In 2013, a meteor about 20-meters in diameter broke up over Chelyabinsk, Russia in a dramatic display that damaged buildings within 100 km and injured more than 1200 people. To better understand the threat presented by such objects, NASA has been conducting 3D, hypersonic simulations like the one shown here. The meteor material is shown in gray and black. Brighter colors like red and yellow indicate the hot, high-pressure shock wave caused when the meteor slams into the atmosphere. Aerodynamic effects quickly erode the meteor, ripping it into pieces that disperse energy explosively in the atmosphere. While you might think the meteor breaking up is good for us, it’s actually the blast waves from its break-up that cause the most damage. (Image and video credit: NASA, source; via Gizmodo)
Category: Research

Schooling in Soap Films
In sports, flocks of birds, and schools of fish, we’re accustomed to thinking that the followers get an aerodynamic or hydrodynamic advantage over the leaders, but this may not always be the case. Here are two flags placed one after another in a soap film flowing from top to bottom. The flags are passive, meaning that their motion is entirely dependent on the flow around them; they cannot exert any resistive force of their own. In this case, scientists observe an effect known as inverted drafting. The lead flag actually experiences less drag – by as much as 50% – than the following flag. This seems to be a result of flow around the second flag having an upstream influence on the motion of the first. (Image and research credit: L. Ristroph and J. Zhang, pdf)

Flow in a Turbine
Fluid flows are complex, complicated, and ever-changing. Researchers use many techniques to visualize parts of a flow, which can help make what’s happening clearer. One technique, shown above, uses oil and dye to visualize flow at the surface. The vertical, black, airfoil-shaped pieces are stators, stationary parts within a turbine that help direct flow. After painting the stator mount surface with a uniform layer of oil, the model can be placed in a wind tunnel (or turbine) and exposed to flow. Air moving around the stators drags some of the oil with it, creating the darker and lighter streaks seen here. Notice how the lines of oil turn sharply around the front of the stator and bunch up near its widest point. Those crowded flow lines tell researchers that the air moves quickly around this corner. (Image credit: D. Klaubert et al., source)

Venturi Splashes

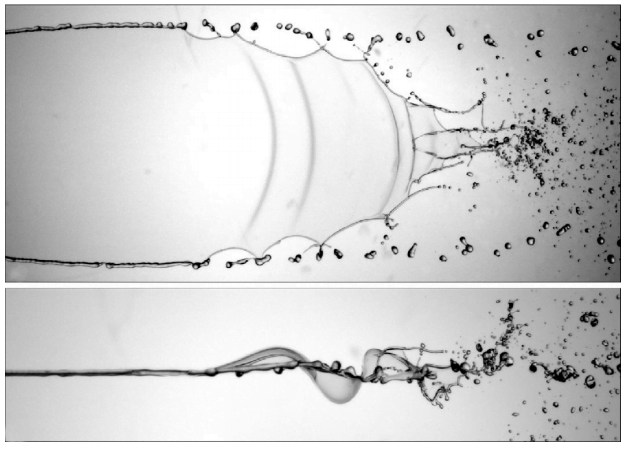
Diving can generate some remarkable splashes. Here researchers explore the splashes from a wedge-shaped impactor. At high speeds, they found that the splash sheet pushed out by the wedge curls back on itself and accelerates sharply downward to “slap” the water surface (top). Studying the air flow around the splash sheet reveals some of the dynamics driving the slap (bottom). The splash sheet quickly develops a kink that grows as the sheet expands. This creates a constriction that accelerates flow on the underside of the sheet. That higher velocity flow means a low pressure inside the constriction, which pulls the thin sheet down rapidly, making it slap the surface. For more, check out the full video. (Image and research credit: T. Xiao et al., source)

The Surge in the Hourglass

When we watch sands running through an hourglass, we think their flow rate is constant. In other words, the same number of grains falls through the neck at the beginning and the end. In many practical granular flows, like those through industrial hoppers (left), this is not the case. Instead, emptying those containers involves a surge near the end where the discharge rate is higher.
The surge is related to the interstitial fluid – the air, water, or other fluid in the space between the grains. On the right, you see an experiment in which brown grains submerged in green-dyed water are emptied. The dark layer is dyed water initially at the top of the grains. As the container drains, that dyed layer moves down more rapidly than the grains; this indicates that the interstitial fluid is actually being pumped by the draining of the grains. Researchers think this is an important factor affecting the final surge. (Image credits: hopper – T. Cizauskas; discharge graph – J. Koivisto and D. Durian, source; research credit: J. Koivisto and D. Durian; submitted by Marc A)

Reconnecting
Vortices are a common feature of many flows. Here we see a helical vortex tube spinning in a swirling flow. The vortex itself is visible thanks to air trapped in its low-pressure core. As the vortex spins, two sections of it come together. This results in what’s known as vortex reconnection: the vortex lines break apart and rejoin in a new configuration – as a small independent vortex ring and a shorter section of helical vortex. Events like this are common but usually hard to observe directly. They’ve been previously visualized using vortex knots and have even been sighted in the quantum vortices of superfluid helium. (Image credit: S. Skripkin, source; research credit: S. Alekseenko et al., pdf)

Breaking Up
Liquid sheets break down in a process known as atomization. Above are top and side views of a liquid sheet created by two identical liquid jets impacting head-on. The jets themselves are off-screen to the left. Their collision generates a thin sheet of liquid that flows from left to right. In the center of the images, the sheet has begun to flap and undulate, shedding large droplets from its edges as it does. At the far end of the sheet, much finer droplets are sprayed out from the center as the sheet collapses completely. This is an example of an instability in a fluid. Initially, any disturbance in the liquid sheet is extremely tiny, but circumstances in the flow are such that those disturbances gather energy and grow larger, creating the large undulations. Those undulations are unstable as well and kick off a fresh set of disturbances that grow until the flow completely breaks down. (Image credit: N. Bremond et al., pdf)

The Japanese Pufferfish
[original media no longer available]
If you’ve ever dived or snorkeled over a sandy lake or ocean bottom, you’ve probably seen some neat patterns there. But it’s hard to compete with the Japanese pufferfish for pure artistry. This small fish creates enormous and elaborate designs in the sand in order to attract a mate. The male fish moves the sand into place by flapping his fins very close to the surface. Above a critical flapping velocity, his fins generate vortices capable of picking up sand, as seen below. With repeated passes, the fish is able to excavate the trough that is key to his creation. It’s a constant fight against the current, though.
Puffers aren’t the only ones who flap their fins to move the sands. Rays and flounders use this technique to bury themselves and hide (Video credit: BBC Earth; image credit: A. Sauret, source; research credit: A. Sauret et al.)

A Real Tatooine

Since at least the release of “Star Wars”, we have wondered what life would be like on a circumbinary planet – a planet orbiting two stars. In the past few decades, we have discovered several such planets, but we are still in the early days of modeling the climate of these worlds. One recent study uses the stars of the Kepler 35 system, which are only slightly less luminous than our sun, to explore the climate of an Earth-like water planet.
According to the study, this fictional planet would maintain Earth-like habitability at a distance of 1.165–1.195 astronomical units from its suns’ center of gravity – just a little further out than our own orbital distance. Variables like the planet’s mean global surface temperature and precipitation vary with two distinct periods – the time required for the stars to orbit one another and the time it takes for the planet to orbit its stars. Both factors affect how much sunlight the planet receives. The planet’s climate response to these changes is complex and varies depending on location, but the overall variations observed in the climate are small. It does show, however, that places like Tatooine don’t have to be desert planets! (Image credit: Tatooine – Star Wars; Kepler 35 system – L. Cook; research credit: M. Popp and S. Eggl)

Sorting by Bubble

Microfluidic devices, also known as labs-on-a-chip, require clever techniques for processes like sorting particles by size. One such technique uses an oscillating bubble to sort particles. When the bubble vibrates back and forth (left) it creates what’s known as a streaming flow – large regions of recirculation (shown as gray ellipses in the right image). If the bubble is placed inside a channel, we say that two flows have been superposed; the device combines both the left-to-right flow of the channel and the recirculating streaming flow.
Introduce a micron-sized particle into this combined flow, and it will get carried to the bubble and then bounced around by its effects (left). In fact, the larger the particle is, the more the bubble deflects it relative to the flow. You can see this in the image on the right as well. Here the frame rate has been matched to the bubble’s vibration, so the bubble appears stationary, and the particle paths look smooth. The gray lines show the fluid’s path, and individual solid particles are introduced at the left. The largest particle gets strongly deflected as it passes the bubble and exits at the top-right. A fainter, smaller particle follows after it. Being smaller, the bubble’s deflection on it is weaker, and this second particle exits along a path to the center-right. The result is a fast and simple method for particle sorting. (Image and research credit: R. Thameem et al., source)