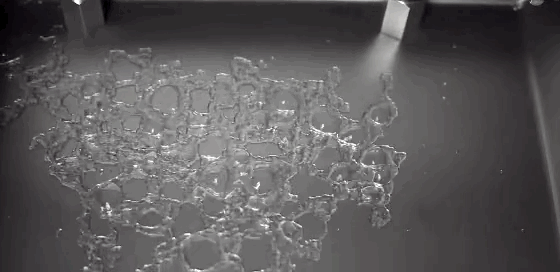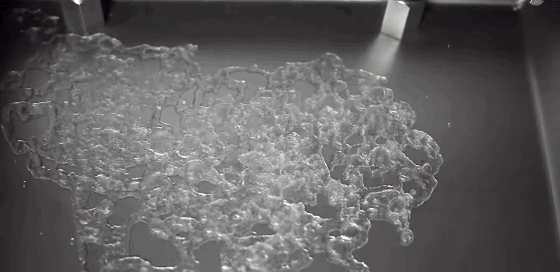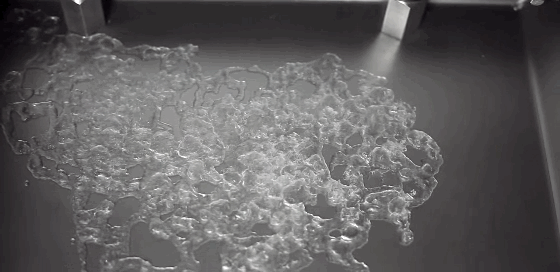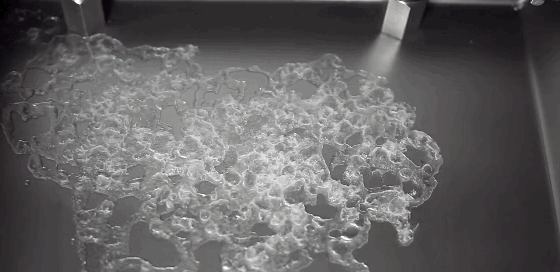Much of the rain that falls on Earth began as snow high in the atmosphere. As it falls through warmer layers of air, the snowflakes melt and form water droplets. The details of this melting process have been difficult to capture experimentally, but a new computational model may provide insight. The basic process has a couple stages. As snow begins to melt, surface tension draws the water into concave areas nearby. When those regions fill up, the water flows out and merges with neighboring liquid, forming water droplets around a melting ice core.
Although this same sequence was observed for many types of snow, scientists also observed some important differences between rimed and unrimed snowflakes. Rime forms when supercooled water droplets freeze onto the surface of a snowflake. Lightly rimed snow still looks light and fluffy, like the animation above, but heavily rimed snow forms denser and more spherical chunks. Because there are lots of porous gaps in heavily rimed snow, water tends to gather there during initial melting. Rimed snow was also more likely to form one large water droplet rather than breaking into multiple droplets like snow with less rime. For more, check out NASA’s video and the Bad Astronomy write-up. (Image credit: NASA, source; research credit: J. Leinonen and A. von Lerber; via Bad Astronomy; submitted by Kam Yung-Soh)