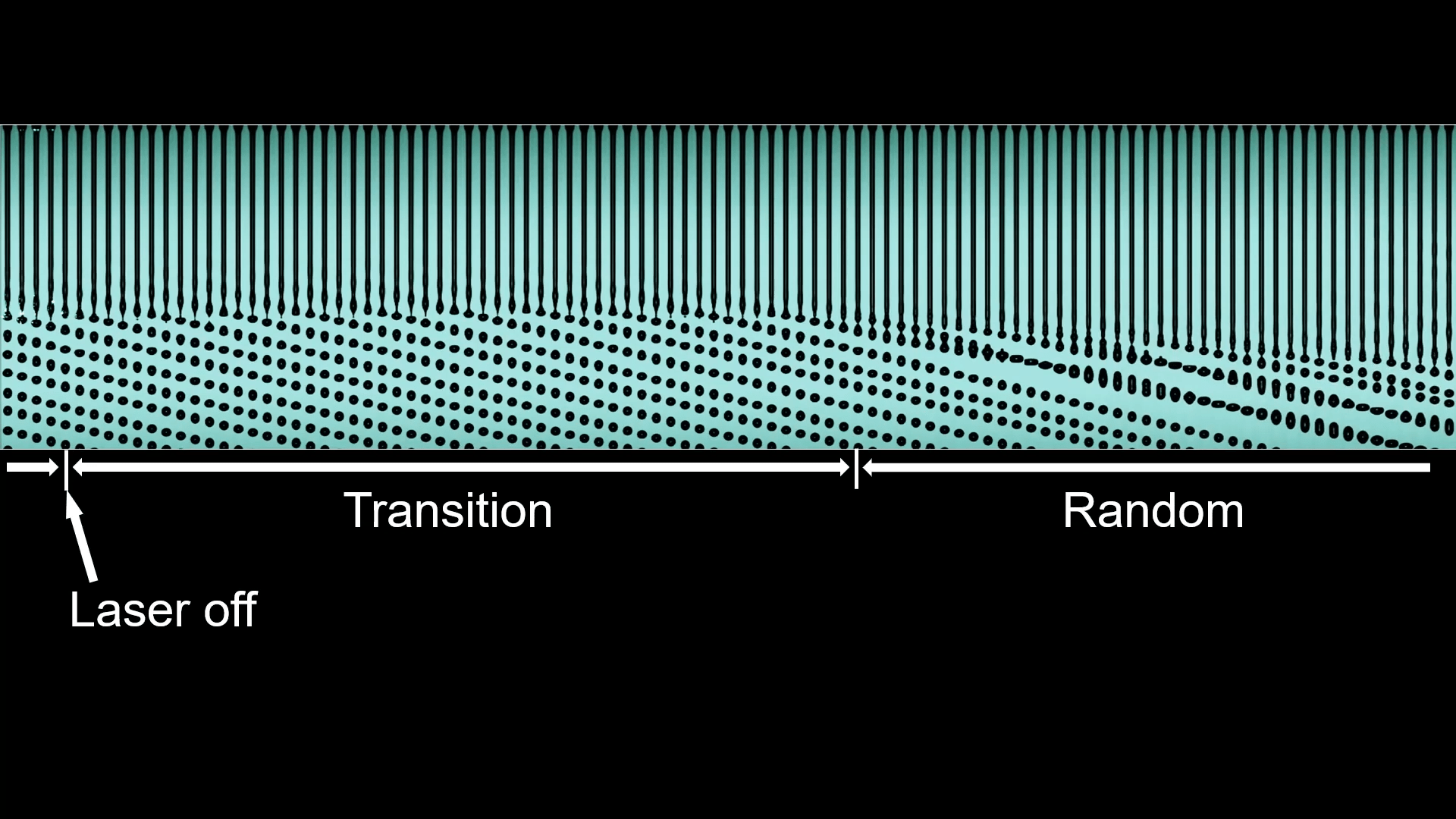
A falling stream of water will naturally break up into droplets via the Plateau-Rayleigh instability. Those droplets are random, unless something like vibration of the nozzle sets their size. In this study, though, researchers found that shining a laser beam on the stream can trigger an orderly break-up with droplets that are consistent in size and spacing.
The optofluidic phenomenon depends on a few different effects. The changing curvature of the liquid stream reflects the laser light, some of which undergoes total internal reflection and travels up the jet as if it were a fiber optic cable. Look closely in the right side of the second image, and you’ll see a periodic flicker of green light at the mouth of the nozzle. Those flashes of green reveal that the liquid jet is guiding the light upstream in bursts, each of which exerts an optical pressure that triggers the Plateau-Rayleigh instability.
When the laser first turns on, there’s a transition period before the orderly break-up begins, and, likewise, turning the laser off triggers a transition from orderly to random (top image). (Image and research credit: H. Liu et al.; via APS Physics; submitted by Kam-Yung Soh)

Volcanic Shocks
A violent underwater eruption at the Hunga Tonga-Hunga Ha’apai caldera on January 15th sent literal shock waves around the world. This animation, based on satellite images from Japan’s Himawari 8, shows the fast-moving shock waves and the growing ash plume coming from the uninhabited island. Although most recent eruptions from this volcano have been small, experts suspect that this latest eruption is part of a major event, similar to the volcano’s last big eruption about 1,000 years ago.
The explosiveness of the eruption comes from the interaction of seawater and fresh magma. When the magma erupts quickly underwater, the hot liquid contacts seawater directly rather than forming a protective layer of vapor (as in the Leidenfrost effect). The resulting explosion tears the magma apart, exposing more hot surfaces to the cold water and further driving the chain reaction. (Image credit: S. Doran/Himawari 8; submitted by jpshoer; see also S. Cronin)

Changing with the Flow
Chemically-reacting flows are some of the toughest problems to unravel. In this new study, researchers found that the very act of flowing through narrow channels can change the speed of chemical reactions. In particular, they found that protein molecules carried through a capillary tube (comparable in size to human capillaries) changed their local shape as a result of the shear forces they experienced. Those changes actually sped up the proteins’ chemical reactions compared to the reaction speed for the chemicals in bulk.
That finding suggests two important takeaways: 1) chemicals may be absorbed in the human bloodstream differently in capillaries than in other parts of the cardiovascular system, and 2) mimicking these tiny capillaries in microfluidic devices could be useful in speeding up certain biochemical reactions. (Image credit: top – KazuN, visual abstract – T. Hakala et al.; research credit: T. Hakala et al.; via Science; submitted by Kam-Yung Soh)


“One Month of Sun”
Get lost in the beauty of our star with Seán Doran‘s film “One Month of Sun”. Constructed from more than 78,000 NASA Solar Dynamics Observatory images, the video shows solar activity from August 2014, particularly the golden coronal loops that burst forth from the sun’s visible surface. These bursts of hot plasma follow the sun’s magnetic field lines, often emerging from sunspots. (Image and video credit: S. Doran, using NASA SDO data; via Colossal)



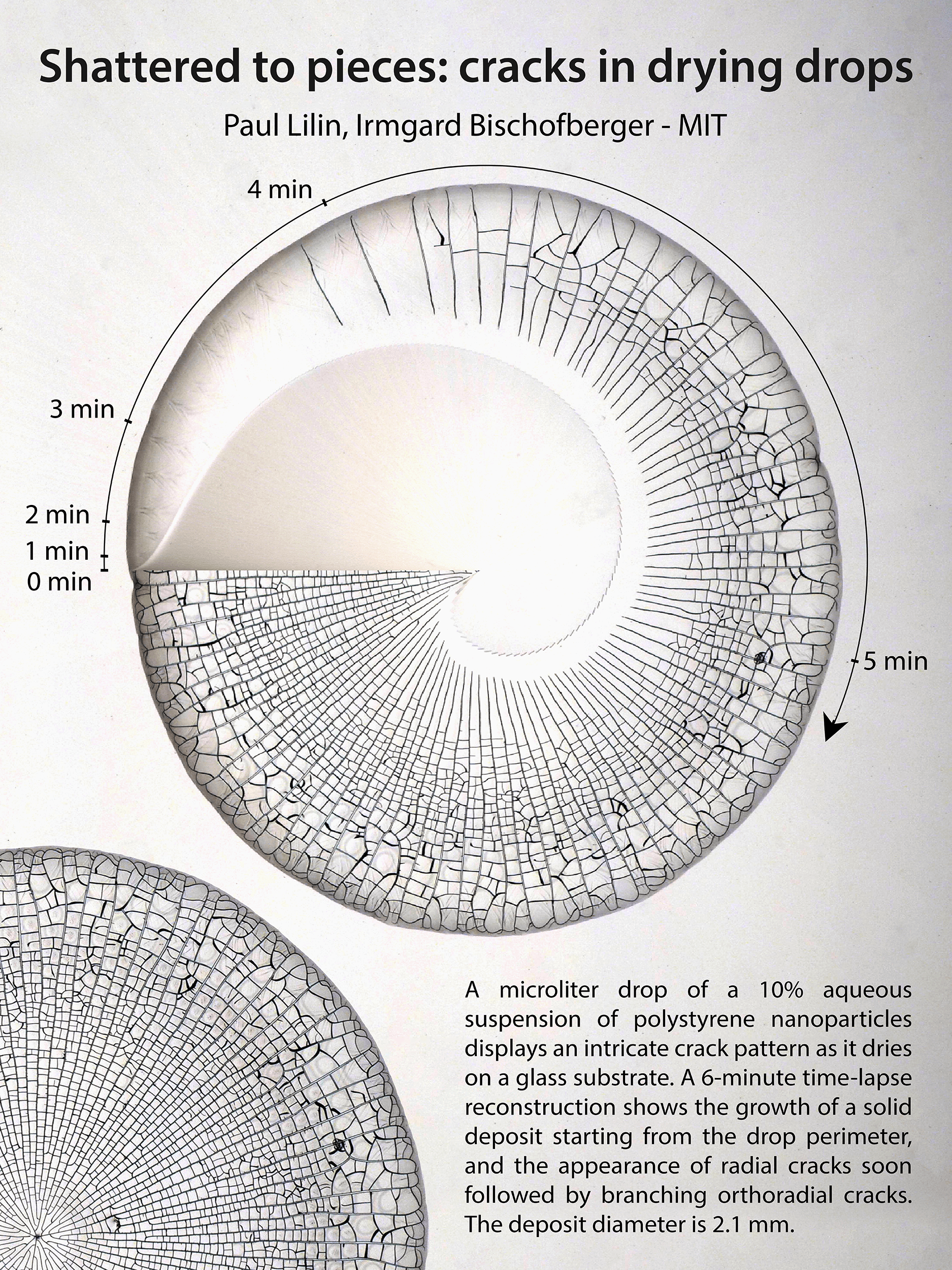
Cracking Droplets
Droplets infused with particles — like coffee — can leave complex stains once they evaporate. Here researchers show the complex cracking pattern that develops as a droplet with nanoparticles evaporates. The central image in the poster actually shows the drop’s pattern changing in time. The initial drop is shown at 9 o’clock, and as you move clockwise around the drop, time passes and the crack structure becomes more complex. What a neat way to visualize the changes! (Image and research credit: P. Lilin and I. Bischofberger)

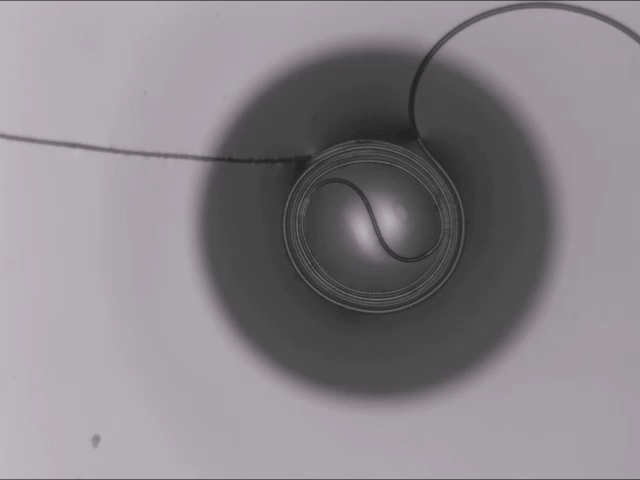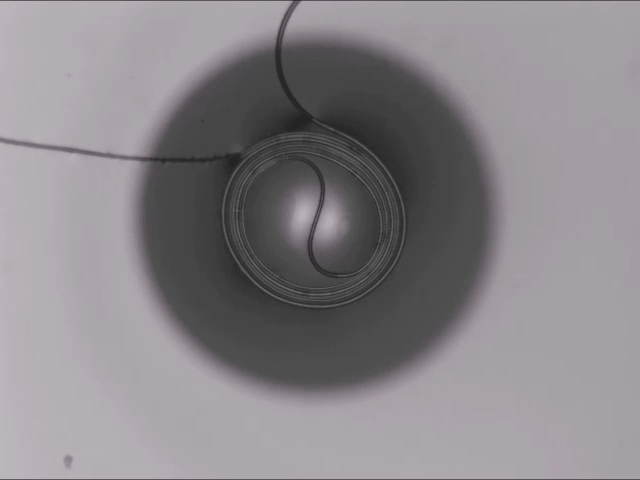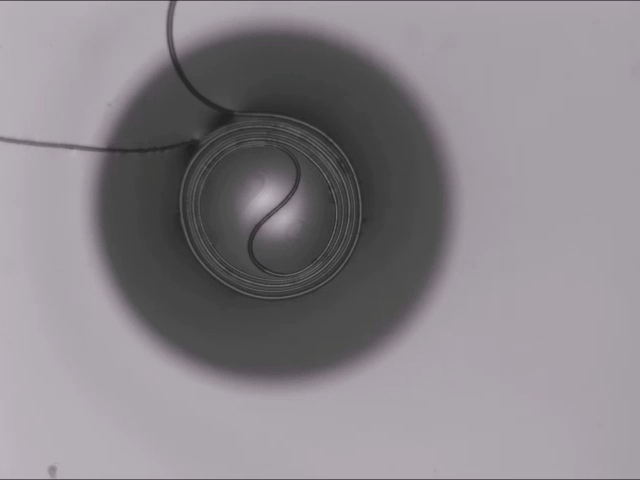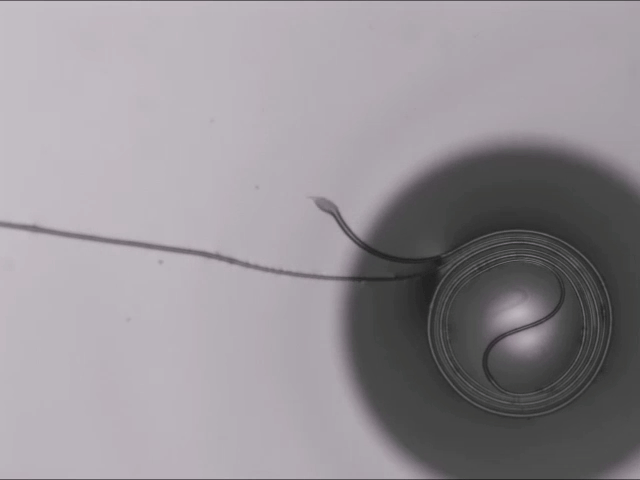
All Wound Up
A thin fiber sitting atop a bubble can spontaneously coil around the bubble thanks to elastocapillarity. (This seemingly bizarre behavior is also why wet strands of hair clump together.) Here’s the situation: The dark circle you see is all bubble; only a portion of the bubble — known as a spherical cap — sticks above the surface of the liquid. When a fiber sits across the top of the bubble, two things can happen: 1) the fiber simply sits there until the bubble bursts, or 2) the fiber starts to bend and wind around the bubble’s cap.
Bending the fiber takes energy. In this case, that bending energy comes from the system as a whole reducing its free energy. The fiber actually sinks into the bubble film in what the researchers call a “bridged” configuration, where the fiber sits inside the liquid film while also touching the air inside and outside the bubble. In this position, the interfacial energy of the fiber-bubble system is lower, leaving enough excess energy savings for the fiber to coil. (Image and research credit: A. Fortais et al.)