Droplets of acetone deposited on a bath of warm water can float along on a Leidenfrost-like vapor layer. The droplets are self-propelling, too, thanks to interactions between the acetone and water. Acetone can dissolve in water, and when acetone vapor beneath the drop gets absorbed into the water bath, it lowers the local surface tension. That drop in surface tension creates a pull in the direction of a higher surface tension; this is what is known as the Marangoni effect. Because of that flow in the direction of higher surface tension, the acetone drop accelerates away. (Image credit: S. Janssens et al., source)
Category: Research

Using Jets to Find Food
Archer fish are well-known for their ability to hit aerial targets with perfectly aimed jets of water, as we’ve discussed previously. But a new study shows they use a similar technique to form underwater jets that help them uncover food. The researchers found that the fish altered the timing of their jet formation based on the type of substrate – fine sand, course sand, or mud – that the food pellet was hidden in. A great next step in this research would be using a technique like particle image velociometry (PIV) to measure the flow field directly and see to what extent the fish’s actions are altering the jet they produce. (Image and research credit: J. Dewenter et al.; GIF source: freshphotons)

Titan’s Bubbly Islands
Titan, Saturn’s largest moon, is a fascinating world with remarkable similarities to our own. It is the only other world we know of with stable bodies of liquid at its surface. Unlike Earth, frigid Titan’s lakes and seas are filled with methane and ethane. Radar data from the Cassini mission has shown oddly changing shorelines on Titan, above, with islands that seem to magically appear and disappear over time.
Researchers at NASA’s Jet Propulsion Laboratory now think these islands may, in fact, be bubbles. As Titan’s lakes cool, they’re better able to absorb nitrogen gas, but when temperatures warm up, that gas comes out of solution and floats to the surface, much like the bubbles of carbon dioxide in a soda. If this hypothesis holds up, there are some interesting implications for Titan’s atmosphere. Here on Earth, bubbles popping in the ocean are a major source of aerosol particles. It’s possible migrating rafts of bubbles could behave similarly on Titan. (Photo credit: NASA/JPL-Caltech/ASI/Cornell; submitted by jpshoer)
I’m excited to announce I will be visiting JPL later this month (March 30th), where I have the honor of giving a Women’s History Month talk. If there are any JPLers who are FYFD fans, I hope to see you there. Be sure to RSVP to the ACW luncheon by the March 24th deadline.

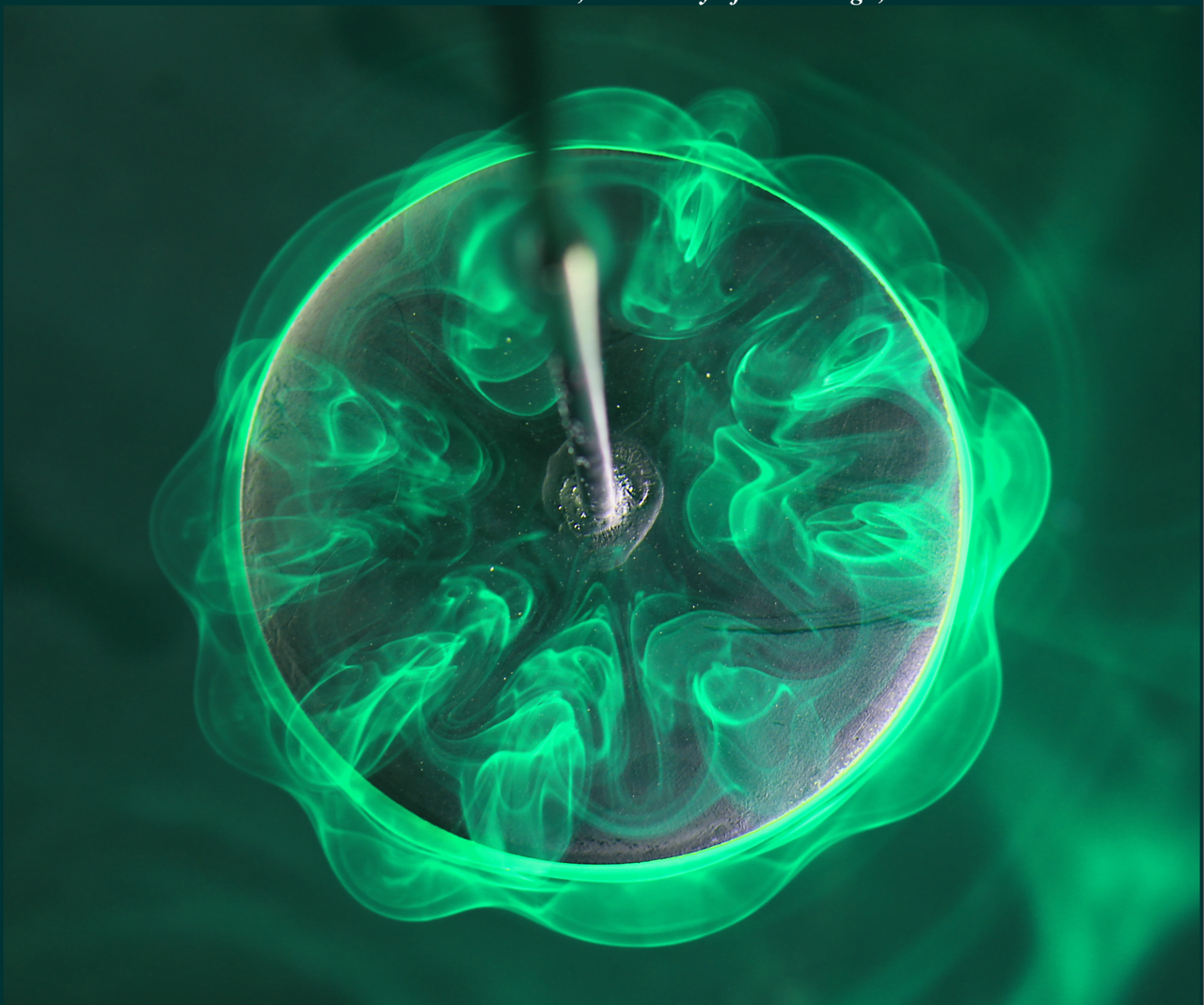
Breaking Down Vortices
Vortex rings are ubiquitous in nature, showing up in droplet impacts, in propulsion, and even in volcanic eruptions. Understanding the interaction and breakdown of multiple vortices with one another is therefore key. The image above shows a circular disk that’s being oscillated up and down (in and out of the page). As the disk moves and changes direction, it generates vortices that interact with one another. Here some of those interactions are visualized with fluorescent dye. The overlapping vortices form complex and beautiful shapes on their way to breakdown. (Image credit: J. Deng et al., poster, paper)

Bursting Droplets
Mixing multiple fluids can often lead to surprising and mesmerizing effects, whether it’s droplets that dance or tears along the walls of a wine glass. A recent paper highlights another such mixture-driven instability – the bursting of a water-alcohol droplet deposited on an oil bath. The Lutetium Project tackles the physics behind this colorful burst in the short video above. The behavior is driven by the quick evaporation rate of alcohol in the droplet and the way this changing chemical concentration affects surface tension in the droplet. Alcohol evaporates more quickly from the edges of the drop, creating a region of higher surface tension around the edge. This pulls fluid to the rim of the drop, where it breaks up into droplets that get pulled outward as the inner drop shrinks.
The oil bath plays an important role in the instability, too. Without it, friction between the drop and a wall is too high for the droplet to “burst”. A thick layer of oil acts as a lubricant, allowing the escaping satellite drops to speed away. (Video and image credit: The Lutetium Project; research credit: L. Keiser et al.; submitted by G. Durey)


Breaking the Wave Speed Limit
Whirligig beetles are small surface swimming insects. As they race across the water surface, they create both visible and unnoticeable waves on the water. These waves are the result of both surface tension and gravity. Typically, it’s the wavelength of the gravity waves that limit a swimmer or boat’s speed. When the wavelength of the gravity waves a swimmer creates meets the size of the swimmer, the waves generated ahead of the swimmer start to reinforce the waves forming at the back of the swimmer. This traps the swimmer (or boat) in a trough between its bow and stern waves and limits the max speed of the swimmer since overcoming this critical hull speed requires excessive amounts of power.
The tiny whirligig beetle overcomes this natural speed limit cleverly. It is smaller than the shortest possible gravity wave in water. Thus, it can never be trapped between its bow and stern waves! This allows the tiny swimmer to zip across the water’s surface at speeds above 0.5 m/s. That’s over 30 beetle body lengths per second! (Image credit: H. L. Drake, source; research credit: V. Tucker; submitted by Marc A.)

Supporting Bubbles
Surface tension holds small droplets in a partial sphere known as a spherical cap. But when droplets become larger, they flatten out into puddles due to the influence of gravity. In contrast, soap bubbles remain spherical to much larger sizes. The bubble pictured above, for example, is more than 1 meter in radius and nearly 1 meter in height.
There is a maximum height for a soap bubble, though, and it’s set by the physical chemistry of the surfactants used in the soap. To support itself, the bubble requires a difference in surface tension between the top and bottom of the bubble. A higher surface tension is necessary at the top of the bubble to help prevent fluid from draining away. The difference in surface tension between the top and bottom of the bubble can never be greater than the difference in surface tension between pure water and the soap mixture – thus those values set a maximum height for a bubble. The researchers found their bubbles maxed out at a height of about 2 meters, consistent with their theoretical predictions. (Image credit: C. Cohen et al.; via freshphotons)

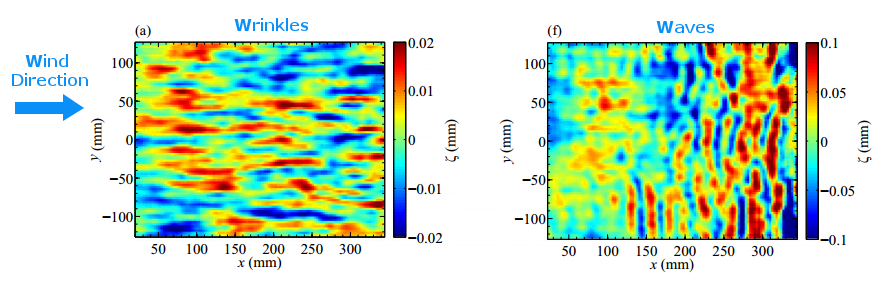
Wrinkling Winds

If you’ve ever sat out on a lake and just watched the water’s surface, you’ve probably noticed how complex and variable it looks. There may be waves that rock your kayak but there are smaller variations, too, like little ripples or even tiny wrinkles that appear on the surface. Much of this activity comes from wind blowing across the water. When the wind exceeds a critical speed, waves form. They generally travel in lines that are aligned perpendicular to the wind (lower right). But what happens when the wind is below the critical speed?
A recent study looked at just this question. By blowing air across the surface of different liquids and observing variations in the surface height as small as 2 micrometers, the researchers were able to measure tiny wrinkles on the water’s surface (lower left) when the wind speed was small. The size and shape of the wrinkles actually corresponds to structures in the turbulent air flow over the water! For fluids like water, there’s a smooth transition from wrinkles to waves as the wind speed increases, so both may be visible at the same time. For higher viscosity fluids, the switch from one to the other is more abrupt. (Image credits: water – M. Soveran; figure – A. Paquier et al. w/ annotations added in blue; research credit: A. Paquier et al.)

Aerodynamic Leidenfrost Effect

If you place a droplet on a surface much hotter than its boiling point, that droplet will skitter and float almost frictionlessly across the surface on a thin layer of its own vapor. This is what is known as the Leidenfrost effect. But you don’t have to heat a surface to get this behavior. There’s also an aerodynamic Leidenfrost effect, shown above, when the surface is moving. As the surface moves, it drags a layer of air along with it, and that layer of air is capable of keeping droplets aloft indefinitely. The thickness of the air layer depends on speed; the faster the plate moves, the thicker the air layer underneath droplets. The aerodynamic forces generated are large enough to drive a droplet up an incline against the force of gravity (bottom image). (Image credit: animation – M. Saito et al., source; chronophotograph – A. Gautheir et al., pdf)

Vortex Impact

When a vortex ring impacts a solid wall (or a mirrored vortex ring), it expands and quickly breaks up. The animations above show something a little different: what happens when a vortex ring hits a water-air interface. As seen in the side view (top image), the vortex starts to expand, but its shear at the interface generates a stream of smaller vortices that disrupt the larger vortex. (They even look like a little string of Kelvin-Helmholtz vortices!) When viewed from above (bottom image), the vortex ring impact and breakdown look even more complicated. Mushroom-like structures get spat out the sides as those secondary vortices form, and the entire structure quickly breaks up into utter turbulence. There’s some remarkable visual similarities between this situation and some we’ve seen before, like a sphere meeting a wall and drop hitting a pool. (Image credit: A. Benusiglio et al., source)