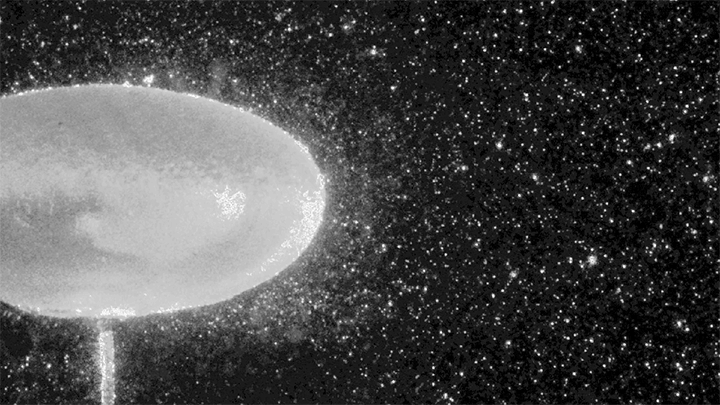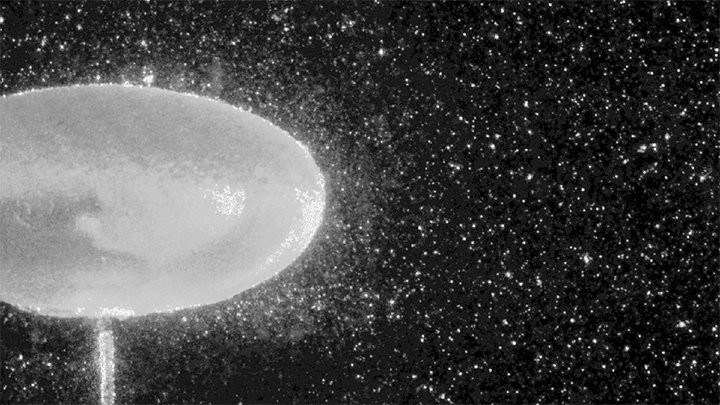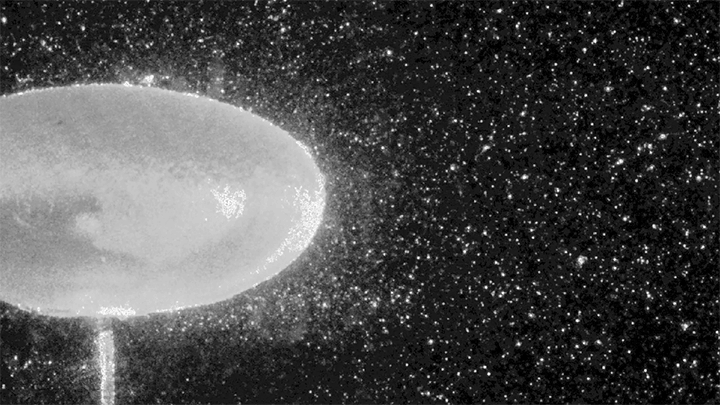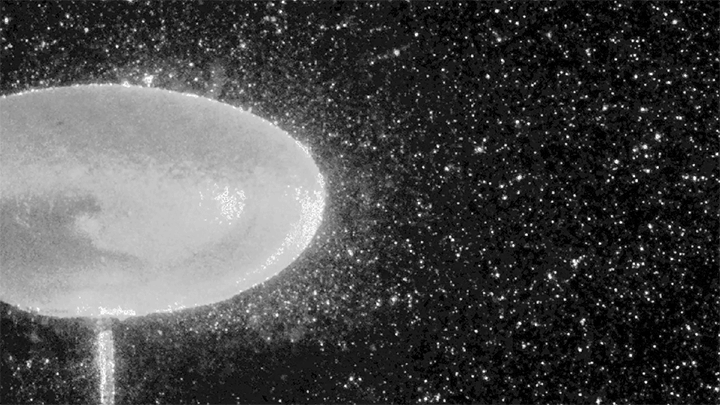
Hydrogen bubbles rise off zinc submerged in hydrocholoric acid in this short film from the Beauty of Science team. In high-speed video, the rise of the bubbles is stately and mesmerizing. Notice how the smallest bubbles appear as perfect spheres; for them, surface tension is strong enough to maintain that spherical shape even against the viscous drag of their buoyant rise. Larger bubbles, formed from mergers both seen and unseen, have a harder time staying round. In them, surface tension must battle gravitational forces and drag from the surrounding fluid. (Image and video credit: Beauty of Science; via Laughing Squid)
Month: January 2020

Tapping a Can Won’t Save Your Beer
It happens to the best of us: sometimes our beer gets shaken up during transit. One common reaction to this is to tap the side of the can repeatedly before opening, but a new scientific study shows that tapping doesn’t affect the volume of beer lost. Danish scientists tested over 1,000 cans of beer in randomized combinations of shaken, unshaken, tapped, and untapped, and observed no difference between tapped and untapped cans.
The foam-up upon opening takes place in shaken beer because carbon dioxide bubbles form in the pressurized beer, especially along defects in the wall where bubbles can nucleate. When the pressure is released, the carbon dioxide becomes supersaturated and comes out of solution, especially into the pre-formed bubbles, which rapidly grow and overflow. In theory, tapping could disturb those bubbles before opening, but in practice, it makes no difference. Your best bet? Give the beer time to settle before you open it. (Image credit: Q. Dombrowski; research credit: E. Sopina et al.; via Ars Technica)

The Physics of Al Dente
It’s a simple weeknight routine: toss a handful of spaghetti noodles in boiling water, wait a few minutes, and enjoy with the sauce of your choice. But there’s a surprising amount of physics in the humble strand of spaghetti, and a new model focuses on the way spaghetti sags and curls as it cooks.
Spaghetti, like most pastas, is made of semolina flour mixed with water, extruded (in commercially produced spaghetti), and then dried. Once immersed in water, the rod of pasta begins to swell and soften as water works its way slowly inward. At the same time, it will lose some of its starches to the surrounding water. If the water is hot enough, the pasta undergoes an additional process, starch gelatinization, which is responsible for cooked pasta’s characteristic texture. That perfect al dente condition occurs right as the hydration front reaches the pasta’s core.
As all of this happens, the initially straight spaghetti strand sags, settles, and curls. Researchers found that, even with a relatively simple model that assumes spaghetti doesn’t stick to the pot, they could capture shape change of individual spaghetti strands, suggesting it’s possible to identify perfectly cooked pasta by shape alone. (Image credit: Pixabay; research credit: N. Goldberg and O. O’Reilly; via Ars Technica)

Swinging Jets
In the tiny realm of microfluidics, flows are, in general, completely laminar. That makes mixing a challenge. But it turns out that pumping water steadily into multiple inlets can spontaneously generate oscillations between the jets, allowing dramatic mixing even at low Reynolds numbers. Two inlets in a parallel channel (first image) oscillate steadily over a small range of conditions, but widening the channels (second image) allows the jets to switch back and forth over a larger range. And adding additional inlets (third image) can create even more complex fluid oscillators! (Image, video, and research credit: A. Bertsch et al.)

River Avon
One of the challenges in fluid dynamics is considering the instantaneous versus the average. Many flows — especially turbulent ones — are different at every point in space and in time. That’s a lot of data to collect and to wrap one’s head around. So often researchers will average turbulent measurements over a period of time and break that information down into two variables: an average velocity and a fluctuating one.
What does that have to do with this image? Well, by capturing the River Avon’s flow near Pulteney Bridge as a long exposure, photographer Peter Leadbetter gives us a look at the river’s “averaged” flow. The long exposure smooths out some of the intermittent features visible in a faster picture, and instead draws our attention to the overall path of the flow and regions that may behave differently, like those near the wall in the foreground. The averaging researchers do is much the same. It will erase or obscure some features while making the large-scale patterns more obvious. (Image credit: P. Leadbetter; submitted by Ioanna S.)

Flowery Splashes
Plunge a disk into water and you’ll get a dome-like splash that closes back on itself. But what happens when that disk has a patterned surface? In this video, researchers added a wedge-like surface pattern to the disk, creating a splash with petals like a flower. Just as the surface of disk is about to submerge completely, a jet of the remaining air spurts out the trough of each wedge. This air jet breaks up the tip of the triangular splashes focused by the wedge. (Image, research, and video credit: H. Kim et al.)

Testing Waves in High Gravity
Where waves crash and meet, turbulence is inevitable. But exactly how large waves interact — whether in the ocean, in plasma, or the atmosphere — is far from understood. A new experiment is teasing out a better physical understanding by tweaking a variable that’s been hard to change: gravity.
To do so, the researchers conduct their experiments in a large-diameter centrifuge (shown above) where they can create effective gravitational forces as high as 20 times Earth’s gravity. This increases the range of frequencies where gravity-dominated waves occur by an order of magnitude.
By studying this extended frequency range, the authors found something unexpected: the timescales of wave interactions did not depend on wave frequency, as predicted by theory. Instead, those interactions were dictated by the longest available wavelength in the system, a parameter set by the size of the container. It will be interesting to see if future work can confirm that result with even larger containers. (Image credit: ocean waves – M. Power, others – A. Cazaubiel et al.; research credit: A. Cazaubiel et al.; via APS Physics; submitted by Kam-Yung Soh)

Spinning Ink Out of Markers
I have to say I’m grateful that my classmates in school never discovered the mess-generating superpower of felt-tipped markers. As the Slow Mo Guys demonstrate here, when you spin or fling these markers, ink will stream out of them. That’s due, in part, to the air vents present near the tips. Markers (and other pens) have those to equalize the pressure between the outside and the ink reservoir; otherwise, the ink won’t flow to the felt tip as it should. Is anyone else surprised by the sheer volume of liquid ink apparently contained in these pens? (Image and video credit: The Slow Mo Guys)

“Viscoelasticity Gives You Wings!”
What happens when you drop a hydrogel bead on a water droplet? Because of the hydrogel’s viscoelasticity and its hydrophilic nature, the rebounding bead carries the droplet with it. As seen in the video above, when the impact energy is small enough, the droplet forms a reverse crown during lift-off, kind of like giving the hydrogel bead a skirt. The key feature for lift-off is the bead’s deformation on impact. Because the hydrogel widens at its base, it is sometimes able to push the entire droplet off its initial footprint and detach it from the surface. (Image, research, and video credit: R. Rabbi et al.)

Self-Assembly Under Stratification
Sometimes mistakes lead to great discoveries. After leaving a failed outreach demo overnight, researchers discovered a new mechanism for self-assembling particles. In the initial set-up, a layer of fresh water is poured atop a layer of denser, saltier water. This creates what’s known as a stably stratified fluid, with progressively denser mixtures of salt water as one moves downward. If you pour in particles of an intermediate density (heavier than fresh water and lighter than salt water), they’ll form a layer at one height, and, if you wait overnight, those particles will slowly form a disk-like raft.

This self-assembly is driven by fluid dynamics — not by any attraction between the particles. Because the particles are unable to absorb salt, their boundaries distort the concentration gradients in the surrounding fluid. This generates subtle currents at the particle boundaries, like in the picture above, where flow moves toward the particle at the equator and away at the poles. Larger particle clusters generate stronger flows, allowing them to attract even more particles.
Although the speeds involved are quite slow, this mechanism may play an important role in nature, where stratified flows are common. The researchers speculate, for example, that the effect could be important in the clustering of microplastics in the ocean. (Image and research credit: R. Camassa et al.; see also R. McLaughlin; submitted by Kam-Yung Soh)