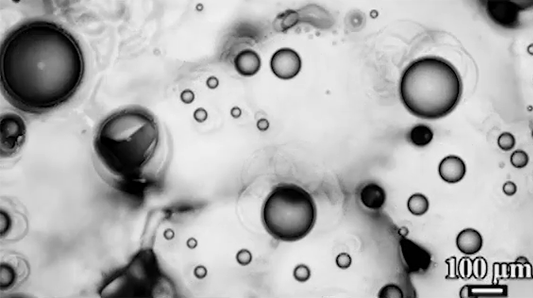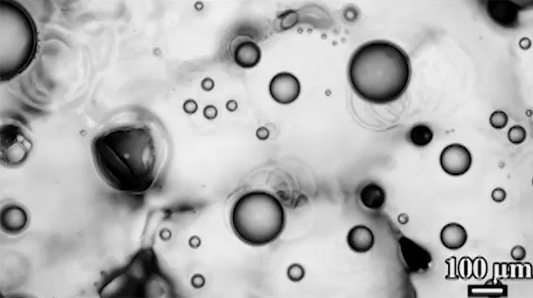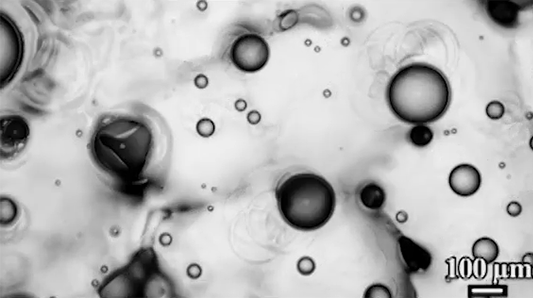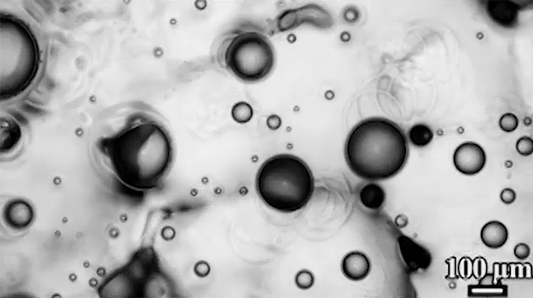
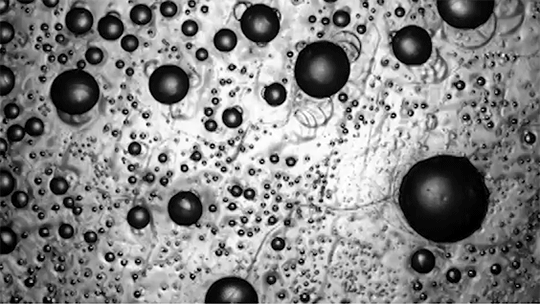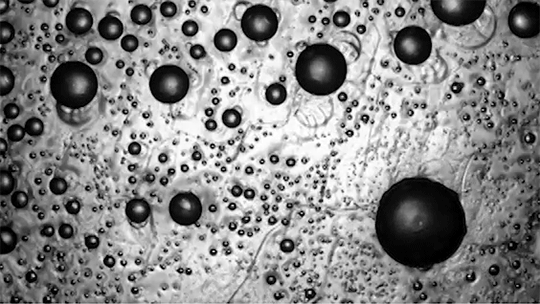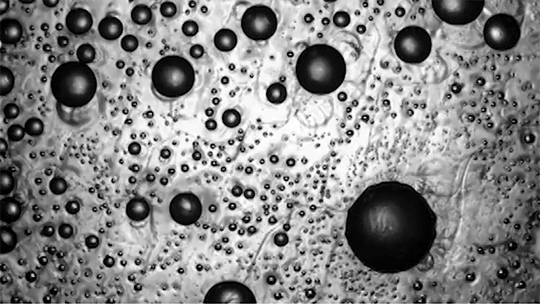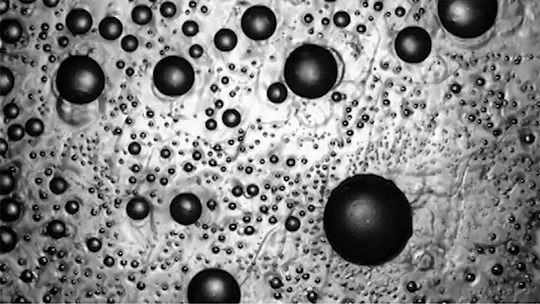
Preventing ice and frost from forming on surfaces – especially airplane wings – is a major engineering concern. The chemical de-icing cocktails currently used in aviation are a short-lived solution, and while superhydrophobic surfaces can be helpful, they tend to be easily damaged and therefore impractical. Another possible solution, shown here, are so-called phase-switching liquids – substances like cyclohexane that have freezing points higher than that of water. This means that they form a solid coating near the freezing temperature of water.
Water droplets on these coatings move in a random stick-slip walk (above) but they tend not to freeze. This is because freezing requires the droplets to release heat, which melts part of the phase-switching liquid. Now, instead of solidifying to the surface, the droplet moves on a film of the phase-switching liquid. Re-freezing that liquid is tough because it’s thermodynamically unfavorable, and the smoothness of the liquid layer makes it harder for ice to find a nucleation point. In lab tests, the phase-switching liquid surfaces resisted ice and frost more than an order of magnitude longer than conventional materials. (Image and research credit: R. Chatterjee et al.; video credit: Univ. of Illinois at Chicago; submitted by Night King)