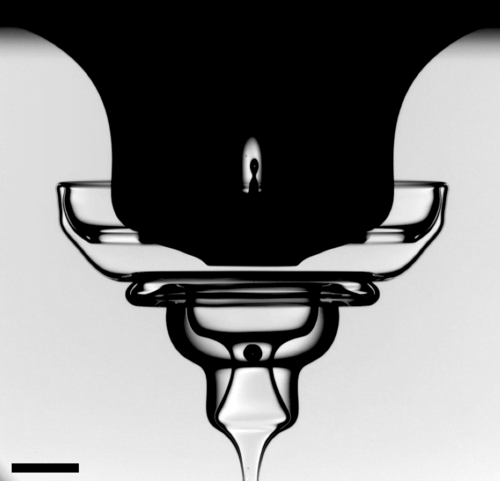
Conventional wisdom – and the Second Law of Thermodynamics – require all fluids to have viscosity, with the noted and bizarre exception of superfluids, which can flow with zero viscosity. In essence, you cannot have work (i.e. flow) for free. Some effort has to be lost to resistance.
But scientists have discovered, bizarrely, that adding bacteria to water can result in zero or even negative viscosities – meaning that effort is required to keep the flow from accelerating. Before you ask, no, this is not a recipe for a perpetual motion machine. What happens when the bacteria-filled fluid is sheared is that the bacteria align and start collectively swimming. The local effects of each bacteria combine en masse to create a fluid that seemingly flows on its own. In the end, though, it’s the bacteria that are supplying that work. It certainly raises interesting prospects, though, for harnessing the power of bacterial superfluids. See the links below for more. (Image credit: M. Copeland, source; research credit: S. Guo et al., A. Loisy et al.; via Quanta; submitted by Kam-Yung Soh)