Spreading paint with a brush or with fingers is familiar activity for most people. It’s also similar to processes used in industry for spreading thin layers of paint and other complex fluids. In a recent study, researchers took a look at how a soft, elastic blade (similar to a paintbrush or one’s fingers) spreads shear-thinning fluids (like paint) and Newtonian fluids (like water). Surprisingly, they found that it actually takes 30% more mechanical work to spread a shear-thinning fluid than the same volume of an equivalent Newtonian one. That’s pretty much the opposite of what we’d expect since the action of spreading (and shearing) the complex fluid should reduce its viscosity. However, they did find that the shear-thinning fluid spreads to a thin layer more consistently than the Newtonian fluid does. (Image credit: A. Kolosyuk; research credit: M. Krapez et al.)
Search results for: “shear thinning”

Beijing 2022: Why Are Ice and Snow Slippery?
Although every Olympic winter sport relies on the slippery nature of snow and ice, exactly why those substances are so slippery has been an enduring mystery. Michael Faraday hypothesized in the nineteenth century that ice may have a thin, liquid-like layer at its surface, something that modern studies have repeatedly found.
One recent study used an entirely new instrument to probe the characteristics of this lubrication layer and found that it is only a few hundred nanometers thick. But the fluid in this layer is nothing like the water we’re used to. Instead it has a viscosity more akin to oil and its response to deformation is shear-thinning and viscoelastic, more like the complex fluids in our kitchens and bodies than pure, simple water. They found that using a hydrophobic probe modified the interfacial viscosity even further, which finally provides a hint at the mechanism behind waxing skis and skates.
Fortunately for us, we’ve found plenty of ways to employ and enjoy water’s slipperiness, even as the mystery of it slowly gives way to understanding. (Image credit: M. Fournier; research credit: L. Canale et al.; via Physics World; submitted by Kam-Yung Soh)

Studying Active Polymers Using Worms
I’ve covered some odd studies in my time, but this might be the strangest: to understand how active polymers affect viscosity, researchers loaded drunk worms into a rheometer. Active polymers are long-chain molecules that, like worms, can move on their own using stored energy or by extracting energy from their surroundings. Their dynamics are tough to study, though, because individual polymers are almost impossible to observe while a suspension of them is being deformed.
Enter the humble sludge worm. Often sold as fish food, these worms — like the polymers they’re meant to imitate — are individually quite wiggly but, given their size, are far easier to observe. Researchers placed them in a custom rheometer in a solution of water and observed how the worm mass responded when sheared by a spinning top plate (Image 3). Like active polymers, the worms exhibited shear-thinning; the faster the plate spun, the lower the worms’ viscosity, likely because the additional force helps align the worms.
But how do active worms compare with passive ones? The obvious solution would be to repeat their tests with dead worms, but the researchers found a more humane method: by adding some alcohol to the water, they temporarily reduced the worms’ activity, allowing them to compare active and passive worms (Image 2). Once rinsed in water, the worms sobered up and returned to their normal activity levels.
The researchers found that both the active and passive worms exhibited shear-thinning as the force on them increased, but the shear-thinning in the active worms was not as pronounced, presumably because the movements of individual worms prevented them from aligning smoothly. (Image and research credit: A. Deblais et al.; via Gizmodo and APS Physics)

Viscoelasticity and Liquid Armor
One proposed method for improving bulletproof armor is adding a layer of non-Newtonian fluid that can help absorb and dissipate the kinetic energy of impact. Thus far researchers have focused on shear-thickening fluids – like cornstarch-based oobleck – filled with particles that jam together if anything tries to deform them quickly. But is it really the shear-thickening properties that matter for high-speed impacts?
To test this, researchers studied projectile impact on three fluids: water (left), a cornstarch mixture (not shown), and a shear-thinning polymer mixture (right). Water is Newtonian, and it slows down the projectile but doesn’t stop it. Both the shear-thickening cornstarch and the shear-thinning polymer mixture do stop the projectile. And by modeling the impacts, researchers concluded that the key to that energy dissipation isn’t their shear-related behaviors: it’s the fact that both fluids are viscoelastic.
That means that these fluids show both viscous (fluid-like) and elastic (solid-like) responses depending on the timescale of an impact. The high speed of the impact triggered a strong viscous response in both fluids, bringing the projectile to a halt. And if, as the researchers suggest, it’s a fluid’s viscoelasticity that matters most, that widens the field of candidates when it comes to developing a fluid-based armor. (Image and research credit: T. de Goede et al.)

The Kaye Effect
Allow a stream of shampoo to fall into a pile and you’ll catch a glimpse of the bizarre Kaye effect. A jet of shampoo will briefly rise up before becoming chaotic and falling. The key to this behavior is the shear-thinning of the shampoo. When the shampoo is just sitting on a surface, it’s quite viscous, but slide your hand across it, and the shampoo will become much less resistant to flowing.
When the jet of falling shampoo hits the pile, it creates a little dimple. Sometimes the incoming jet hits that dimple and slips along it, thanks to a sudden decrease in viscosity. That can send an outgoing jet of shampoo riding off the dimple like a ramp. As the dimple deepens, the outgoing streamer rises up until it hits the incoming jet and becomes unstable. The shampoo streamer collapses, only to be restarted when a new dimple forms. (Image and video credit: S. Mould; h/t to Guillaume D.)


Chocolate Fountain
Amidst your holiday celebrations, you may have encountered a chocolate fountain. In a recent paper, applied mathematicians have laid out the physics behind these delicious decorations, and it turns out they are an excellent introduction to many fluids concepts. Molten chocolate is a mildly shear-thinning, non-Newtonian fluid, meaning that it becomes less viscous when deformed. This adds a wrinkle to the mathematics describing the flow, but only a little one. The researchers divide the flow into three regimes: pipe flow driving the chocolate up the inside of the fountain, thin-film flow over the fountain’s domes, and, finally, the curtain of falling chocolate where foodstuffs are dipped. The final regime is the most mathematically challenging and may be the most fascinating. The authors found that the free-falling curtain of liquid pulls inward as it falls due to surface tension. Their paper is quite approachable, and I recommend those of you with mathematical inclinations check it out. (Image credit: P. Gorbould; research credit: A. Townsend and H. Wilson)

The Kaye Effect
Those who have poured viscous liquids like syrup or honey are familiar with how they stack up in a rope-like coil, as shown in the top row of images above. What is less familiar, thanks to the high speed at which it occurs, is the Kaye effect, which happens in fluids like shampoo when drizzled. Shampoo is a shear-thinning liquid, meaning that it becomes less viscous when deformed. Like a normal Newtonian fluid, shampoo first forms a heap (bottom row, far left). But instead of coiling neatly, the heap ejects a secondary outgoing jet. This occurs when a dimple forms in the heap due to the impact of the inbound jet. The deformation causes the local viscosity to drop at the point of impact and the jet slips off the heap. The formation is unstable, causing the heap and jet to collapse in just a few hundred milliseconds, at which point the process begins again. (Image credit: L. Courbin et al.)

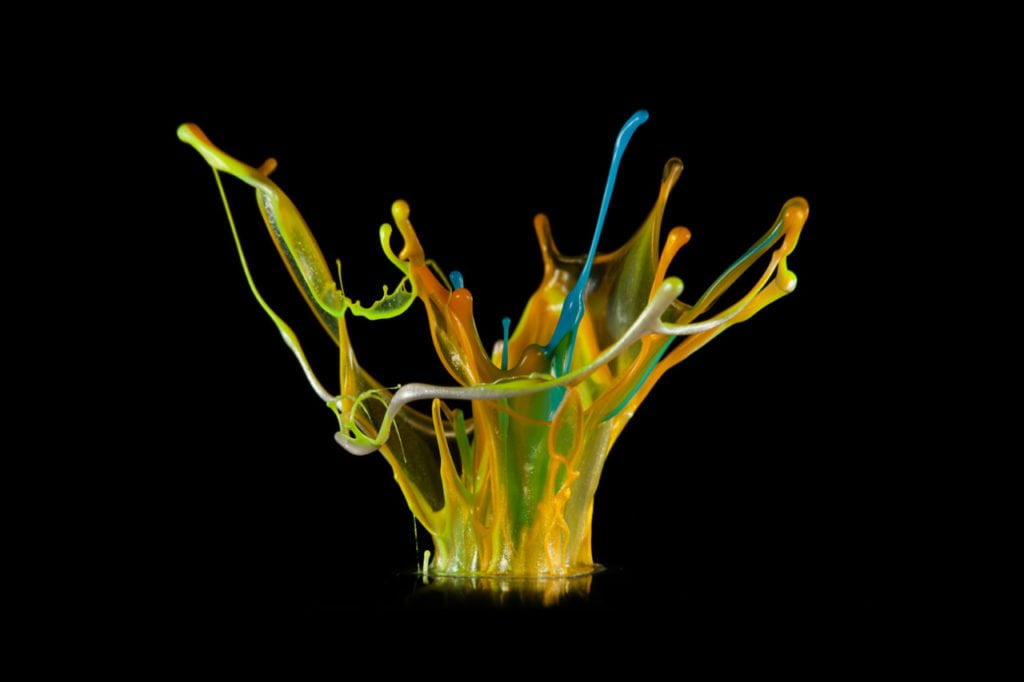
Paint on Speakers
Paint seems to dance and leap when vibrated on a speaker. Propelled upward, the liquid stretches into thin sheets and thicker ligaments until surface tension can no longer hold the the fluid together and droplets erupt from the fountain. Often paints are shear-thinning, non-Newtonian fluids, meaning that their ability to resist deformation decreases as they are deformed. This behavior allows them to flow freely off a brush but then remain without running after application. In the context of vibration, though, shear-thinning properties cause the paint to jump and leap more readily. For more images, see photographer Linden Gledhill’s website. (Photo credit: L. Gledhill; submitted by pinfire)

4th Birthday: The Kaye Effect
Today’s post continues my retrospective on mind-boggling fluid dynamics in honor of FYFD’s birthday. This video on the Kaye effect was one of the earliest submissions I ever received–if you’re reading this, thanks, Belisle!–and it completely amazed me. Judging from the frequency with which it appears in my inbox, it’s delighted a lot of you guys as well. The Kaye effect is observed in shear-thinning, non-Newtonian fluids, like shampoo or dish soap, where viscosity decreases as the fluid is deformed. Like many viscous liquids, a falling stream of these fluids creates a heap. But, when a dimple forms on the heap, a drop in the local viscosity can cause the incoming fluid jet to slip off the heap and rebound upward. As demonstrated in the video, it’s even possible to create a stable Kaye effect cascade down an incline. (Video credit: D. Lohse et al.)

Why Ketchup is Hard to Pour
Oobleck gets a lot of attention for its non-intuitive viscous behaviors, but there are actually many non-Newtonian fluids we experience on a daily basis. Ketchup is an excellent example. Unlike oobleck, ketchup is a shear-thinning fluid, meaning that its viscosity decreases once it’s deformed. This is why it pours everywhere when you finally get it moving. Check out this great TED-Ed video for why exactly that’s the case. In the end, like many non-Newtonian fluids, the oddness of ketchup’s behavior comes down to the fact that it is a colloidal fluid, meaning that it consists of microscopic bits of a substance dispersed throughout another substance. This is also how blood, egg whites, and other non-Newtonian fluids get their properties. (Video credit: G. Zaidan/TED-Ed; via io9)